SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-4A: Lewis Structures
Description
In this activity you will use your knowledge of valence electrons and the periodic table to learn how to represent atoms and molecules using Lewis structures.
Page content
A. Introduction
Gilbert Lewis was one of the pioneers of knowledge in the field of chemical bonding. His theories have helped shape many of the important concepts used throughout chemistry to this day. Specifically, his theory promoted the idea that atoms are most stable when they have a full set of valence electrons. This knowledge formed the basis of the octet rule, which states that many atoms are at their most stable when they are surrounded by 8 electrons in their valence shell (hydrogen and helium follow the duet rule, where they are most stable with 2 electrons in their valence shell). This idea that atoms are most stable with a full valence shell helped us to understand covalent bonding, whereby atoms share electrons between them to produce full valence shells in both sharing partners.
B. Valence Electrons
You should recall that valence electrons are the outermost electrons of an atom. There is a simple method for determining the number of valence electrons in the first 20 atoms of the periodic table. If you number the columns (or groups) above the first 20 elements from 1 to 8 this will indicate the number of valence electrons for every element in that column. For example, Be, Mg & Ca all have 2 valence electrons. The lone exception to this rule is helium (He) which has 2 valence electrons even though it is in the 8th column.

C. Drawing Lewis Structures
Lewis Structures are a method of representing atoms and molecules to reflect valence electrons and covalent bonds. In Lewis Structures, electrons are represented by dots and bonds are represented by lines.
D. Drawing Lewis Structures Step-By-Step: Single Atom Elements
The following instructions summarize the steps outlined in the video above
To draw the Lewis Structure for a single atom of an element, for example Fluorine (F) follow these steps:
- Determine the number of valence electrons.
- Fluorine is in the 7th column, therefore it has 7 valence electrons
- Write the symbol for the element
- Add electrons around the 4 sides of the symbol, one electron at a time, once all 4 sides have a single electron you begin pairing them. Stop adding electrons once you have added the total number of valence electrons.
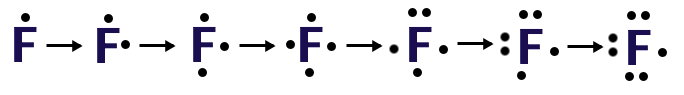
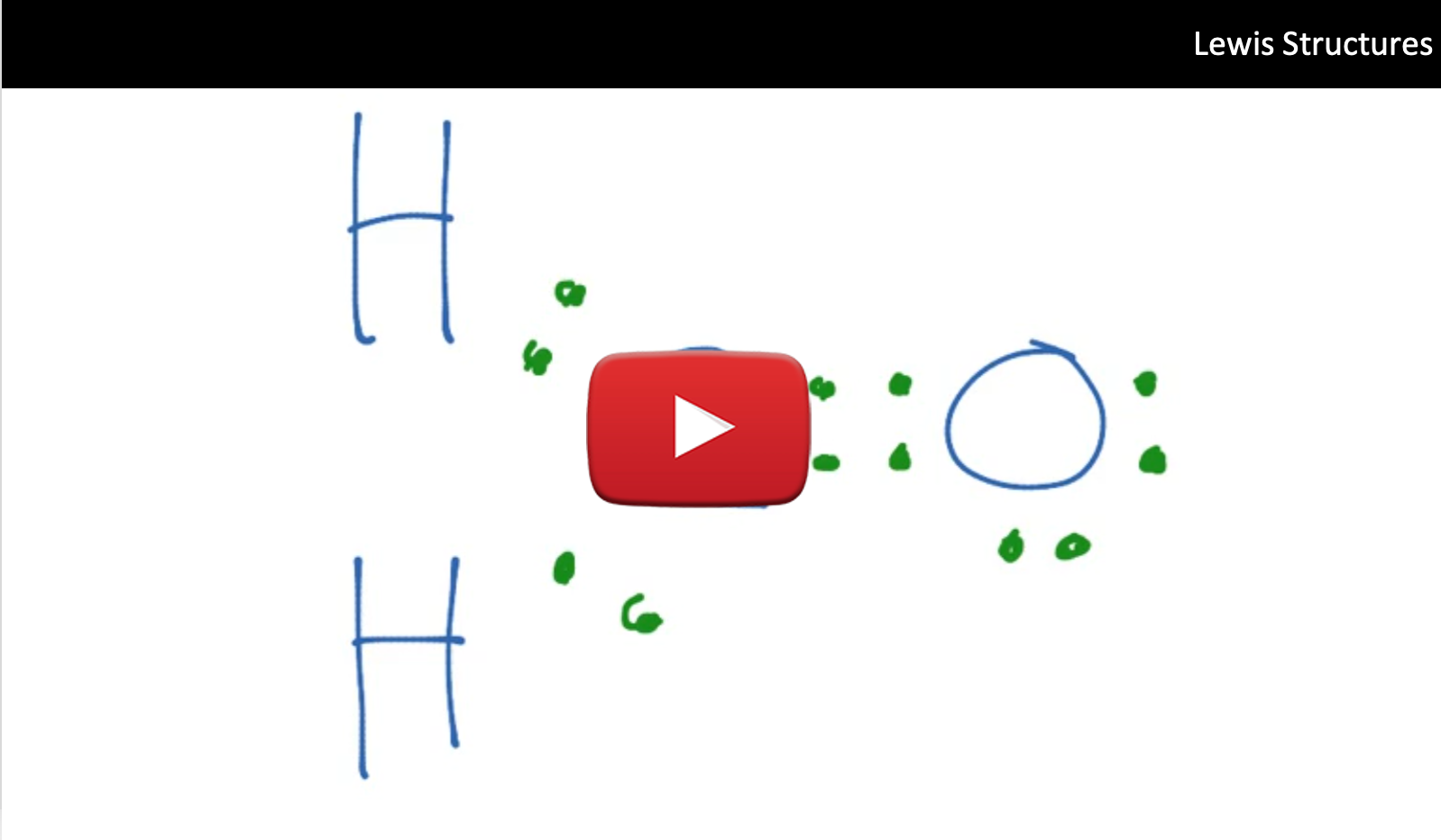
E. Drawing Lewis Structures Step-By-Step: Polyatomic Molecules
To draw the Lewis Structure for polyatomic molecules, for example (NO3)- follow these steps (*note the - (minus) sign indicating a net charge of -1):
-
- N = 5 valence electrons
- O = 6 valence electrons. Since there are 3 oxygen molecules, you multiply the valence number of oxygen by 3. O = 3*6 = 18 valence electrons total
- Total valence electrons = 5 + 18 = 23
- However this molecule has a charge of -1, which means it has an extra valence electron, so we have to add 1 more
