SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-2A: Shells & Subshells
Description
In this activity you will further explore the structure of the atom by learning about how electrons are organized into different shells and subshells surrounding the nucleus.
Page content
In the last lesson we learned that Bohr concluded that the individual bands that were observed from the hydrogen emission spectrum were caused by electrons dropping from a higher energy level (orbit) to its lowest energy level (ground state). When the electrons drop down a level, they emit energy in the form of a photon and these photons correspond to the bands of the emission spectrum. This following brief video introduces some of the concepts you will learn about in the next few learning activities.
A. Shells and Subshells (Orbitals)
Bohr imagined the energy levels as circular orbits that the electrons could be found in where electrons circled the nucleus in the same way that planets orbit the sun. However, Bohr’s model was an incomplete picture of the way that atoms are structured, how electrons behave and what electron orbits actually look like. Instead of orbiting the nucleus in a circular path, it turns out that individual electrons are actually randomly moving about the nucleus in defined 3-D areas called orbitals.
Bohr’s model proposed that electrons exist in orbits, represented by the letter n, with higher values for n representing orbits of higher energy and further away from the nucleus. This model can be represented in two ways:
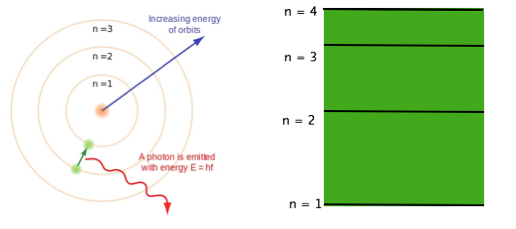
The true picture is a little more complicated. The energy levels represented by n do in fact exist, and are referred to as shells. These shells are further divided into subshells, which represent the actual 3-D orbitals where electrons are likely to be found. Each orbital has a unique shape and orientation with differing energy values. As you move up to higher energy levels (shells), more orbitals are found in each shell. For example, the n = 1 shell has only one orbital, while the n = 3 shell has 9 orbitals. Every shell can have n2 orbitals, and every orbital can have 2 electrons. Therefore, every shell can have 2n2 electrons.
Our diagram of energy levels now changes to show both the shells and subshells. Note that as you go up in values of n, the total number of orbitals also increases.
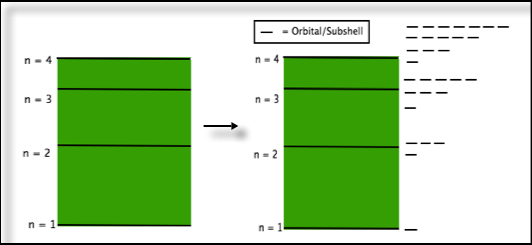
B. Summary
- n = shell, higher n means higher energy, further away from the nucleus
- each shell contains subshells
- subshell = orbital
- Every orbital can have 2 electrons
- Every shell can have n2 orbitals
- Every shell can have 2n2 electrons
Test your knowledge:
Question 1: For n = 5 how many orbitals are found in that shell?
a) 5
b) 10
c) 50
d) 25
Check your answers with this answer key: Answer: 1-2A:1
- office.jpg
- Screen Shot 2014-09-24 at 1.36.41 PM.png
- Screen Shot 2014-09-24 at 1.38.50 PM.png
- Screen Shot 2014-09-24 at 1.40.23 PM.png
- Screen Shot 2014-09-24 at 1.41.02 PM.png
- Screen Shot 2014-09-25 at 1.13.23 PM.png
- Screen Shot 2014-09-25 at 11.49.16 AM.png
- Screen Shot 2014-09-25 at 11.49.35 AM.png
- Single_electron_orbitals1.jpg
- Understanding the Atom.mp4
