SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-3A: Electron Configurations & Orbital Diagrams
Description
In this activity you will explore how electrons are arranged withing an atom and how these arrangements can be represented using orbital diagrams.
Page content
A. Introduction
We have now learned that atomic orbitals have their own unique energy, shape, orientation and spin values associated with them. The next step in understanding atomic structure is exploring how the electrons in an atom are organized within orbitals. In this learning activity we will learn how to draw diagrams that show how the electrons in an atom are organized. The key points to remember from this lesson are:
- How to order atomic orbitals in terms of their energy levels
- How to apply the Aufbau Principle, Pauli Exclusion Principle and Hund's Rule
- How to draw orbital diagrams
- How to interpret electron configurations and orbital diagrams
Review
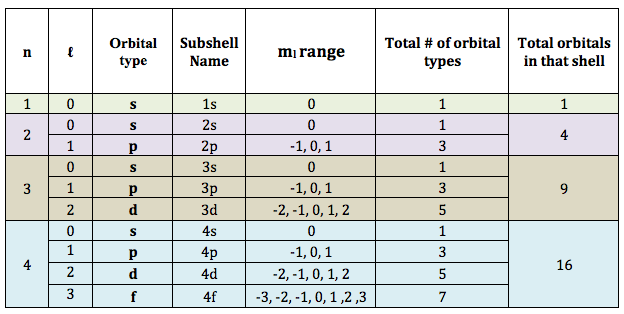
Remember this table from the last lesson, which tells us how many of each orbital are in each shell, so for example, there are 5 d-orbitals in the 3rd and 4th shell:
B. The Aufbau Principle
The Aufbau Principle teaches us how to organize the electrons in an atom and here we will learn the basic skills that will allow us to construct both simple and complex atoms.
The Aufbau Principle states that electrons are added to atomic orbitals starting with the lowest available energy orbital before filling up higher energy orbitals. Therefore, the first 2 electrons in any atom will fill the 1s orbital, the next 2 electrons will fill up the 2s orbital and so on. We learned earlier that n-values correspond to shells of increasing energy. It turns out that the orbitals within a shell have different energy values and that some orbitals in a lower shell can have higher energy values than orbitals in a higher shell (for example, the 5s orbital has a lower energy value than a 4d orbital). To help us order the orbitals in terms of energy levels scientists have created a chart called an Aufbau Diagram which you can see in the image below. To create an Aufbau Diagram use the following steps:
- Start at the top with the n = 1 shell and list the possible orbitals for that shell across the page, therefore the first line would only have the 1s orbital because that is the only orbital that exists in that shell
- As you move down the page, the n-values increase by one, and again you list the orbitals for that level, so the second line has 2s and 2p orbitals
- Continue down to the n = 3 level, listing all the possible orbitals for that level, so the n = 3 level will have 3s, 3p and 3d orbtials
- For each subsequent level you only need to list up to the f-orbitals
- The last line in your Aufbau Diagram should have 7s, 7p, 7d and 7f orbitals listed
- Starting from the top, draw diagonal arrows as you see in the diagram below, these arrows indicate increasing energy levels, when you get to the end of an arrow, you move to the back of the next arrow down. For example after the 5s orbital you would move to the 4d orbital. This indicates that the 5s orbital is at a lower energy level than the 4d orbital
In an Aufbau Diagram, orbital energies can be determined by following the arrows. As you can see below, orbital energy levels are ordered as follows:
1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s < 5f < 6d < 7p < 6f < 7d < 7f
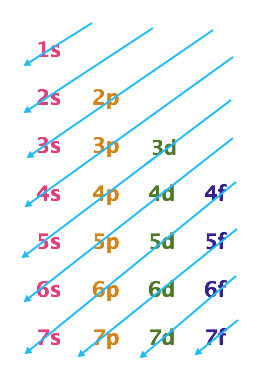
To summarize what the information above is telling us, when we are building up atoms electrons will fill up the orbitals in order of increasing energy and we will now put that information into use. As stated above, the Aufbau Principle says that electrons always fill the lowest energy orbital first.
