SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-2A: Alkanes
Description
In this activity you will learn the correct procedures for how to name and draw simple hydrocarbons known as alkanes.
Page content
A. Naming Simple Alkanes
Click here to access a worksheet and Powerpoint presentation that will help organize your thoughts and summarize this activity.
There are over 1 million different organic compounds that we know of. Can you imagine trying to come up with names for every single one? Chemists have developed a systematic method to name organic compounds that can be translated into molecular formula as well as structural formula.
For simple alkanes there is a naming system that you can think of as them having a first (prefix) and last (suffix) name, merged into one. The prefix of a simple alkane tells us simply how many carbons are in that alkane, while the suffix tells us what type of hydrocarbon it is. For example, the simples alkane is methane. The prefix meth- tells us there is 1 carbon atom, while the suffix -ane tells us this is an alkane. You are expected to know the names of the first 10 simple alkanes which are summarized in the table below:
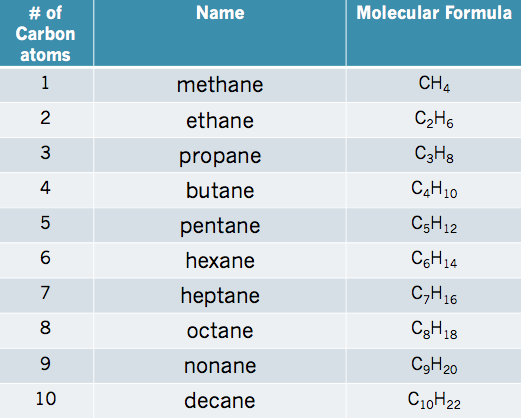
To remember the names of the first 10 alkanes it is useful to use a memory tool, for example in the following strange sentence:
Monkeys Eat Peeled Bananas, Pandas Hear Happy Otters Napping Daily
the first letter of every word corresponds to the order of the first letter of the alkane name as you move down the chart (Methane, Ethane, Propane, Butane, Pentane, Hexane, Heptane, Octane, Nonane, Decane). Try to come up with your own silly sentence to help you remember these names, you can post your own mnemonic in a forum, by clicking here. Remember every time you move down a column of the chart you are adding 1 carbon atom.
Check your understanding
Question 2-1B-1 Name the following molecules
a) C5H12
b)
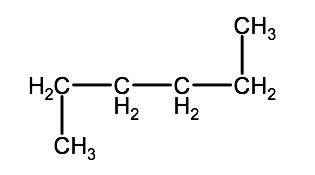
Answer 2-1B:1
B. Branched Alkanes
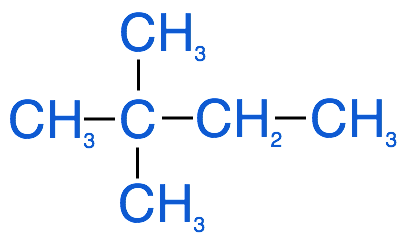
Alkanes can exist in different conformations, lines (e.g. propane), rings (e.g. cyclopropane) and branches (e.g. 2,2-dimethylbutane):
Alkyl Groups
As you can see in the image above of 2,2-dimethylbutane, alkanes can have groups that branch off from the main structure. If these groups are also hydrocarbons, they are called alkyl groups. Naming alkyl groups follows the same convention as naming simple alkanes, the prefixes are all the same, but the suffix for these branched alkyl groups is simply -yl. For example if the branched alkyl group has 2 carbons it is called an ethyl group, if it has 10 carbons it is called a decyl group.
C. Naming Complex Alkanes
To deal with the more complex compounds chemists have devised a structured naming system that was developed by the International Union of Pure and Applied Chemistry (IUPAC). This system is devised in such a way that you can take the name of a complex organic compound and be able to translate that into its structure. The steps you will now learn may seem complicated but stick with it, once you have practiced a few times you will understand the patterns and methods better. In fact by the end of this unit, you will be able to look at a complex name such as 2-ethylbutyl 3-methylpentanoate and be able to easily convert that into a structural formula.
Watch this video for a tutorial on how to properly name alkanes using IUPAC conventions. The step-by-step instructions follow below.
Note: when naming organic compounds commas are used to separate numbers and dashes (-) are used to separate numbers and words
When naming complex alkanes the important information we need to identify is:
- The longest continuous carbon chain. This is known as the parent chain and will form the root name of the molecule
- The number of carbons in each branched alkyl group
- The carbon in the parent chain that a branched alkyl group branches from
From this information we will be able to derive the name of any complex alkane.
