SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-4C: VSEPR Shapes
Description
In this activity you will learn about the different molecular shapes that can be predicted by applying VSEPR theory.
Page content
Common VSEPR Shapes
In the previous learning activity, you learned how to apply VSEPR theory by configuring Lewis Structures to minimize electron repulsion. This learning activity will introduce some common molecular shapes predicted by VSEPR theory. Remember, the goal of VSEPR is to reduce the repulsive effect of electron groups around the central atom.
Molecular Shape Simulator
Use this simulator as you move through the content below to help you to see how VSPEPR theory is applied and to test your knowledge:
Source: http://phet.colorado.edu/en/simulation/molecule-shapes
PhET Interactive Simulations
University of Colorado Boulder
http://phet.colorado.edu
A. Linear
Linear shapes are predicted when the central atom has only 2 electron groups. Bond angles in a linear molecule are 180˚. An example of a linear molecule is CO2.
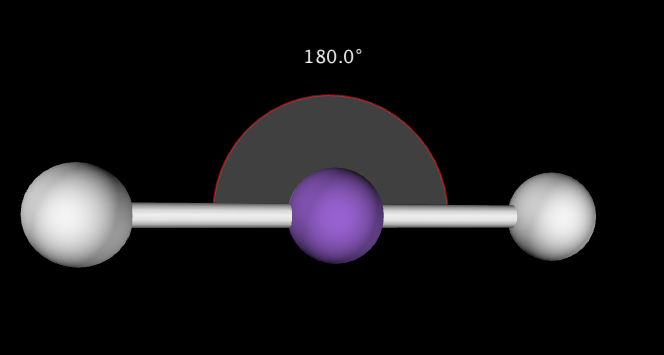
It should be noted that VSEPR can be used to predict the shapes of molecules without a central atom as well. For example, N2 has the following Lewis Structure, which also is linear with 180˚ bond angles

B. Bent
There are 2 scenarios that produce bent shapes:
1.Central atom has 3 total electron groups, 2 bonding pairs of electrons and 1 lone pair of electrons. This produces a molecule with 120˚ bond angles. Example: SO2
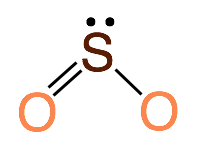
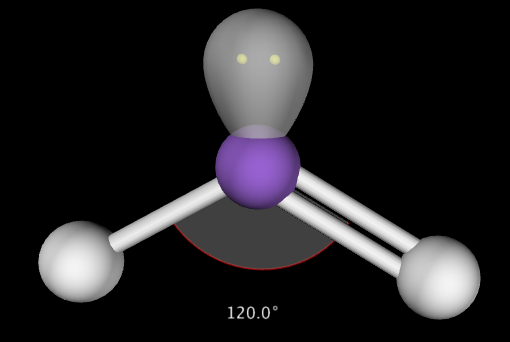
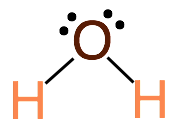
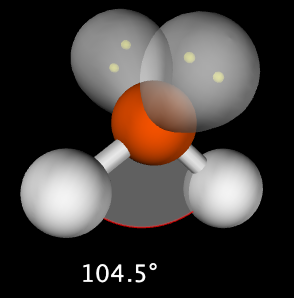
2. Central atom has 4 total electron groups, 2 bonding pairs of electrons and 2 lone pairs of electrons. This produces a molecule with 104.5˚ bond angles. Example: H2O
