SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-1B: Atomic Structure: Rutherford's Gold Foil Experiment
Description
In this activity you will explore the methodology and results of Rutherford's famous gold foil experiment that helped lead to our current understanding of the structure of an atom.
Page content
A. Introduction
Welcome to SCH4U! We are going to start the course by exploring the fundamental building block of all matter - the atom. All solids, liquids and gasses in the entire universe are made up of individual atoms, all of which share the same basic structure. The atom itself is made up miniscule building blocks organized in a very specific way. This lesson will outline some of the important experiments that helped us understand the structure of the atom. You will remember from Grade 11, the atom is composed of 3 main building blocks – Protons (positive charge), Neutrons (no charge) and Electrons (negative charge).
The main lesson from this activity is that protons are concentrated in the tiny nucleus of an atom while electrons can be found outside the nucleus, with mostly empty space occupying the area between the two. It is important to remember that our understanding of the atom came through extensive experimentation and observation - 2 of the fundamental properties of all science.
By the end of the next few lessons, you'll know how we came go from plum pudding to orbiting planets, to quantum mechanics. Here we go!
B. Name That Shape - The Game
Our knowledge of the structure of atoms has come a long way from earlier, simple models. Through experimentation and observation a very important scientist named Ernest Rutherford helped to advance our understanding of the structure of the atom.
C. Rutherford's Gold Foil Experiment Simulator
Early theories on the structure of the atom pictured atoms as blobs of positive charge with negatively charged electrons scattered throughout, this was known as the "Plum Pudding Model". In 1911, Ernest Rutherford designed an experiment known as the "Gold Foil Experiment" to test if this model was correct. Your next task will be to try out a simulation of Rutherford's experiment to help you understand how his observations led to the advancement of the model of the atom.
For this task you will need the following:
- Open this file , and follow the instructions included.
- Click here to open the Rutherford Gold Foil Experiment Simulator to begin the simulation.
- Answers to the handout can be found in the check up time section of this lesson.
D. Rutherford's Gold Foil Experiment: Explanation
In 1911, the generally accepted model of the atom pictured it as a blob of positive charge with negatively charged electrons scattered throughout. Ernest Rutherford designed an experiment to test this theory. In his experiment, a beam of positively
charged alpha particles was fired at a thin sheet of gold foil. Alpha particles are positively charged, extremely tiny particles, made up of just 2 protons and 2 electrons and the gold foil was made up of gold atoms bound to each other. By observing the path of the positively charged
alpha particles as they passed through the gold foil, Rutherford believed he could uncover the mystery of the atom's structure. His experiment was very similar to the game you played at the beginning of this lecture where you tried to determine the
shape of an unknown object by observing how fired particles interacted with the unknown
. A description of Rutherford's experiment follows:
Experimental Design: a stream of positively charged alpha particles are fired at a thin sheet of gold foil, the path
of the alpha particles was observed by surrounding the apparatus with a surface that detects alpha particles. See the diagram below.
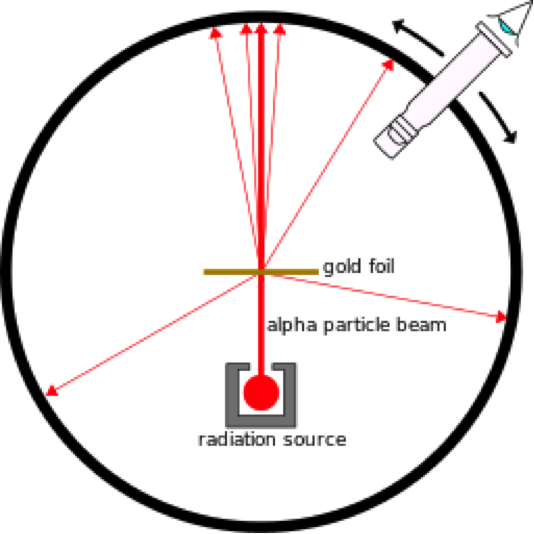 .Source: Kurzon (Own work) [CC-BY-SA-3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons
.Source: Kurzon (Own work) [CC-BY-SA-3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons
Hypothesis: If the plum pudding model is correct, positively charged Alpha particles should rip through the atoms of the gold foil like a bullet passing through paper, as you observed in the simulation above when you clicked on the "Plum Pudding Atom" tab.
Rutherford's Observations: Alpha particles were deflected in all directions when fired at a sheet of gold foil (as you saw in the simulation above when you clicked on the "Rutherford Atom" tab), others passed right through without breaking the foil at all.
Conclusions:
- Some alpha particles were deflected because they were repelled by an area of dense positive charge within the gold atoms (the nucleus). This debunked the theory that atoms were made up of a uniform blob of positive charge.
- Some alpha particles passed right through the atoms of the gold foil because there were large areas of empty space within each atom, between the dense nucleus and the surrounding electrons.
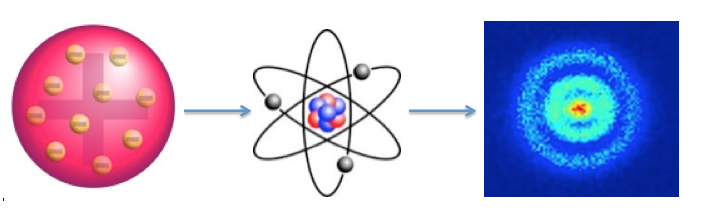
As a result of Rutherford's experiment, our picture of the atom evolved from:
 to this
to this 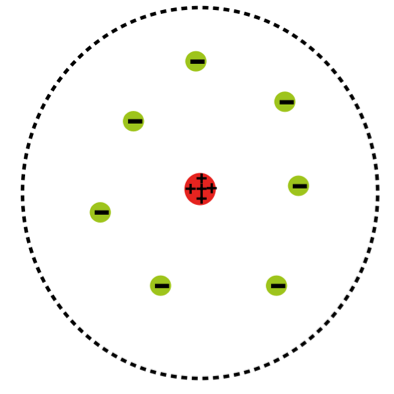
The following animation further sums up Rutherford's gold foil experiment:
Source: CBSE Youtube
Test your knowledge:
Question 1-1B-1) Positively charged alpha particles were deflected when fired at the gold foil in Rutherford's experiment because:
a) The gold atoms are stronger than the alpha particles
b) The alpha particles were repelled by the electrons of the gold atoms
c) The alpha particles were repelled by the concentrated area of protons in the nucleus
d) The alpha particles were repelled by randomly scattered protons in the gold atoms
Click for answer 1-1B:1
E. Summary:
- Early models of the atom pictured them as blobs of positive charge with negatively charged electrons scattered throughout
- In Rutherford's experiment, positively charged alpha particles were deflected when passing through a thin sheet of gold foil
- Rutherford's experiment led to the understanding that the positive charge of an atom is concentrated in a tiny space at the center of the atom, known as the nucleus.
