SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-1C: Isomers
Description
In this activity you will learn how compounds with the same formulas can have very different structural and physical properties depending on the arrangement of the atoms within them.
Page content
A. Isomers
So far we have been introduced to the basics of organic chemistry, including how to represent organic compounds in various different formats as well as describing the various classes of organic compounds. This next acitivity introduces the concept of isomerism. Isomerism is a great example of how the properties of a compound are dependent on its structure and composition and these properties can vary widely based on tiny structural differences, which is a theme that should be very familiar to you at this point in the course. Isomers are 2 or more compounds with the same molecular formulas that differ in the arrangement of their atoms within the molecule. A good way to think of isomer is to consider a Lego set with 5 different pieces, you could theoretically arrange those 5 pieces into many different shapes, but all of those different shapes would still be made up of the same 5 pieces. Each of your different shapes in this scenario would be considered isomers of each other. There are 2 different types of isomers that are relevant to this course, we are going to introduce the concepts in this learning activity and we will learn more about them in future lessons.
B. Structural Isomers
Structural isomerism occurs when 2 different molecules have the same molecular formula (ie they are made up of the same type and number of atoms) but the atoms are joined together in different ways. Take for example an organic compound with the molecular formula C4H10, this compound can be organized in the following ways:
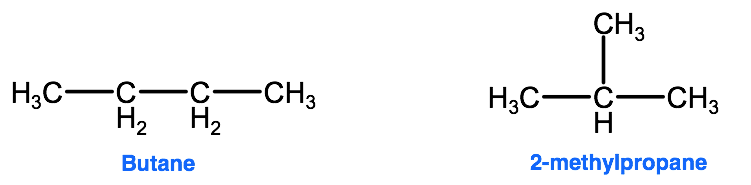
When organized as a straight chain, the compound is known as butane, which has a boiling point of -1°C; however when organized with a branched structure, the compound is known as 2-methylpropane which has a boiling point of -11.7°C. These molecules both have the exact same composition, yet the subtle difference in how the atoms are organized gives them very different properties. In other words, the properties of these 2 molecules is directly related to its structural organization.
C. Stereoisomers
A second type of isomerism exists known as stereoisomerism that is typically found in alkenes, which as we learned are hydrocarbons containing at least one double bond. Structural isomers are molecules that have the same molecular formula, and the atoms bonded in the same order, however the arrangement of the atoms in 3-D space are different. For example, the two molecules below both have the chemical formula C4H8 and may appear to be identical but if you look closely you will notice that the molecule on the left, named cis-2-butene, has two alkyl (-CH3) groups on top, whereas the molecule on the right, named trans-2-butene, has one alkyl group on the top and one on the bottom. This subtle difference in structure is enough to cause a difference in the boiling points in the 2 molecules, with cis-2-butene boiling at 3.7°C and trans-2-butene boiling at 0.9°C.
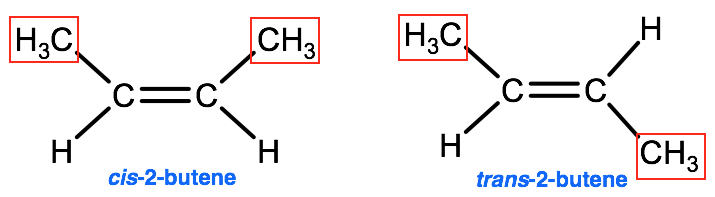
For these types of stereoisomers, when both alkyl groups are on the same side of the double bond, they are given the prefix cis-, whereas when the alkyl groups are on either side of the double bond they are given the prefix trans-.
D. Summary
- Isomers are molecules that have the same molecular formula but different arrangement of atoms
- Structural isomers have the same formula but the atoms or groups of atoms are bonded in different orders
- Stereoisomers have the same molecular formula but the arrangement of the atoms in 3-D space differ
- The structural differences between two isomers causes noticeable and quantifiable changes in the properties of each isomer