SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-2C: Quantum Numbers Part 2: Magnetic & Spin Quantum Numbers
Description
In this activity you will further your knowledge of quantum numbers as you discover how electrons can be described by the orientation of their orbitals and the direction of their spin.
Page content
A. Introduction:
In the last activity, we learned about the first 2 quantum numbers n and ℓ. In this lesson we will learn about the last 2 quantum numbers which tell us how many of each type of orbital exists within a shell (ml) as well as the spin of a given electron within a shell (ms).
B. The Magnetic Quantum Number (ml)
Magnetic Quantum Number (ml) describes the orientation of an orbital in space (ie what plane does it lie in on a 3-D axis). This number tells us how many of a particular type of orbital are in each shell (for example there are 3 p-orbitals in every shell from n = 2 up). Values for ml range from –ℓ to +ℓ , including 0. For example if the secondary quantum number (ℓ) = 2, the (ml) has values that range from -2 to +2, therefore there are 5 different d-orbitals in a given shell ( -2, -1, 0, 1, 2).
As summarized in the chart below, when ℓ = 2, this corresponds to the d-orbital, which has 5 different possible orientations (look in the last column, 3rd row), which are described by (ml)
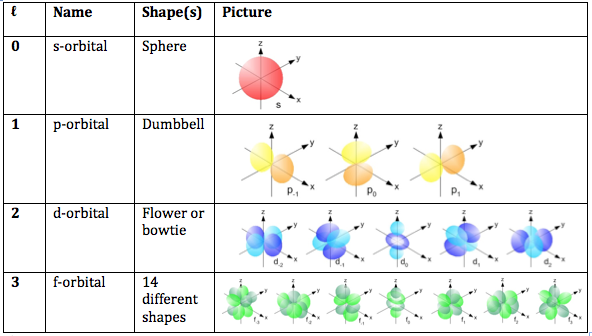
When ℓ = 0: (ml) values range from 0 to 0; therefore there is 1 s-orbital in every shell
When ℓ = 1:(ml) values range: -1, 0, 1; therefore there are 3 p-orbitals in every shell from n = 2 and above
When ℓ = 2:(ml) values range: -2, -1, 0, 1, 2: therefore there are 5 d-orbitals in every shell from n = 3 and above
When ℓ = 3:(ml) values range from -3, -2, -1, 0, 1, 2, 3: therefore there are 7 f-orbitals in every shell from n = 4 and above
Check your understanding:
Question 1-2C-1: When ℓ = 3, how many ml values exist for these orbitals?
a) 3
b) 6
c) 7
d) 4
Answer 1-2C:1
Question 1-2C-2: How many p-orbitals exist in any shell in any shell above the n = 1 energy level?
a) 1
b) 2
c) 3
d) 4
Answer 1-2C:2
To summarize everything we have learned so far in one tidy table:
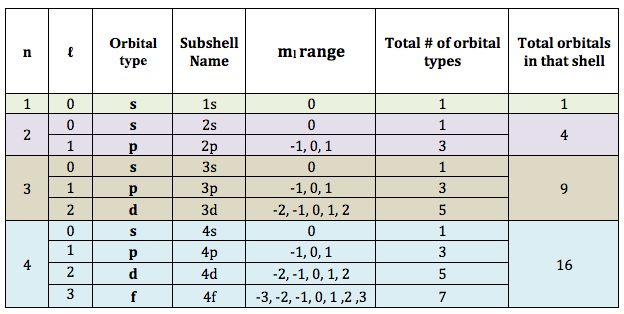
Example
Looking at the table above, we can see that for example, when n = 3:
-
- ℓ-values range from 0 to 2,
- this shell would have 1 s-orbital, 3 p-orbitals and 5 d-orbitals
- the shell would have 9 orbitals total and could contain up to 18 total electrons (2 per orbital)
Remember we said earlier, every shell (n) can have n2 orbitals and every orbital can hold 2 electrons, therefore every shell (n) can hold 2n2 electrons
C. The Spin Quantum Number (ms)
4. The Spin Quantum Number (ms) describes the direction a given electron is spinning. Electrons have “spin” values associated with them that represent the direction that a given electron spins. For the ms number there are only 2 possible values +1/2 and -1/2. An arrow pointing up or down can also represent the spin of an electron.
As we learned earlier each orbital can only contain 2 electrons, and it turns out that if an orbital has 2 electrons they must spin in opposite directions, therefore, the Pauli Exclusion Principle states: no 2 electrons in the same atom can have the same 4 quantum numbers.
To recap, we can describe an electron in an orbital using 4 quantum numbers:
- Principal Quantum Number (n): Describes the distance of a shell from the nucleus
- Secondary Quantum Number (ℓ): Describes the shape of an orbital (subshell)
- Magnetic Quantum Number (ml): describes the orientation of the orbital in space (ie. what axis does it lie on)
- Spin Quantum Number (ms): describes the direction an electron is spinning
Check your understanding:
Question 1-2C-3 How many possible orientations of the f-orbital exist?
a) 3
b) 7
c) 4
d) 1
Check Answer 1-2C:3
Question 1-2C-4 How many electrons can reside in the n = 2 shell?
a) 8
b) 4
c) 16
d) 2
Check Answer 1-2C:4
D. Summary
From these 2 sections on quantum numbers the most important information to remember is the following:
- n = 1 shell has 1 s-orbital
- n = 2 shell has 1 s-orbital and 3 p-orbitals
- n = 3 shell has 1 s-orbital, 3 p-orbitals and 5 d-orbitals
- n = 4 shell has 1 s-orbital, 3 p-orbitals, 5 d-orbitals and 7 f-orbitals
- Each shell can have n2 orbitals and hold 2n2 electrons
- Each orbital can hold 2 electrons, with opposite spin values
- The Pauli Exclusion Principle tells us that 2 electrons in the same orbital cannot have the same spin value.
