SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-2D: Summary
This page is part of the content downloaded from 1-2D: Summary on Monday, 3 March 2025, 9:21 AM. Note that some content and any files larger than 50 MB are not downloaded.
Description
Summary
Page content
Summary
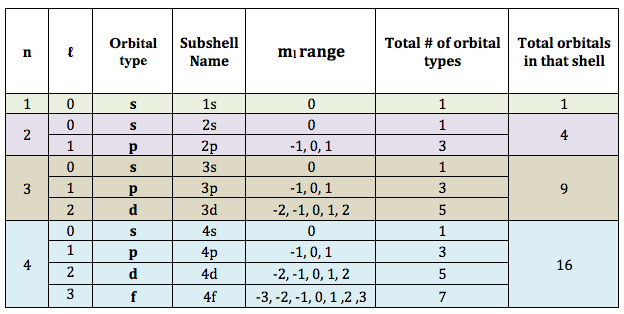
- n-values: Describe an orbital shell
- Each shell contains subshells
- Subshell = orbital
- Every orbital can have 2 electrons
- Every shell can have n2 orbitals
- Every shell can have 2n2 electrons
- The 4 quantum numbers used to describe an electron are as follows:
- Principal Quantum Number (n): Describes the distance of a shell from the nucleus
- Secondary Quantum Number (l): Describes the shape of an orbital (subshell)
- Magnetic Quantum Number (ml): describes the orientation of the orbital in space (ie. what axis does it lie on)
- Spin Quantum Number (ms): describes the direction an electron is spinning
- n = 1 shell has 1 s-orbital
- n = 2 shell has 1 s-orbital and 3 p-orbitals
- n = 3 shell has 1 s-orbital, 3 p-orbitals and 5 d-orbitals
- n = 4 shell has 1 s-orbital, 3 p-orbitals, 5 d-orbitals and 7 f-orbitals
- The secondary Quantum Number (ℓ) describes the name and shape of an orbital
- ℓ-values range from 0 to (n-1)
- ℓ = 0: s-orbital
- ℓ = 1: p-orbital
- ℓ = 2: d-orbital
- ℓ = 3: f-orbital
- When ℓ = 0: (ml) values range from 0 to 0; therefore there is 1 s-orbital in every shell
- When ℓ = 1:(ml) values range: -1, 0, 1; therefore there are 3 p-orbitals in every shell from n = 2 and above
- When ℓ = 2:(ml) values range: -2, -1, 0, 1, 2: therefore there are 5 d-orbitals in every shell from n = 3 and above
- When ℓ = 3:(ml) values range from -3, -2, -1, 0, 1, 2, 3: therefore there are 7 f-orbitals in every shell from n = 4 and above
- ms values indicate the spin of an electron, values can only be -1/2 or +1/2
- 2 electrons in the same orbital must have opposite (ms) values
- The Pauli Exclusion Principle tells us that 2 electrons in the same orbital cannot have the same spin value.
The table below summarizes all the information you will need for the section on quantum numbers: