SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-4B:VSEPR Theory
Description
In this activity you will learn about how the repulsive effect of neighbouring electrons can help predict the shapes that poly-atomic molecules will form.
Page content
A. Introduction
In the last learning activity we reviewed Lewis Structures which helps us create 2-D image of atoms and molecules. However, as you may have guessed the real picture is a little more complex. Atoms and molecules exist in 3-D space, and knowing the 3-D shapes they take on helps us understand some of the properties that these molecules display. Producing accurate, real images of individual molecules is extremely difficult, but scientists have developed a model to help predict the shapes of molecules known as VSEPR (pronounces Ves-Per) theory. The VSEPR theory produces shapes that act to minimize the repulsive forces within molecules by placing electrons as far away as possible from each other. In fact, a Canadian scientist by the name of Ronald J. Gilespie was one of the key contributors to this theory. Click here to learn more about Ronald J. Gilespie's contributions to VSEPR theory.
B. VSEPR Theory
As mentioned above the VSEPR theory predicts molecular shapes by arranging the atoms around a central molecule in such a way as to reduce the repulsion between neighbouring electron groups. Electron groups consist of either bonding pairs of electrons or lone pairs of electrons (keep in mind that a double or triple bond only count as single electron group). A simple way of thinking of VSEPR theory is that electron pairs want to be as far away from each other as possible because they have the same charge and we can predict the shapes of molecules based on this repulsive effect. However, not all electron groups are created equally, it turns out that lone pair electrons have a stronger negative charge than bonded electron pairs, so these lone pairs actually have a stronger repulsive effect.
C. Predicting Shapes With VSEPR
There are a simple set of steps we can follow to predict shapes using VSEPR theory. For this example lets use CCl4 (carbon tetrachloride) and try to predict its shape.
1. Draw the simplified Lewis Structure of the molecule.
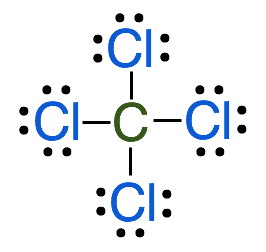
2. Count the electron pairs surrounding the central atom including both bonded and lone pair electrons.
-
- In this case, there are 4 bonding electron pairs around the central atom and zero lone pairs
3. Arrange the surrounding atoms and any lone electron pairs around the central atom to minimize the repulsion between them by placing them as far apart as possible in a 3-D space. To arrange the atoms as far apart as possible in 3-D space we need to consider the bond angle, which is the angle formed between 2 adjacent atoms with the central atom as the vertex.
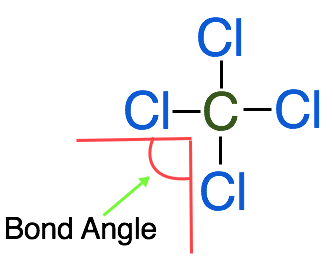
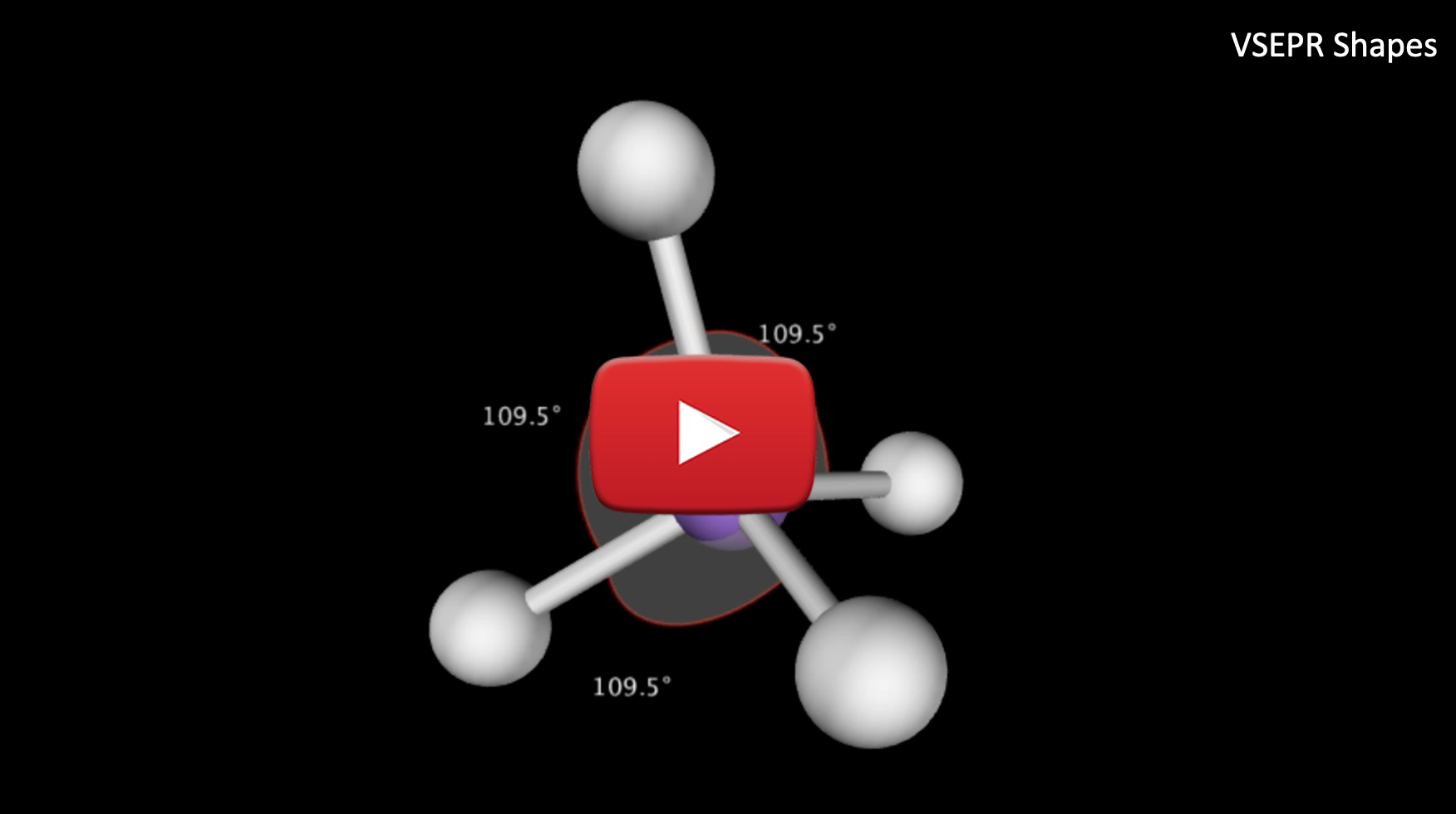
Looking at the Lewis Structure above the bond angles for each of the surrounding atoms is 90˚. To create the furthest distance between the surrounding molecules we want to maximize the bond angle. To do this we need to consider the molecule in 3-D space. VSEPR theory finds that for a central atom surrounded by 4 electron groups, the shape that minimizes repulsion is represented on a Lewis diagram as follows:
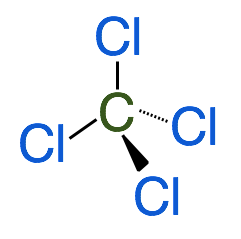
In the diagram above, the dashed line represents an atom that is extending towards the back of the page and the wedge represents an atom that is extending out, away from the page. In this conformation, the bond angles are 109.5˚ and the maximum separation between surrounding atoms is achieved. The 3-D model of the shape drawn above looks like this:
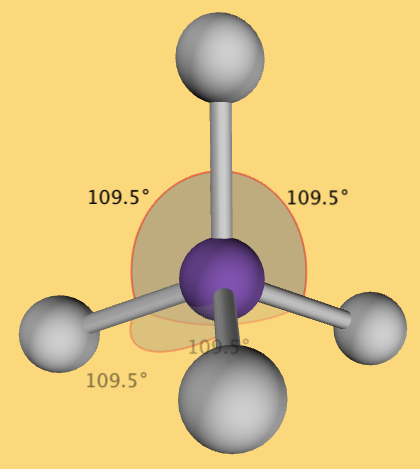
4. Determine the name of the structure and the bond angles.
-
- The name of the shape above is called a tetrahedral and it has bond angles of 109.5˚
Let's review what you've learned so far. Source: PBS Learning Media
We will investigate the shapes of simple covalent molecules. Specifically, we will be learning about something called VSEPR theory, and how it allows us to predict the shapes that covalently bonded molecules will have.
