SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-6D: Intermolecular Forces Investigation
Description
In this investigation, you will explore how intermolecular forces impact the properties of matter.
Page content
Intermolecular Forces Lab Investigation
For this investigation, your role is to use your knowledge of intermolecular forces and the properties of matter to predict how boiling points will vary based on their chemical and structural makeup. You will also use available data to test your predictions and analyze the trend in boiling points found in these different compounds. Record all of your data, graphs and answers to analysis questions for future reference.
Introduction: In all liquids, intermolecular forces are important, but these forces become negligible in the gas state for the conditions at which liquids boil. Therefore in this case we are looking at a situation where intermolecular forces must be overcome by adding energy, but no new bonds are formed. The temperature at which a liquid boils reflects the strength of the intermolecular forces present among the molecules. Higher temperatures mean more energy has been added and the intermolecular forces must have been stronger.
Purpose: The purpose of this lab investigation is to test the theory and rules for London dispersion and dipole-dipole forces. The question this lab seeks to answer is: What is the trend in boiling points of the hydrogen compounds of elements in group 14-17 of the periodic table?
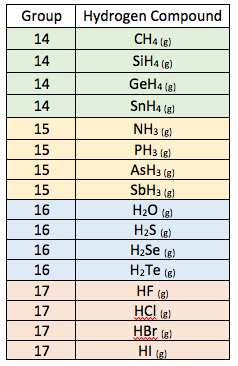
Hypothesis: Based on London dispersion and dipole-dipole forces write a prediction for the trend in boiling points within and between groups. For example, your prediction could include a general sketch of a graph of boiling point vs. number of electron per molecule. Explain the reasoning behind your prediction.
Data: Click here to open the data table containing the actual boiling points of hydrogen compounds of elements in groups 14-17
Analysis Questions:
- Using the boiling point data above, complete a graph of the results by plotting boiling point versus the number of electrons per molecule.
- Based upon your graph, what is the trend in boiling points of the hydrogen compounds of elements in group 14-17 of the periodic table?
- Assess the validity of your prediction that you made above, and the reasoning you used relating to intermolecular forces to make the prediction.
- Are there any unexpected data points in the results? If so, suggest a possible explanation for these.