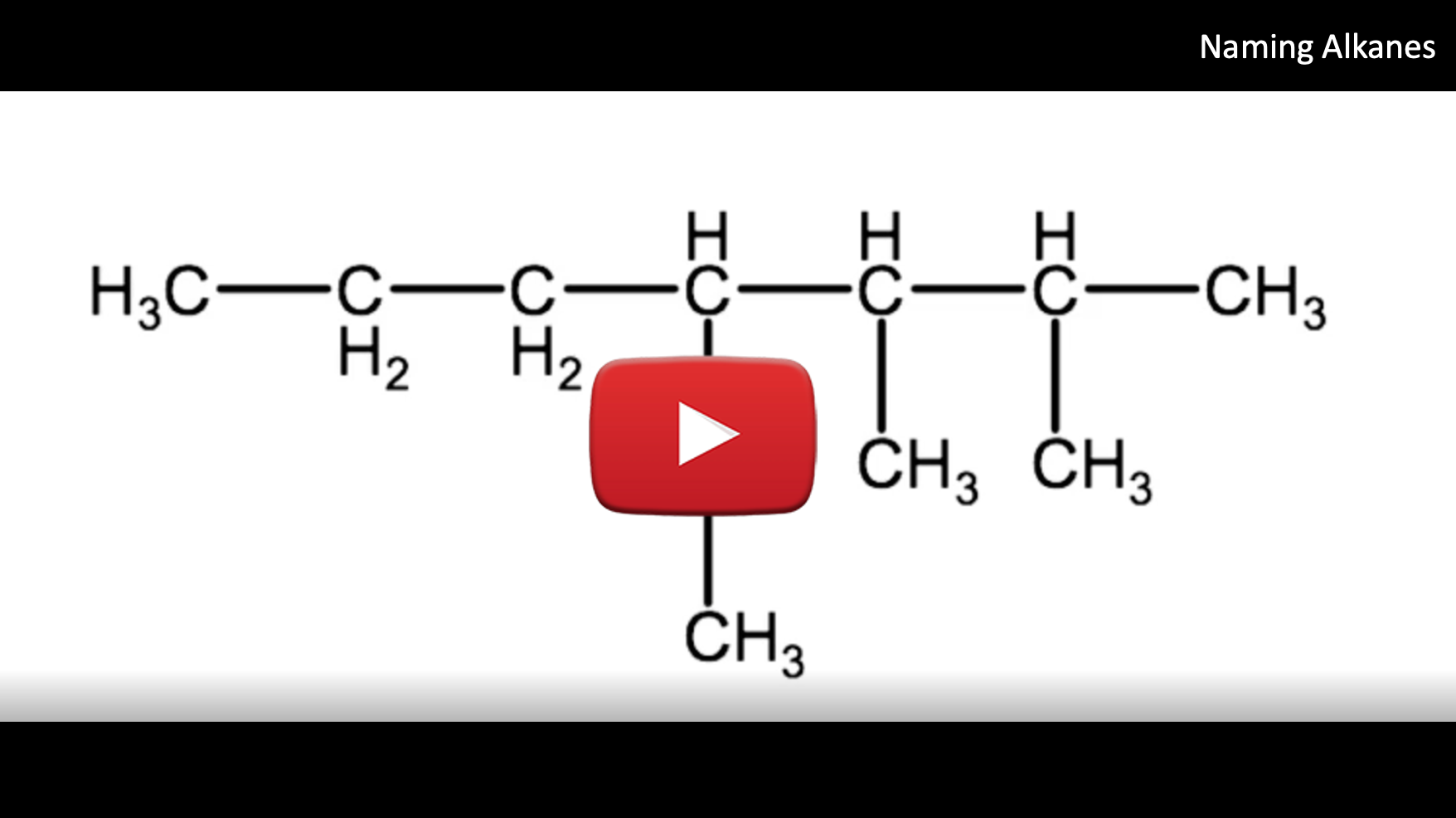
The image below represents the branched alkane that we want to name follow the steps below to derive the correct IUPAC name: 4-ethyl-2,3-dimethylheptane
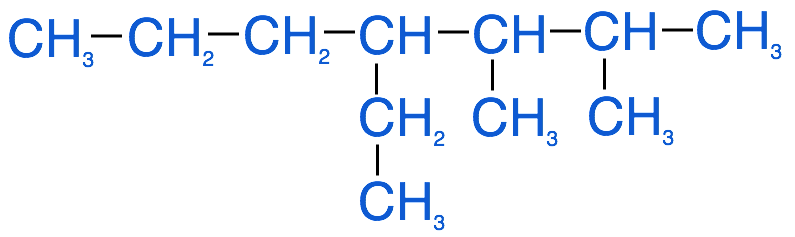
1. Identify the parent chain. This is the longest continuous carbon chain within the molecule. This is very important. Continuous implies carbons in a row, but not necessarily in a straight line. You will need to examine the molecules carefully to determine the longest chain. For example in the molecule above, you would have to check if it is a longer continuous chain going straight across the row or is it longer if you were to follow the branch with 2 carbons that branches below the top row.