VSEPR stands for Valence Shell Electron Pair Repulsion. It tells us that areas of a molecule that have a higher concentration of electrons will repel each other and that these repulsions will affect the shape of the molecule.
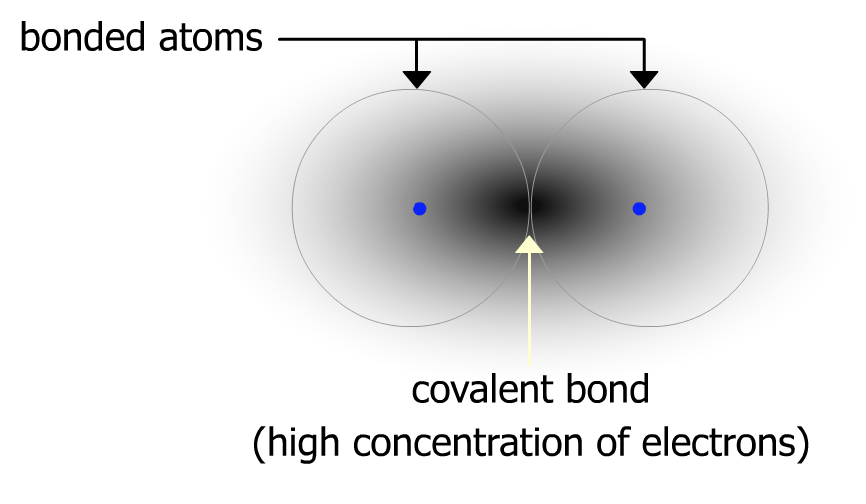
We have already learned that a covalent bond is actually electrons that are being shared by two atoms. These electrons are most likely to be found somewhere between the two bonded atoms as shown below.
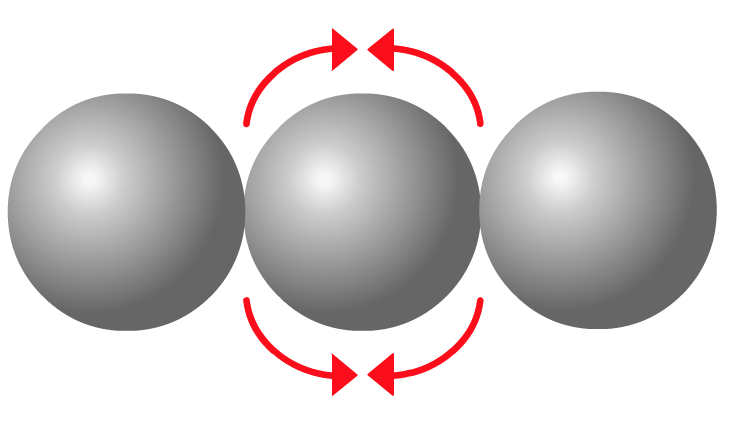
If three atoms are bonded together, the negatively charged electrons in each of the bonds will repel each other.
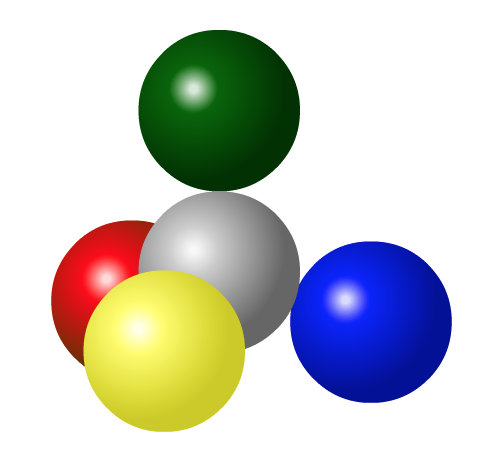
The image on the left has two bonds, when the third bond is added the same principle applies. The electrons in the bonds repel each other. In order to be as far apart as possible, the three outside atoms will take positions 120o apart. And when the fourth bond is added, they space themselves out three-dimensionally to be 109.5o apart.
