SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-1B: Classes of Organic Compounds
Description
In this activity you will explore some of the different classes of organic compound as distinguished by the nature of the bonds within them and the functional groups attached to them.
Page content
A. Introduction
Organic chemistry is the study of the properties and reactions of organic compounds. An organic compound is any compound that contains carbon atoms (excluding CO, CO2 and HCN), these compounds are everywhere around us and form the basis of life as we know it. There are over 1 million organic compounds and they are found in everything from our DNA to the fuel that powers our cars. We will begin this unit by looking at the various classes of organic compounds and how they are different from each other. These classes of organic compounds have properties that are unique to each class and as we've seen so far throughout this course, the differences in properties between these classes is directly related to their diverse compositions and structures. So, lets begin by introducing the classes of organic compounds.
B. Hydrocarbons
Alkanes
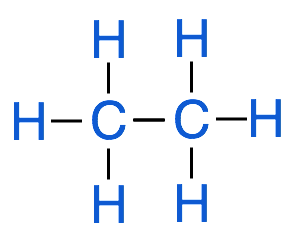
A hydrocarbon is an organic molecule that contains only hydrogen and carbon atoms. Even though hydrocarbons only contain 2 types of elements there are thousands and thousands of different types of hydrocarbons in existence. There are 3 basic types of hydrocarbons, alkanes, alkynes and alkenes. An alkane is what is known as a saturated hydrocarbon. In this case saturated means that all of the bonds between carbon atoms are single bonds. As you should recall from previous chemistry courses, carbon atoms have 4 valence electrons, therefore a carbon atom can be covalently bound to a maximum of 4 other atoms. We use the term saturated because all of the carbon atoms in an alkane are bound to the maximum number of atoms possible. Below is a diagram of a simple alkane, known as ethane.
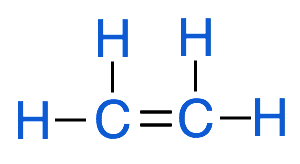
Alkenes
An unsaturated hydrocarbon is any compound made up of only hydrogen and carbons that has at least one double or triple bond in the structure, for example, a hydrocarbon that has double bonds is called an alkene, the image below represents an alkene with 2 carbons, which is known as ethene.
Alkynes
A hydrocarbon with at least one triple bond is known as an alkyne. These are relatively rare in nature. Below is an image of a 2 carbon alkyne, known as ethyne. Note the difference in naming these three types of hydrocarbon, the first part of the name (or prefix) indicates the number of carbons, while the last part of the name (the suffix) represents the types of bonds present.

Alkyl Groups
Not all organic compounds are straight chains as we've seen above, some organic compounds have groups that branch off of them, known as functional groups. If the branched group is made up of a hydrocarbon, it is known as an alkyl group (e.g. -CH3)
C. Alcohols
Alcohols are a common class of organic compounds that can be found in nature as well as manufactured synthetically. The defining characteristic of alcohols are that they are organic compounds that contain a hydroxyl group (-OH). The properties of alcohols are determined by how the carbon atoms are arranged within the molecule and we will learn more about these in later lessons. Below is a diagram of the alcohol known as ethanol, which is what is the organic compound commonly found in alcoholic drinks.
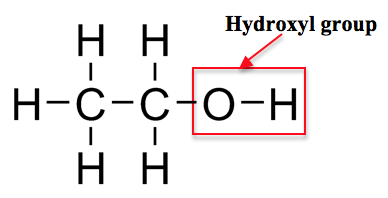
D. Ethers
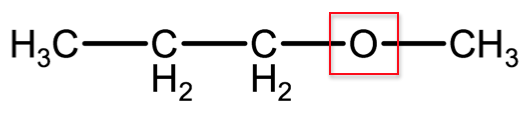
Ethers are a class of organic compounds that are formed by the condensation reaction of alcohols. The distinguishing characteristic of ethers is an organic compound with a functional group that contains an oxygen atom bound between 2 carbon atoms. Below is the structural formula for a common ether known as methoxypropane.
