SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-3C: Blocks of the Periodic Table
Description
In this learning activity you will explore how the periodic table can be organized into blocks based on their electron configurations and properties.
Page content
A. Blocks of the Periodic Table
The properties and characteristics of individual elements is highly related to their structure. Elements with similar electron configurations have similar properties and the periodic table is organized in such a way to group together elements with similar characteristics.
Having completed Lesson 1-3B, hopefully you were able to identify a pattern in how elements are organized in the periodic table. This lesson will highlight the key information relating to blocks of the periodic table and some of the common characteristics of elements within each block.
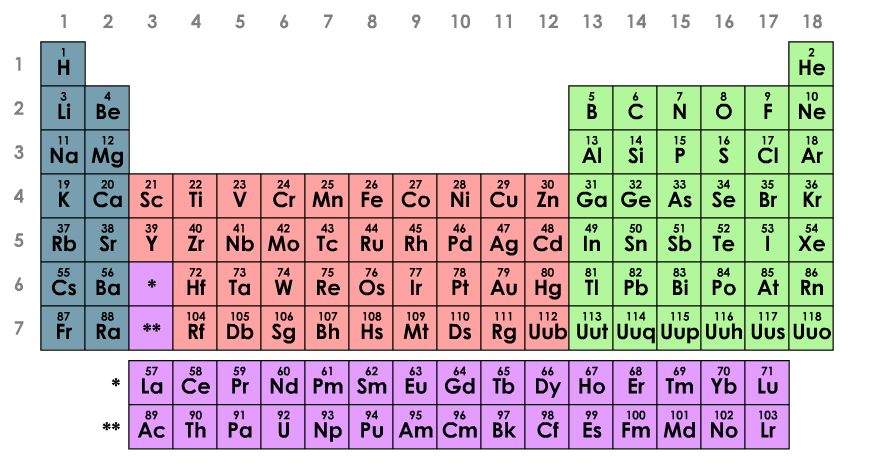
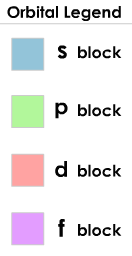
The Periodic Table can be viewed as a composition of 4 blocks: s-, p-, d- and f-blocks. The name of each block should be familiar to you by now, as the names relate to atomic orbitals. The blocks are organized in such a way that the name of the block indicates which orbital type is filled last for every element in that block. For example, Sc (Scandium), which is in the d-block has the electron configuration Sc: 1s22s22p63s23p64s23d1. (Remember the Aufbau principle states that electrons will fill the lowest energy orbital first, therefore the 3d orbital is filled before the 4p. Refer to an Aufbau Diagram to determine the order in which orbitals are filled). In fact we can use our knowledge of the blocks, and the structure of the periodic table to tell us exactly which orbital is the last to be filled with electrons for an atom.
B. Characteristics of the Blocks of the Periodic Table.
s-block:
-
- Made up of Alkali (Group 1) & Alkaline Metals (Group 2)
- Have outer electrons in s-orbitals
- Alkali Metals have 1 valence electron, Alkaline have 2 valence electrons
- Alkali Metals: Low melting and boiling points
- Alkaline Metals: Higher melting and boiling points
p-block:
-
- Have outer electrons in p-orbitals
- Made up of metals, metalloids & non-metals
- Melting and boiling points increase as you move down the group
d-block
-
- Have outer electrons in d-orbitals
- Made up of transition metals
- High melting and boiling points
f-block
-
- Have outer electrons in f-orbitals
- Made up of the rare earth metals
- High melting and boiling points
The key message from these blocks is that elements with similar electron configurations display similar characteristics. Even though the same members of a block may have vastly different atomic masses, number of protons and electrons, the fact that their outer electrons are organized in a similar way gives them very similar characteristics. The periodic table is organized to group those elements with similar electron configurations together.
C. Summary
- The periodic table of the elements can be divided up into 4 blocks: s block, p block, d block & f block
- The last orbital to be filled with electrons in an element is what determines the block it is in
- Elements within a block have similar characteristics
- Elements with similar electron configurations have similar qualities
- Electron configuration can affect specific characteristics including electronegativity, melting point and boiling point