Now, let's try to predict a few molecular structures based on their chemical formulas.
Let's start with CO2
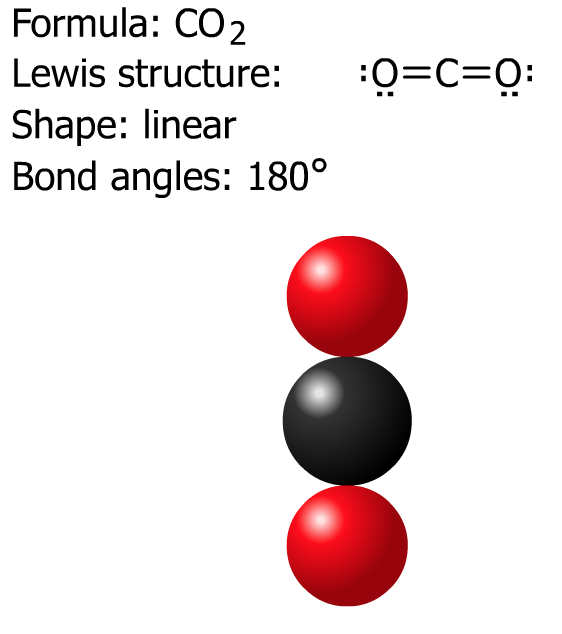
Next, try SO2 and NO3- then check the solutions below.
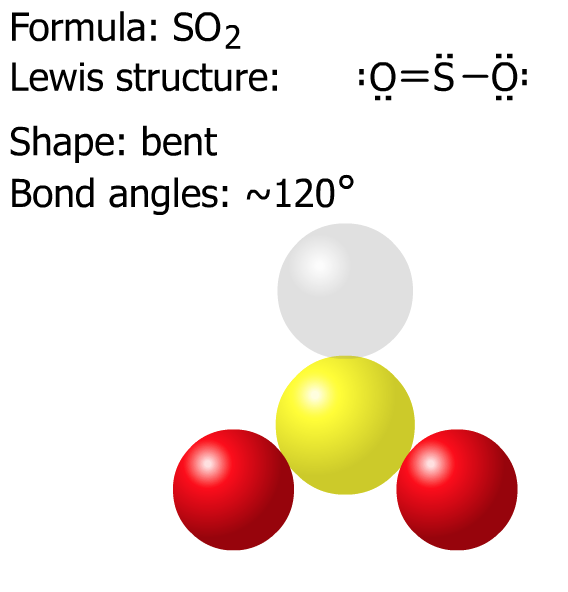
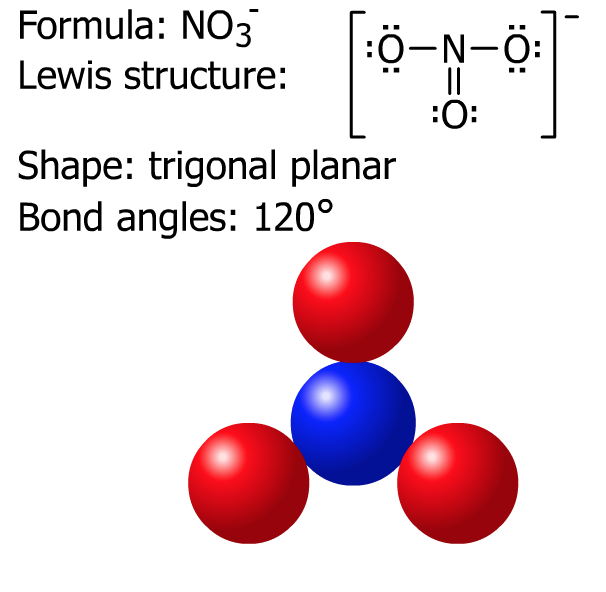
D. Summary
- VSEPR = Valence-Shell Electron-Pair Repulsion
- VSEPR theory is based on the fact that electron groups are negatively charged and repel each other, therefore they try to orient themselves as far away as possible from each other
- Molecular shapes predicted by VSEPR are based on the number and type of electron groups within a molecule