Test your knowledge:
Question 1-3A-1: Which of the following atomic orbitals has the highest energy:
a) 4f
b) 4d
c) 5p
d) 6s
Answer 1-3A:1
C. Electron Configuration
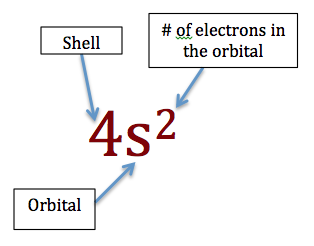
Scientists use a notational system to describe the electrons in an atom which tells us about the number of electrons in each orbital. For example Fluorine (which has 9 electrons) can be written as F: 1s22s22p5. When writing these configuration, we order the orbitals by energy, lowest to highest. For example, Nickel (Ni) would be written as: 1s22s22p63s23p64s23d8 (note that the 3d orbital is listed after 4s because it has a higher energy, therefore it would be filled up after the 4s orbital. See the image below for an explanation of the notation.
Test your knowledge:
You may need to use the Periodic Table of the Elements for this activity.
Question 1-3A-2: What atom does the following electronic configuration represent: 1s22s22p63s23p4?
a) Carbon (C)
b) Sulfur (S)
c) Aluminum (Al)
d) Chlorine (Cl)
Answer 1-3A:2
Question 1-3A-3: What does the electron configuration for Bromine (Br, atomic #35) look like?
a) 1s22s22p63s23p64s24p53d10
b) 1s22s22p63s23p64s23d104p5
c) 1s22s22p63s23p64s23d54p10
d) 1s22s22p63s23p64s24p5
Answer 1-3A:3
D. Orbital Diagrams
Orbital diagrams are used to show the organization of electrons in an atom. They can tell us which orbitals are occupied, how many electrons in each orbital and the spin of each of those electrons. In orbital diagrams, we use boxes to represent individual orbitals and arrows to represent individual electrons.
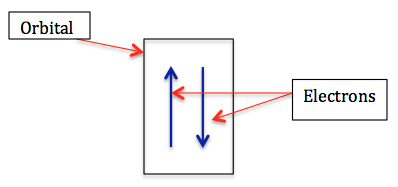 For these diagrams we need to apply the Pauli Exclusion Principle and the Aufbau Principle. We also need to apply a new rule, known as Hund's Rule, which states that the lowest energy state for an atom is the one with the maximum number of unpaired electrons. An unpaired electron means it is an electron by itself in an orbital. Another way of thinking about Hund's rule is that if the p-orbitals of a shell are empty, and you have 2 free electrons, the electrons will each fill one of the p-orbitals instead of pairing up in one.
For these diagrams we need to apply the Pauli Exclusion Principle and the Aufbau Principle. We also need to apply a new rule, known as Hund's Rule, which states that the lowest energy state for an atom is the one with the maximum number of unpaired electrons. An unpaired electron means it is an electron by itself in an orbital. Another way of thinking about Hund's rule is that if the p-orbitals of a shell are empty, and you have 2 free electrons, the electrons will each fill one of the p-orbitals instead of pairing up in one.
Remember from the last lesson: according to the Pauli Exclusion Principle, one orbital can only hold 2 electrons, and these electrons must have different spin values. We use up and down arrows to represent different spin values.
Now, lets put these 2 methods into practice and see how to use them:
Lets start with the easiest element first, hydrogen. Hydrogen has 1 electron, and according to the Aufbau Principle, this electron will occupy the orbital with the lowest energy which is the 1s orbital, so we can represent hydrogen like this:
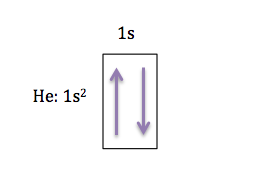
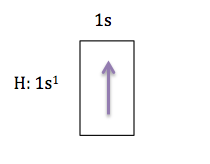 Moving across the periodic table, the next element is helium (He), which has 2 electrons, the orbital diagram for He would look like this:
Moving across the periodic table, the next element is helium (He), which has 2 electrons, the orbital diagram for He would look like this:
Moving across the the row, lets show the orbital diagrams from lithium all the way to Neon. As you can see these diagrams incorporate the Pauli Exclusion Principle, the Aufbau Principle and Hund's Rule.
