SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-1C: Atomic Structure: Niels Bohr and Electron Orbits
Description
In this activity you will learn about experimental results that helped lead to our current understanding of how electrons are arranged within an atom.
Page content
A. Hydrogen Emission Spectrum
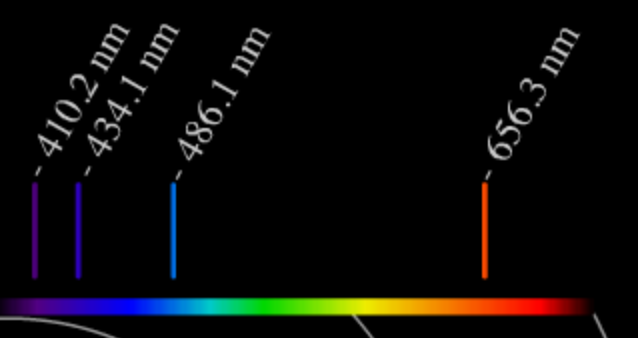
From Rutherford's experiments,it was now known that the atom had a dense area of concentrated positive charge in the center, but we still did not have the full picture of how atoms are structured. The next step in determining the structure of the atom was figuring out where the electrons belong. It was the interpretation of the emission spectrum of hydrogen by Niels Bohr that provided the evidence for atomic orbitals. An emission spectrum is the unique pattern of lines produced from pure samples of both elements and compounds when they are exposed to a jolt of energy in the form of a spark. Following the spark scientists observed, that the sample would emit energy in the form of light, and if this light was passed through a prism, the unique spectrum that you see below would be produced. Scientists were baffled as to why only certain lines would appear for certain samples and not the entire continuous spectrum seen at the top of the image. It took the brilliant mind of Niels Bohr to interpret these observations to help us understand the line spectrum for hydrogen and in the process develop the structural model of the atom beyond Rutherford.

B. Bohr's Model of the Atom
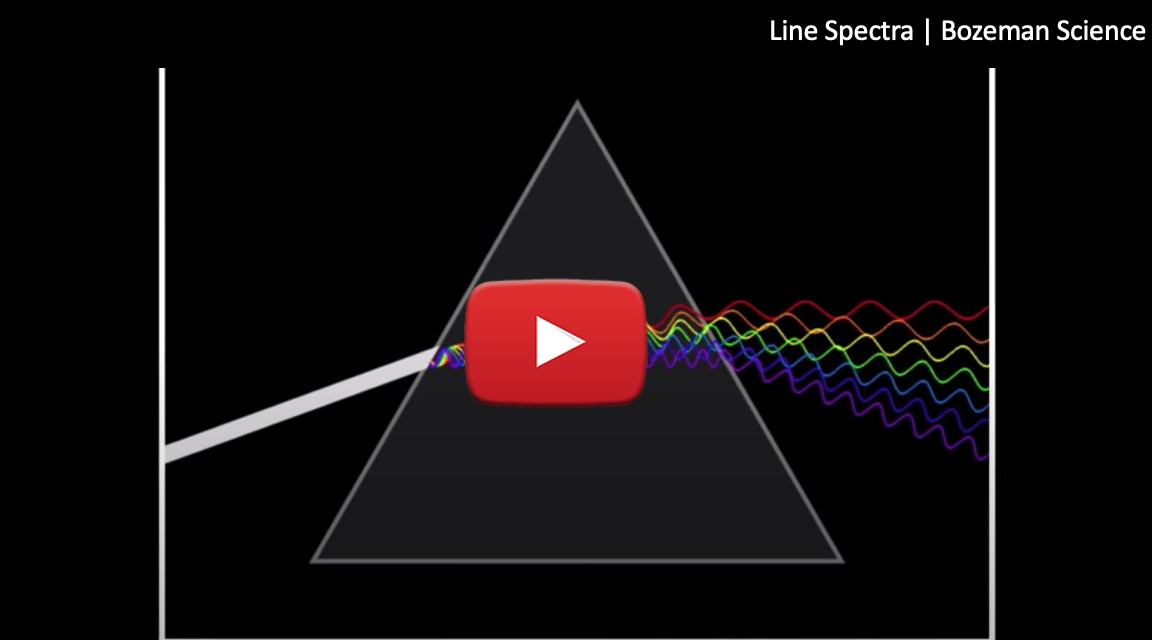
Bohr wanted to know why hydrogen only produced specific lines in its emission spectrum. Watch the video below to
how the Hydrogen emission spectrum is produced and at the end see how Bohr interpreted the results.
Source: Bozeman Science
To summarize the animation: Bohr's theory was that the electrons in the Hydrogen atom were confined to specific levels or orbits. When the spark was applied to the sample of hydrogen gas, electrons in the hydrogen atoms jumped from lower energy levels to higher energy levels. It turns out that electrons prefer to be in the lowest possible energy orbital, which is known as the ground state.