-
-
- In this example the longest carbon chain (highlighted in red) has 7 carbons. Therefore the root name of this molecule is heptane
-
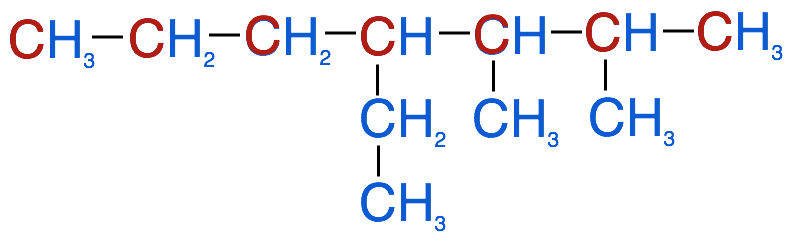
2. Identify all of the branched groups (also known as the substituent groups) attached to the parent chain
-
-
- In this example there are 2 methyl groups (CH3) and 1 ethyl group (C2H5)
-
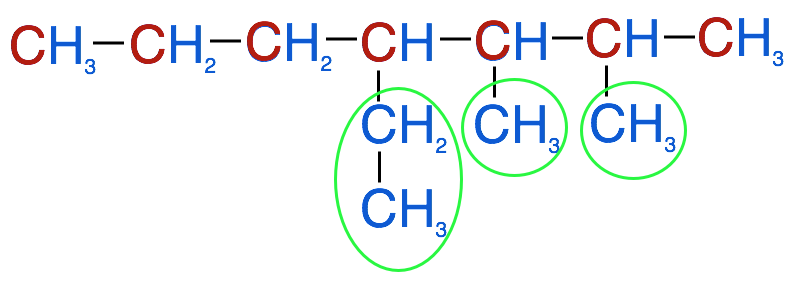
3. Number the parent chain's carbons from the end so that the substituents are attached to the lowest possible numbers.
-
-
- In this example, a methyl group is attached at carbons 2 and 3 and the ethyl group is attached at carbon 4. We will use these numbers to identify location of each substituent group, so in our case we have 2-methyl, 3-methyl and 4-ethyl
-
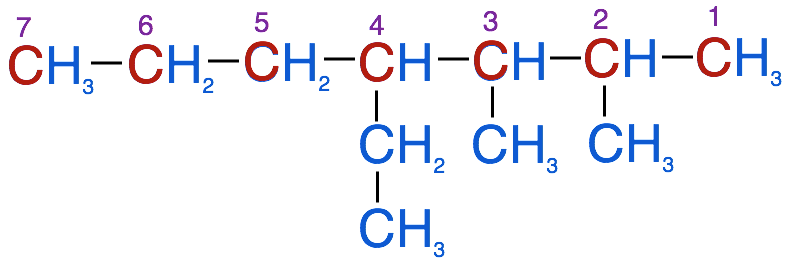
-
-
- Note if you were to begin numbering the carbons in the other direction, you would end up with the ethyl group on carbon 4 and the methyl groups on carbons 5 & 6. This would violate the rule of having the substituents on the lowest possible numbered carbon.
-
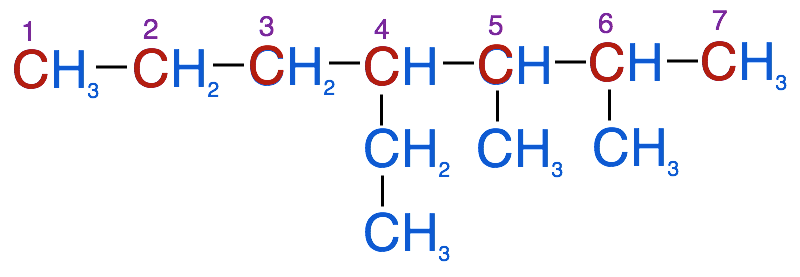
4. If there is more than one of the same substituent type, in this example we have 2 methyl groups, we use a prefix to indicate the total number of that substituent (2 = di-, 3 = tri-, 4 = tetra-) and a number to indicate the location of each substituent
-
-
- In this example we have 2 methyl groups, located at carbons 2 and 3, so we use the prefix di- and the numbering system 2,3. Combine these 2 rules and we have 2,3-dimethyl to identify the 2 methyl groups.
