SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-6A: Intermolecular Forces
Description
In this learning activity you will discover about the different types and relative strengths of the forces that hold molecules and atoms together.
Page content
A. Introduction
Intermolecular forces are the forces that occur between molecules that cause them to interact with each other, whereas intramolecular forces are the forces within a molecule that hold it together as we've seen already with covalent bonds. Intermolecular forces are responsible for many of the properties of matter including boiling points, melting points, and surface tension. To visualize the effects of intermolecular forces imagine an ice cube melting into a puddle of liquid water and that same puddle of water being heated up until it boils and evaporates. The water has changed states from solid to liquid to gas, but the molecular structure of the water never changed, it was always H20, yet its physical appearance and characteristics changed throughout. These changes are due to changes in intermolecular forces which we will examine in this lesson. The three forces which we will learn about in this activity are dipole-dipole forces, hydrogen bonding and London dispersion forces, combined these 3 make up a class of forces known as Van Der Waals forces.
Watch this brief video for an introduction to the concepts we will explore in this lesson:
Source: youtube
We learned in the previous lesson that some molecules can have a polar quality depending on the shape of the molecule and the electronegativity of the atoms within it. If you have a substance that is made up of polar molecules, the positive and negative poles of the molecules will be attracted to each other and will arrange themselves so that the positive and negative ends are close to each other.
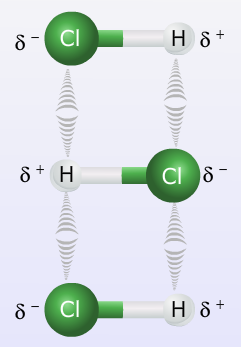
Dipole-dipole forces are very weak compared to a covalent bond, in fact they are about 1% as strong and can be greatly influenced by temperature and pressure. The strength of the force weakens considerably as the distance between molecules increases. For example if a polar substance is in its gaseous form at low pressure, the molecules of the substance will be spread out and the dipole-dipole forces will be much weaker than if the pressure was increased forcing the molecules closer to each other.
C. Hydrogen Bonding
Hydrogen bonding is a special type of intermolecular force that is stronger than a typical dipole-dipole force. Hydrogen bonding occurs in molecules where a hydrogen atom is covalently bonded to an atom that has a very high electronegativity value and is small enough to get close to the positive dipole of an adjacent molecule. Hydrogen bonding only occurs when hydrogen is bound to oxygen, nitrogen or fluorine.
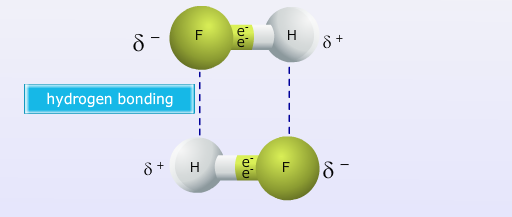
This type of force tends to be stronger than typical dipole-dipole forces because of the large difference in electronegativity values and the ability for opposite poles to align very close to each other. Hydrogen bonding plays a very important role in giving water its unique qualities as well as influencing many other substances' physical properties. The image below shows how hydrogen bonding occurs between water molecules, note the dashed lines are used to represent a hydrogen bond.
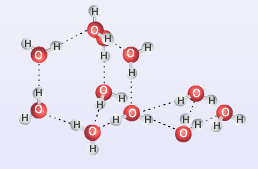
Due to the strength of the hydrogen bond, substances with hydrogen bonding between molecules have higher boiling points than expected based on their molecular mass. This is because it takes more energy to break the hydrogen bonds and force the molecules far enough apart from each other to go from liquid to gas than a substance that only has dipole-dipole forces holding it together. It is still important to note though that as strong as hydrogen bonds are, they are nowhere near as strong as the intramolecular forces that hold atoms within a molecule together. In fact they are 10 to 20 times weaker than most covalent bonds. Hydrogen bonds are crucial for many important biological molecules and processes including creating the double-helix structure of DNA and the structure and function of proteins and enzymes.
D. London Dispersion Forces
So far we've looked at 2 types of intermolecular forces that exist due to the polar molecules; however, intermolecular forces also exist between non-polar molecules, and these are called London Dispersion Forces, named after the man who discovered them, Fritz London. London dispersion forces explain how all substances can exist as liquid and solid under the right circumstances. The basic explanation behind London dispersion forces is that even in non-polar molecules, the constant movement of electrons can create temporary dipole-like arrangements of charge. The electrons of an atom are not evenly distributed around the nucleus of an atom at all times. As the electrons move around the nucleus it is possible for the distribution of them to create what is known as a temporary dipole. This temporary dipole can then influence the electrons of neighbouring molecules and induce a dipole in them as well. These dipoles are very weak and don't typically last very long, but the effect can be significant for large atoms or at very low temperatures. The measure of how easy it is to induce a dipole in a molecule is called its polarizability. Large atoms with many electrons have a higher polarizability than smaller atoms.
