SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-4D: Lewis Dot Review
This page is part of the content downloaded from 1-4D: Lewis Dot Review on Monday, 3 March 2025, 9:20 AM. Note that some content and any files larger than 50 MB are not downloaded.
Description
The following resource will review the protocol for drawing Lewis structures and offer some practice questions to test your understanding.
Page content
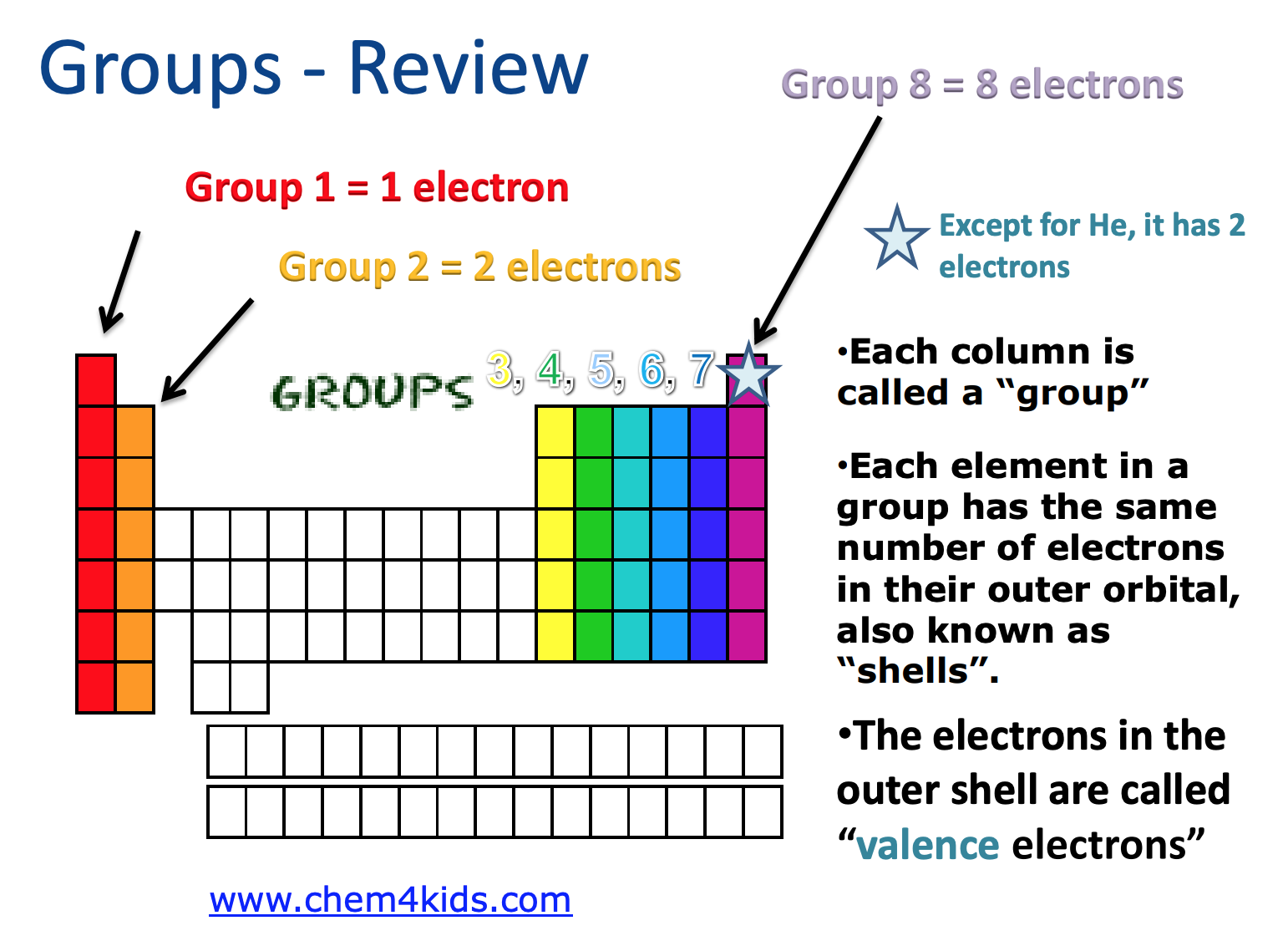
Electron Dot Diagrams
1) Find your element on the periodic table.
2) Determine the number of valence electrons.
- Valence electrons are the number of electrons in the atom’s outer most shell.
3) This is how many electrons you will draw.
Electron Dot Diagram
1) Write the element symbol.
2) Carbon is in the 4th group, so it has 4 valence electrons.
3) Starting at the right,
draw 4 electrons, or
dots, counterclockwise around the
element symbol.

Try these elements on your own:
a) H
b) P
c) Ca
d) Ar
e) Cl
f) Al
Answers:






You should now be able to
draw an electron dot
diagram and a Bohr model
for the first 20 elements in
the periodic table.