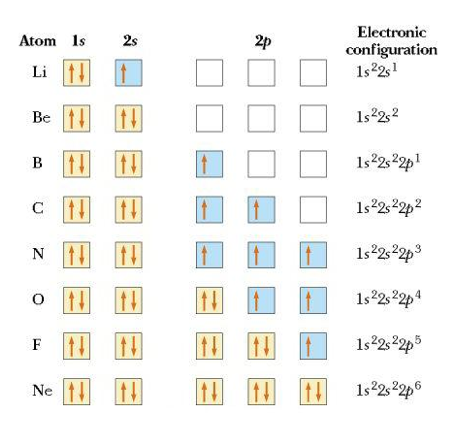
Lets examine the orbital diagram for Nitrogen (N) above. Nitrogen has 7 total electrons, the first 2 fill up the 1s orbital, the next 2 fill up the 2s orbital, for the next 3 electrons they each fill up their own 2p orbital. This is the application of Hund's rule, instead of 2 electrons pairing up in one of the 2p orbitals, they prefer to be alone if possible. Orbital diagrams can be written across a row as we see above or down a column, with the lowest energy orbitals at the bottom as seen in question 1-3A-4 below.
Test your knowledge:
For these questions, you may require the Periodic Table of the Elements
Question 1-3A-4: What atom does the following orbital diagram represent:
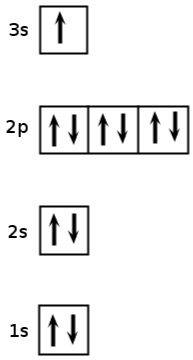
a) Neon (Ne)
b) Silicon (Si)
c) Sodium (Na)
d) Magnesium (Mg)
Answer 1-3A:4
E: Exception to Aufbau Rule
Some d and f block elements do not always follow the Aufbau Rule. Examples of these elements include: chromium, copper, and gold. These elements experience increased stability provided by the half-filled and completely filled d and f subshells. This increased stability is due lower electron repulsion in the orbitals and results in a lower overall energy of the atom. There are also smaller energy differences between the orbital at higher atomic number.
Let’s look at some examples of exceptions to the Aufbau rule:
Chromium
Based on the Aufbau Rule, the electronic configuration of chromium should be [Ar]3d44s2 . However, due to the exception, its electronic configuration is actually [Ar]3d54s1 .
Notice that the 3d orbital contains 1 additional electron to hold 5 electrons, which is exactly a half-filled shell. The 4s orbital now only has 1 electron. 3d is more stable with 5 electrons than with 4 electrons. In addition, the energy gap between 3d and 4s subshells is lower. The half filled sub-shells also feature lower electron-electron repulsions in the orbitals which increases the stability.
Copper
Based on the Aufbau rule, the electronic configuration of copper would be: [Ar]3d94s2. Instead, the actual configuration is [Ar]3d104s1. You will notice that the 3d subshell has 10 electrons instead of the expected 9. This is because a completely filled 3d orbital is more stable than when it has only 9 electrons.
Some other exceptions are silver, palladium molybdenum, rhodium, and platinum. Try predicting the electron configuration of these elements, knowing that they are exceptions to the Aufbau rule.
F. Summary
- The Aufbau Principle states that electrons fill up orbitals with the lowest energy first
- Aufbau Diagrams can be used to determine the relative energy of the atomic orbitals
- Electron configurations display how the electrons in an atom are organized
- Orbital Diagrams also display how electrons are organized in an atom
- Hund's Rule states that electrons will fill up an empty orbital before pairing up with another electron in an orbital with the same energy