SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-2B: Quantum Numbers Part 1: Primary & Secondary Quantum Numbers
Description
In this activity you will explore how quantum numbers can be used to describe the positions of electrons within an atom.
Page content
Lesson Key Points:
- Orbitals are 3-D shapes where electrons are located
- Orbitals exist within shells
- Multiple orbitals can reside within a shell
- Each orbital can hold 2 electrons
- Orbitals are described by their shell and orbital shape (ie 3p = a p-orbital in the 3rd shell)
- n = 1 shell has 1 s-orbital
- n = 2 shell has 1 s-orbital and 3 p-orbitals
- n = 3 shell has 1 s-orbital, 3 p-orbitals and 5 d-orbitals
- n = 4 shell has 1 s-orbital, 3 p-orbitals, 5 d-orbitals and 7 f-orbitals
A. Introduction: Quantum Numbers
In this lesson you will learn more about the structure of atomic orbitals using quantum numbers. The quantum numbers act as a system to describe the characteristics of a given orbital including its distance from the nucleus, shape, orientation and the direction an electron spins. This lesson introduces the first 2 quantum numbers: n & ℓ. It is important to understand that within a shell, multiple orbitals can exist. So, on to the quantum model of the atom....here we go!
Recall the last lesson where we found out that atomic orbitals or subshells can be found within the shells that are described by n. From this value for n we were able to deduce how many orbitals exist in that shell (n2) and how many electrons can reside there (2n2). The next step in understanding atomic structure is learning the system that is used by scientists to describe individual orbitals and electrons, which leads us to the quantum model of the atom.
B. Basics of the Quantum Model of the Atom:
The quantum model of the atom describes an electron within an orbital using 4 characteristics. Each quantum number describes a specific characteristic: distance from the nucleus, shape of the orbital, orientation of the orbital in space and the spin of an individual electron.
- The 4 quantum numbers used to describe an electron are as follows:
- Principal Quantum Number (n): Describes the energy level an orbital is found in.
- Secondary Quantum Number (l): Describes the shape of an orbital (subshell)
- Magnetic Quantum Number (ml): describes the orientation of the orbital in space (ie. what axis does it lie on)
- Spin Quantum Number (ms): describes the direction an electron is spinning
C. The Principal Quantum Number: Shells
1. The principal Quantum Number (n) energy level (shell) an electron is found in. The values of (n) can be any positive whole number beginning at 1. The higher the value of n, the further the distance from the nucleus a shell is. It turns out that the primary quantum number (n) can help us determine the other possible quantum numbers for a given electron.
In the diagram below, we can see that n relates to a shell, inside which orbitals (also known as subshells) can be found. Within a given shell, multiple orbitals can exist. The higher the value of n, the greater the number of orbitals. In fact there is a formula to determine how many orbitals exist within a shell: n2, and since we know each orbital can only hold 2 electrons, every shell can hold 2n2 electrons
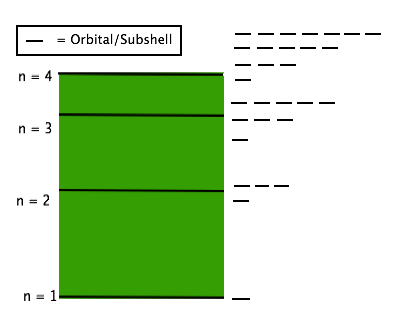
Summary:
Principal Quantum Number (n):
- distance of a shell from the nucleus
- whole numbers ranging from 1 to infinity
- orbitals exist within shells. Orbitals are sometimes referred to as subshells
- n2 orbitals exist inside every shell (n)
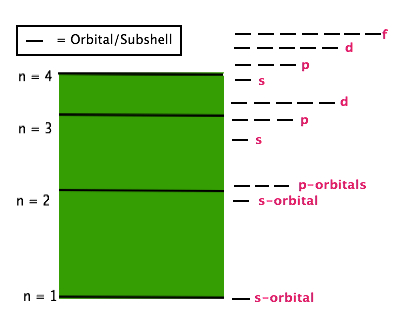
D. Secondary Quantum Number (ℓ): Subshells/Orbitals
The secondary Quantum Number (ℓ) describes the name and shape of an orbital. Each value of ℓ corresponds to a specific orbital shape with a specific name.
Values of (ℓ) are whole numbers beginning with 0, with each number corresponding to a given name.
ℓ = 0 orbital is called an s-orbital and is sphere shaped
ℓ = 1 orbital is called a p-orbital and is dumbbell shaped
ℓ= 2 orbital is called a d-orbital and has 2 different shapes
ℓ= 3 orbital is called an f-orbital and has many different shapes
For every shell, described by n, the values of ℓ range from 0 to (n - 1). For example when n = 3, the range of values of ℓ are 0 to (3-1) = 0 to 2. Therefore the n = 3 shell can have orbitals with ℓ-values of 0, 1 and 2. Another way of saying this is that the n = 3 shell contains 3 different types of orbitals: s-orbitals, p-orbitals and d-orbitals
Summary:
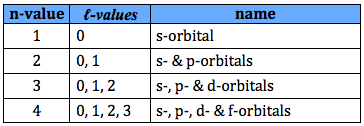
- ℓ-values range from 0 to (n-1)
- When n = 1, ℓ-values range from 0 to 0, therefore the n = 1 shell can only have s-orbitals
- When n = 2, ℓ-values range from 0 to 1, therefore the n = 2 shell can have s- and p-orbitals
- When n = 3, ℓ-values range from 0 to 2, therefore the n = 3 shell can have s-, p- and d-orbitals
- When n = 4, ℓ-values range from 0 to 3, therefore the n = 4 shell can have s-, p-, d- and f-orbitals
Check your understanding
Question 1-2:B-1 An orbital has a ℓ-value of 2, what type of orbital is this?
a) s-orbital
b) p-orbital
c) d-orbital
d) f-orbital
Answer 1-2:B:1
Question 1-2:B-2: For a shell with n = 6, what are the possible range of values for ℓ?
a) 0 to 6
b) 0 to 7
c) 0 to 5
d) 0 to 0
Answer 1-2:B:2
Question 1-2:B-3: How many different types of orbitals exist in the n = 4 shell?
a) 4
b) 3
c) 2
d) 1
Answer 1-2:B:3