- Total valence electrons = 5 + 18 + 1 = 24 total valence electrons
2. Identify the central atom and write its symbol, surrounded by the symbols of the other atoms in the molecule. The central atom will be the atom that all the other atoms bind to. The central atom is typically the least electronegative atom, you should recall from previous chemistry courses that electronegativity increases as you move left-to-right and bottom-to-top across the periodic table. In the case of (NO3)-, nitrogen is lower on the periodic table than oxygen, therefore nitrogen has a lower electronegativity value than oxygen and it will be our central atom in this molecule. Therefore the most electronegative element and In this case we will have a N atom bound to 3 O atoms.

3. Place 1 pair of electrons between each pair of atoms. These represent the shared, bonding electrons. At this point, we have placed 6 of the 24 valence electrons.
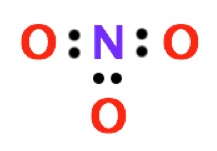
4. Place pairs of the remaining electrons on the surrounding atoms, following the octet rule (or the duet rule for hydrogen).
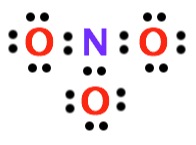
5. Determine how many valence electrons are still available to be placed. If there are valence electrons left over, add them to the central atom. In this case, we have placed 24 valence electrons, and we started with 24 so we are out of valence electrons to place.
6. Determine if the central atom has an octet. In this case, the central atom (N) only has 6 electrons surrounding it. This is not an octet which makes N very sad. If the central atom does not have an octet, move one of the lone pairs of electrons from a surrounding atom between that atom and the central atom. There will now be 4 electrons shared between these two atoms. These 4 shared electrons now represent a double bond.
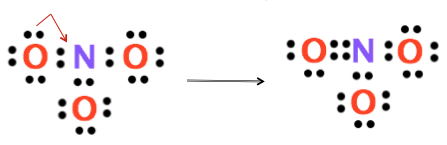
7. For polyatomic ions, once the Lewis structure is completed, square brackets are placed around the drawing with the charge written as a superscript outside the brackets.
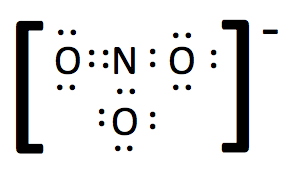
We now have a stable molecule as all of the constituent atoms have stable octets. The next step is to simplify the diagram into a structural formula by converting the shared electrons, which represent covalent bonds into lines and removing lone pair electrons from the outer atoms. If there are lone pair electrons on the central atom, do not remove them from the simplified drawing.
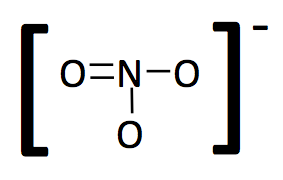
Test your understanding:
Question 1-4A-1: Draw the simplified Lewis Structures for the following molecules:
a) O2
b) CH4
c) NH3
Answer: 1-4A:1
F. Summary
- Electron configuration has an important relationship to the structure and properties of atoms and molecules
- Lewis Structures are diagrams that display atoms, electrons and chemical bonds
- Atoms share electrons between them to form covalent bonds
- The octet rule states that many atoms are most stable when they are surrounded by 8 electrons in their valence shell
- Lewis Structures can be used to show single atom elements or multi-atom molecules
G. Additional Resources