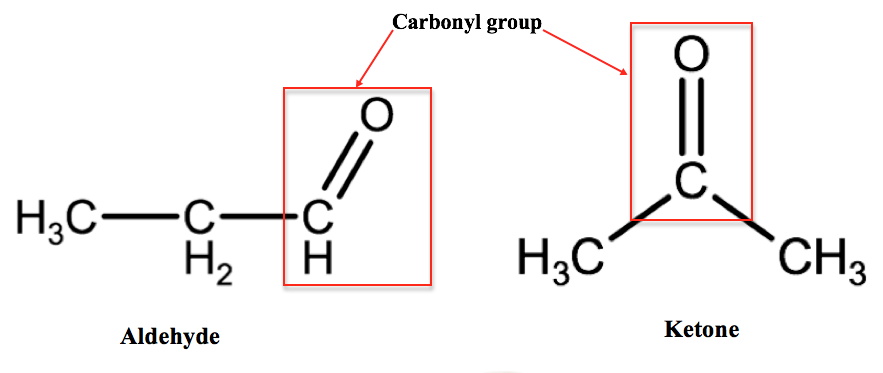
E. Aldehydes & Ketones
Aldehydes and ketones are classes of organic compounds that are defined by the presence of a carbonyl group they differ from each other based on what other atoms the carbonyl group is bound to. A carbonyl group is a carbon atom that is double bonded to an oxygen atom (-C=O) and in an aldehyde, the carbonyl group is bound to at least one hydrogen atom, in aldehydes, the carbonyl group is found at the end of a carbon chain. In a ketone, the carbonyl group is bound to 2 carbon atoms and is located somewhere within the middle of a carbon chain.
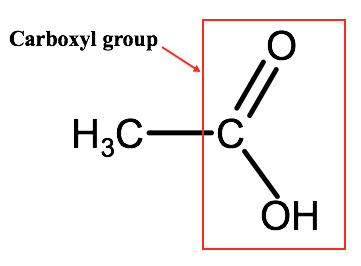
F. Carboxylic Acids
Carboxylic acids are organic compounds that are defined by the presence of a carboxyl group (-COOH). A carboxyl group contains a carbon atom double bonded to one oxygen atom and single bonded to a hydroxyl group. Below is an image of a simple 3 carbon carboxylic acid known as ethanoic acid.
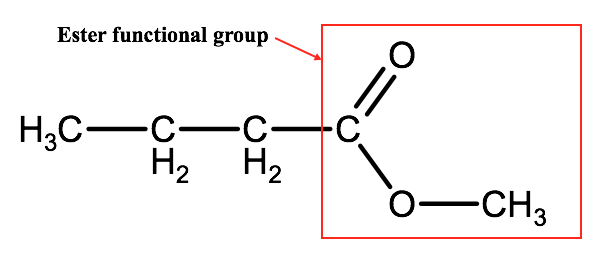
G. Esters
Esters are a class of organic compound very similar to carboxylic acids. Esters are defined by the presence of a functional group that is similar to a carboxyl group, but with the hydrogen atom of the carboxyl group replaced by an alkyl group. Functional groups can be represented by their full structural formulas or simply by the letter R. The image below represents the ester methyl butanoate.
H. Amines & Amides
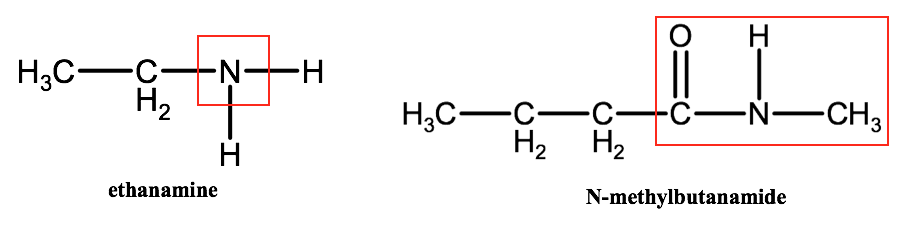
Amines and amides are organic compounds that are characterized by the presence of nitrogen atoms within them. Amines are derivatives of ammonia that contain one nitrogen atom bound to one or more alkyl groups. Amides are somewhat similar to esters in that they contain a carbonyl group bound to a nitrogen atom. Below are diagrams of the simple amine: ethanamine and the simple amide: N-methylbutanamide.
I. Summary