Once the electrons jumped up, their tendency was to drop back down to their ground state. When the electrons dropped to their ground state, energy was emitted from the atom, and this energy was released
in the form of a photon. The higher energy level an electron drops from, the higher the energy of the photon is released.
These photons are what produced the unique lines of the hydrogen emission spectrum. Different colours are produced depending on how many energy levels the electron dropped from. Bohr theorized that the individual lines produced corresponded to the
specific amount of energy released from an atom when an electron dropped down from a higher energy level to a lower energy level. He called these energy levels orbits and theorized that electrons circled the nucleus in the same way that planets orbit
around the sun. Each of these orbits or energy levels was given a number (n) starting at n = 1, as you move farther away from the nucleus, the value of n increases. A key part of Bohr's conclusion was that electrons cannot
exist anywhere in between the defined energy levels. Thus the n-values can only be represented as whole numbers, there is no n = 1.5 for example.
Niels Bohr's contributions to the structure of the atom led us from Rutherford's model of a nucleus surrounded by randomly placed electrons, to Bohr's model of a nucleus surrounded by electrons traveling in planetary-like orbits.
 --------->
--------->

In summary: From Bohr's observations, we came to understand that electrons typically reside in the lowest energy orbit possible, this is called the ground state. When these electrons are excited, by the addition of energy, they can jump up to higher energy orbits. When these electrons drop back down to their ground state (the lowest energy level orbit) they emit energy in the form of a photon. This photon is what produces the unique lines of the hydrogen emission spectrum.
The following summarizes how Bohr's theory helps explain the emission spectra observed by scientists:
Niels Bohr proposed the first successful model of the hydrogen atom in 1913. It was a simple planetary model like the Earth orbiting around the Sun: a light negative electron in a circular orbit around a heavy, nearly stationary, positive proton. Instead of a gravitational force, an attractive electric force holds the electron in its orbit.
In order for the model to work, Bohr had to make a radical proposal... the electron can only occupy certain stable orbits with prescribed energies.
The electron orbit closest to the proton has the lowest energy. Energy is needed to 'pull' or 'lift' the electron away from the proton, so orbits with larger radii have higher energies.
If an electron is to jump from a lower orbit to a higher orbit it will require an energy source. The energy source is light. The electron can absorb a photon of light and use all of that photon's energy to 'jump' form a lower energy orbit up to a higher energy orbit.
Conversely, when the electron 'falls', it will emit all of its extra energy as a corresponding photon of light. It is simply conservation of energy. The energy of the photon is completely absorbed by the electron allowing it to jump to the next energy level.
Only particular photons can give their energy to the electron - that photon's energy must exactly match the energy required for the electron to jump.
You may remember that there is an inverse relationship between energy and wavelength.
So we say that the electron absorbs a particular wavelength of color of light in order to gain the energy required to jump energy levels.
Let's look at an example... Say that the electron is in the second energy orbit, it can absorb the energy of a red photon and jump to the third energy orbit. When it falls back to the second level again, it must emit a red photon such that the total amount of energy remains the same.
Similarly, an incoming blue photon will excite the electron into the fourth energy orbit. If the electron falls back it will emit a blue photon.
The result: the hydrogen atom can absorb photons of light with particular wavelengths or energy.
It can also emit photons of light with those same particular wavelengths.
So for an excited hydrogen atom, we could expect to see particular wavelengths of light produced as the electrons fall back and emit photons of light corresponding to those energies. This 'emission spectra' of Hydrogen is exactly what we see.
Notice that only particular wavelengths of light are emitted - wavelengths that correspond to the required amount of energy to jump between levels. Bohr was able to calculate the exact wavelengths of the spectral lines of hydrogen in the visible region of the electromagnetic spectrum.
What does this have to do with the Sun?
The dark lines we have seen in the Sun's spectra are direct evidence that hydrogen atoms are absorbing energy from the sunlight. They match line for line the emission spectra of hydrogen produced in the laboratory. These spectral lines become a valuable tool that we can use to learn about the electronic structure of the atom.
While to Bohr's model was extremely successful at predicting the precise wavelengths of light in the spectra of hydrogen, it failed to predict the light spectra of any atom with more than one electron. The success would have to wait for the birth of 'Quantum Mechanics.'
Test your understanding:
Question 1-1C-1 True or false: An electron that dropped from the n = 6 orbital to the n = 2 orbital would produce the same line in an emission spectrum as an electron dropping from the n = 4 level to the n = 2 level.
Check your answer 1-1C:1
For another look on the topic of Bohr's model of the hydrogen atom, click here.
Summary
- Bohr's model had electrons orbit around the nucleus of an atom in confined orbitals similar to planets orbiting the sun
- Electrons can move between the orbitals, jumping up when energy is added and jumping down by emitting energy
- When an electron drops down from a higher orbital, energy is given off in the form of a photon
- The unique line spectrum produced by hydrogen is a result of energy being released in the form of photons when excited electrons return to their ground state
- The different lines of the hydrogen emission spectrum are the result of electrons dropping down from different orbitals