C. Trigonal Planar
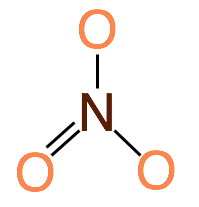
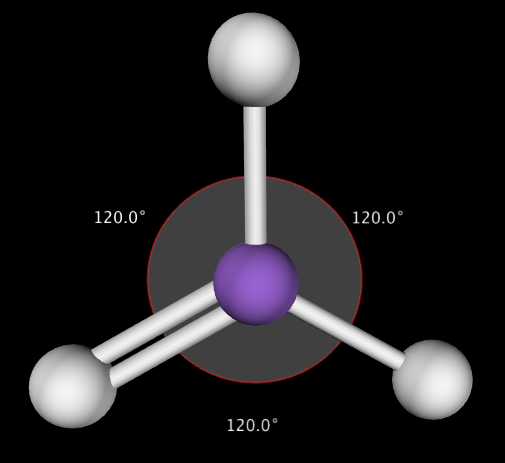
Trigonal Planar shapes are predicted when the central atom has 3 bonding electron pairs surrounding it. This produces a molecule with 120˚ bond angles. Example NO3-
D. Tetrahedral
Tetrahedral shapes are predicted by VSEPR when the central atom has 4 bonding electron groups. This produces a molecule with 109.5˚ bond angles. Example CCl4
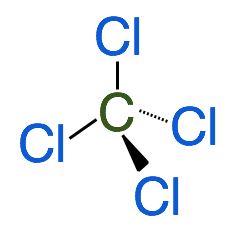
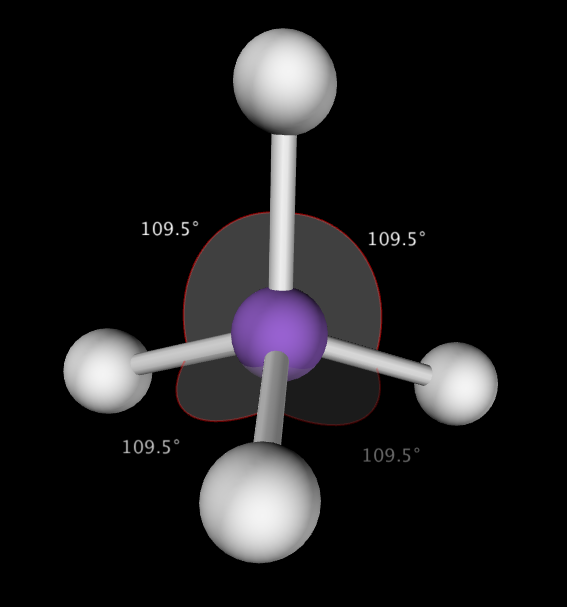
E. Trigonal Pyramidal
Trigonal Pyramidal shapes are predicted by VSEPR when the central atom has 3 bonding electron groups and one lone pair of electrons. This produces a molecule with 109.5-107.8˚ bond angles. Example: NH3
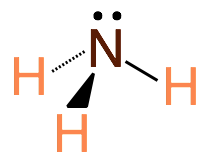
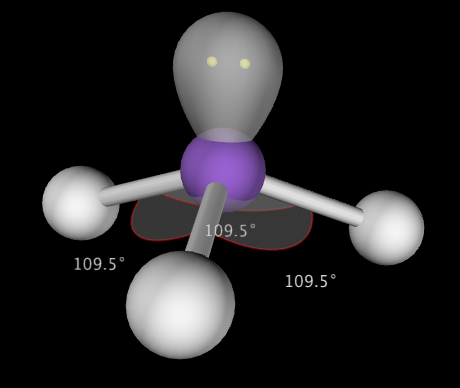
Note: There are several other molecular shapes predicted by VSEPR theory, but they are beyond the scope of this course and you are not expected to know them.
Test your understanding
1-4C-1 Use your knowledge of VSEPR to predict the 3-D shapes of the following molecules:
a) H2
b) PH3
c) BCl3
Answer: 1-4C:1
For a review of the shapes predicted by VSEPR watch this brief video:
F. Summary
- Shapes predicted by VSEPR depend on the number and type of electron groups surrounding the central atom
- Basic shapes predicted by VSEPR include
- Linear: Central atom has 2 electron groups or a molecule made up of only 2 atoms (Bond angle: 180˚)
- Bent: Central atom has 2 bonding electron pairs and 2 lone electron pair (bond angle: 104.5˚)
- Trigonal Planar: Central atom has 3 bonding electron pairs (bond angle: 120˚)
- Tetrahedral: Central atom has 4 bonding electron pairs (bond angle: 109.5˚)
- Trigonal Pyramidal: central atom has 3 bonding electron pairs and 1 lone electron pair (bond angle: 107.8˚)