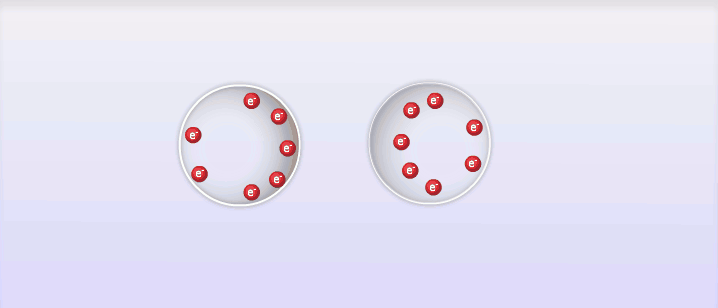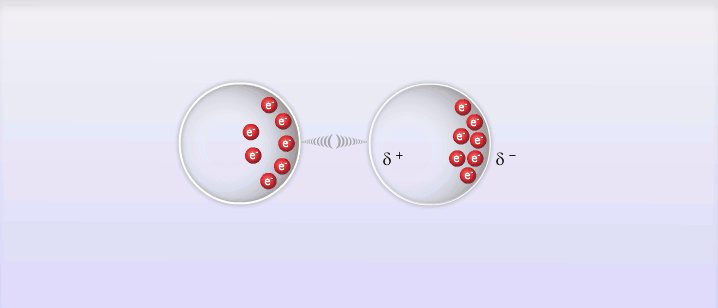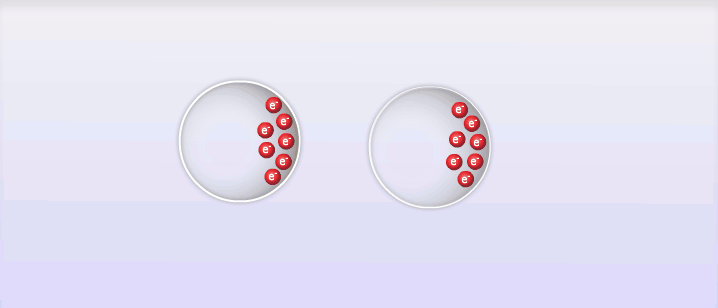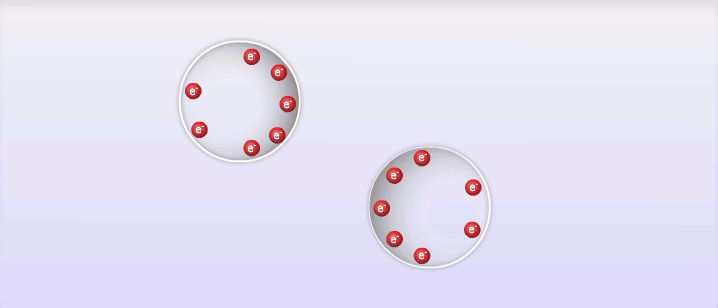
E. Intermolecular Forces and Physical Properties:
The strength of the intermolecular forces within a substance impacts many of its physical and chemical properties. Substances with stronger attractive forces between molecules will have higher boiling points and melting points. As we have seen above, hydrogen bonds are stronger than dipole-dipole forces which are stronger than London dispersion forces. What this means is that molecules with hydrogen bonds tend to stick together more than those with dipole-dipole forces and London dispersion forces. The strength of the forces that keep molecules together within a substance affects many physical properties. For example, recall that for matter to transition from solid to liquid or liquid to gas requires the molecules within to move farther and farther apart. Therefore, in substances with hydrogen bonds, the melting and boilind points will be higher than those with dipole-dipole or London dispersion forces because it takes more energy to separate the molecules from each other. Other properties that are affected by intermolecular forces include: viscosity, surface tension and solubility. We can use the information we know about these types of properties to predict the type of intermolecular forces present within a molecule.
F. Summary
- Intermolecular forces are the attractive forces between molecules that cause them to interact
- The intermolecular forces known as Van Der Waals forces are: dipole-dipole forces, hydrogen bonding and London dispersion forces
- Dipole-dipole forces occur between polar molecules where opposite poles are attracted to each other and molecules arrange themselves in such a way as to maximize this attraction
- Hydrogen bonding is a special type of intermolecular force that occurs in molecules that have a hydrogen atom bound to an oxygen, nitrogen or fluorine atom
- Hydrogen bonding is similar to dipole-dipole forces but the bond is stronger
- Hydrogen bonding gives water many of its unique qualities
- London dispersion forces occur when temporary dipoles form in non-polar molecules
- Temporary dipoles are caused by the random distribution of electrons around the nucleus, when electrons become disproportionately distributed, temporary +ve and -ve poles form which can then induce temporary poles in neighbouring molecules
- Intermolecular forces impact many physical characteristics such as boiling point and melting point. For example, stronger intermolecular forces cause higher melting and boiling points
Image Source:http://www.educationscotland.gov.uk/highersciences/chemistry/animations/intermolecularforces.asp
