SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-1A: Introduction to Organic Chemistry
Description
In this activity you will be introduced to the topic of organic chemistry and will learn about some of the different ways that organic compounds can be represented using formulas and drawings.
Page content
A. Introduction
Organic chemistry is the study of the properties and reactions of organic compounds. An organic compound is any compound that contains carbon atoms (excluding CO, CO2 and HCN). Organic compounds are everywhere around us and form the basis of life as we know it. There are over 1 million organic compounds and they are found in everything from our DNA to the fuel that powers our cars. We will begin this unit by looking at the different ways that chemists represent organic compounds. Hydrocarbons are compounds that only contain hydrogen and carbon atoms. Hydrocarbons are extremely prevalent in our world, for example, most fuels are made up of hydrocarbons.
B. Representing Organic Compounds
Chemists have developed several different methods for representing organic compounds in an attempt to simplify what can sometimes be very complicated structures. For example, a hydrocarbon known as pentane can be represented by a:
- molecular formula: C5H12
- structural formula:
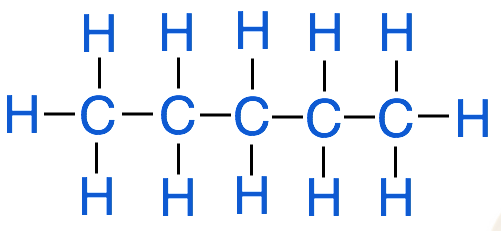
- condensed structural formula:
![]() or
or ![]()
- Line angle formula: there is a carbon represented at the end of each line and at the intersection of every line. Each carbon atom is surrounded by the maximum number of hydrogen atoms, so the carbons at the end have 3 hydrogens and the carbons in the middle have 2 hydrogens, unless other wise indicated
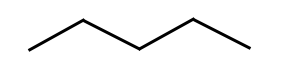
Remember, all of the diagrams and formula above represent the same pentane molecule.
C. Cyclic Organic Compounds
Organic compounds can exist as lines, branched lines as well as in rings. As above there are several ways to represent a hydrocarbon in a ring structure. In this example we will consider cyclopentane, which is a hydrocarbon ring with 5 carbons. Cyclopentane can be represented by a:
- molecular formula: C5H10
- structural formula:
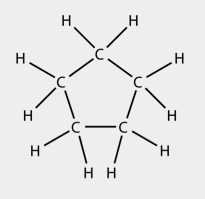
- condensed structural formula
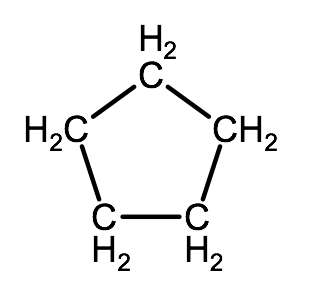
- Line-angle formula: There is a carbon represented at every line intersection, every carbon is surrounded by the maximum number of hydrogen atoms
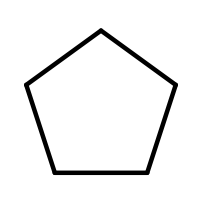
Aromatic Hydrocarbons
There is a special type of cyclic hydrocarbon known as an aromatic hydrocarbon. This is a class of compounds with similar structures, many of which are associated with the strong smells produced by spices such as vanilla, cinnamon and ginger. The simplest aromatic hydrocarbons are all structurally based on a compound with a unique structure known as a benzene ring. Benzene has a molecular formula of C6H6 and can be drawn with 2 different structural formulas (notice the difference in the placement of the double bonds):
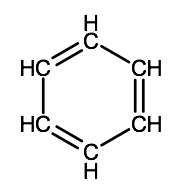
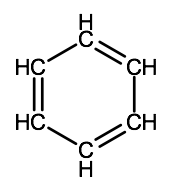
It turns out though that the electrons involved in the double bonds of benzene rings are actually shared equally by all of the carbon atoms that form the ring of a benzene molecule. Therefore, benzene molecules are often represented showing the double bonds shared within the ring.
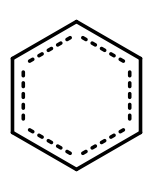
F. Summary
- Organic chemistry is the study of carbon-containing compounds
- Hydrocarbons are compounds made up of only hydrogen and carbon
- Organic compounds can be represented by molecular formulas, structural formulas, condensed structural formulas and line-angle formulas
- In line-angle formulas, there is a carbon represented at the end of every line and at the intersection of every line, every carbon is surrounded by the maximum number of hydrogen atoms
- Aromatic hydrocarbons are a special type of cyclic hydrocarbon in which the electrons involved in the double bonds are shared equally among the carbon atoms that make up the ring