- Note: when naming organic compounds commas are used to separate numbers and dashes (-) are used to separate numbers and words
-
5. List the names of the substituents in alphabetical order, ignoring the prefixes
-
-
- In our example, we will list 4-ethyl first, followed by 2,3-dimethyl followed by the root name: heptane. So for this molecule, the proper IUPAC name is:
-
4-ethyl-2,3-dimethylheptane
Check your understanding
Question 2-1B-1: Name the following alkanes using IUPAC convention
a)
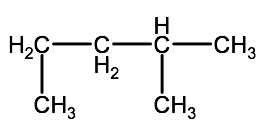
Click here for a link to a video showing the solution to this problem
b)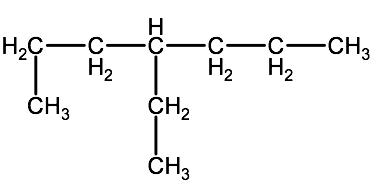
Click here for a link to a video showing the solution to this problem
c)
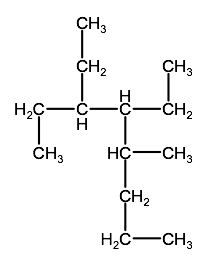
Click here for a link to a video showing the solution to this problem
Answer 2-1B:2
D. Translating IUPAC Names into Structural Formulas
Now that we know how to name complex alkanes from their structural formulas, we can use this knowledge to translate IUPAC names into structural formulas.
Watch this video for a tutorial on how to convert IUPAC names into structural formulas, followed by the step by step instructions with images.
Here we go. Example convert the IUPAC name: 2,3-dimethylhexane into a structural formula.
1. Find the root word in the name, then draw the corresponding carbon chain to create the parent chain.
-
-
- 2,3-dimethylhexane: our root is hexane which we know is a 6 carbon chain
-
![]()
2. Number the carbons in the chain
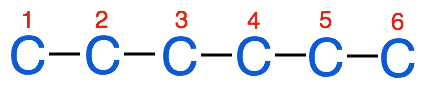
3. Identify the substituent groups and attach them to the parent chain at the appropriate carbon
-
-
- 2,3-dimethylhexane: in this example we have 2 methyl groups (di-methyl) located at carbons 2 and 3
-
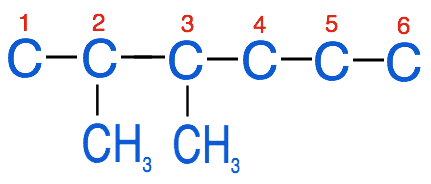
4. Add hydrogen atoms to any carbons that aren't saturated
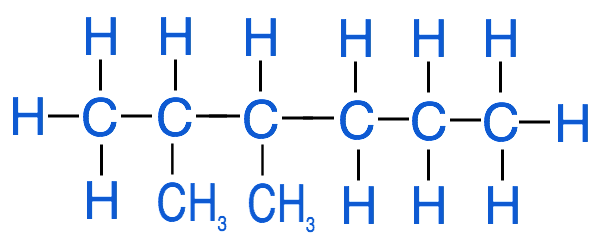
Check your understanding
2-1B-3: Draw structural formula for the following molecules
a) 3-ethyloctane
b) 2,4-dimethyl-4-propyldecane
Answer 2-1B:3
E. Naming Cyclic Alkanes
Naming cyclic alkanes from structural formulas is the same process as naming linear and branched alkanes. The root name comes from the number of carbon atoms that form the ring, preceded by the word cyclo-. Carbon atoms are numbered in such a way so that any substituent atoms are on the lowest possible numbered carbons. For example the molecule below would be named 1,2-dimethylcyclohexane
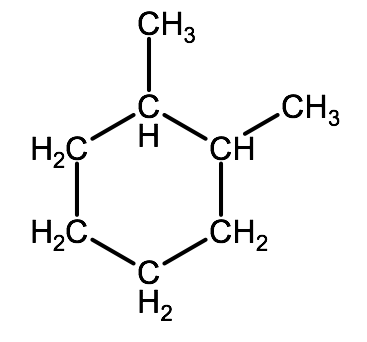
F. Summary
- Alkanes are saturated hydrocarbons with a general formula of CnH2n+2
- Simple alkanes are named using a prefix that denotes the number of carbons in the alkane
- Complex alkanes are named using:
- the longest continuous carbon chain. This is known as the parent chain and will form the root name of the molecule
- the number of carbons in each branched alkyl group
- the carbon in the parent chain that a branched alkyl group branches from
G. External Resources
If you would like to view some more examples with different method, please view the following below:
Organic Chemistry Naming Examples (Naming)
More Organic Chemistry Naming Examples (Naming)
Start with Name and Draw Molecular Structure
Alkane and Cycloalkane Nomenclature I