SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-2B: Alkenes & Alkynes
Description
In this activity you will learn the correct procedures for how to name and draw more complex hydrocarbons, containing double and/or triple bonds, known as alkenes and alkynes.
Page content
A. Introduction
Alkenes & Alkynes are organic compounds that fall into the category of unsaturated hydrocarbons. Unsaturated in this sense means that not all of the carbons in these compounds are bound to 4 other atoms. In an alkene, there is at least 1 carbon that has a double bond and in alkynes, at least 1 carbon has a triple bond. As you may have guessed, these structural differences affect the properties of these hydrocarbons, which we will explore in a later learning activity. Unsaturated hydrocarbons have many uses in everyday life, for example ethyne, which is the simplest alkyne, is found in the torches used for welding metals, it is commonly referred to as acetylene.
B. Naming Unsaturated Hydrocarbons
As we saw in the previous learning activity, because of the sheer number of different types of organic compounds, chemists rely on a structured and formulaic naming system to identify the many different compounds and this applies to the unsaturated hydrocarbons as well. The naming system for alkenes and alkynes is very similar to that for alkanes.
Watch the following video for a tutorial on how to name alkenes and alkynes, followed by step by step instructions below:
Step 1. Identify the parent chain or ring that contains the double or triple bond. This will be used for the last part of the name. For example, if the parent chain has 5 carbons, we will use the prefix pent-.
Step 2. Identify whether the parent chain contains a double or triple bond, if there is a double bond, we will use the suffix -ene, if a triple bond -yne. If there is more than one double or triple bond, we will identify this using the di-, tri- and tetra- convention. For example if there are 2 double bonds we will use the identifier: -diene, if there are 3 double bonds we will use the identifier: triene.
Step 3. Number the parent chain or ring carbons so that the first carbon with a double or triple bond has the lowest number possible. Write this number before the suffix. As with alkanes, if there are for example 2 double or triple bonds, we will identify this by using the numbers where the bonds are, separated by commas. For example 2,3-pentadiene.
Step 4. Using the same rules as we did for alkanes, name and number any substituent groups.
Lets go through an example to try these steps out.
Name the following organic compound:
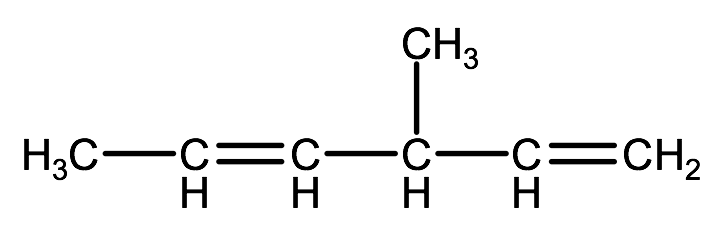
Step 1. Identify the parent chain or ring that contains the multiple bond, and count the number of carbons.
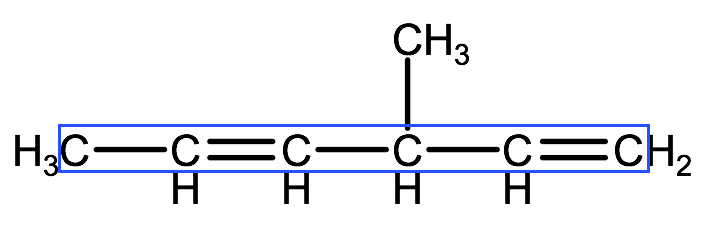
In this example, the parent chain has 6 carbons, so the end of the name will have the prefix: hexa.
Step 2. Identify whether the parent chain contains a double or triple bond. If there is more than one double or triple bond, we will identify this using the di-, tri- and tetra- convention. In this example, there are 2 double bonds so our name will end in -diene
Step 3. Number the parent chain or ring carbons so that the first carbon with a double or triple bond has the lowest number possible.
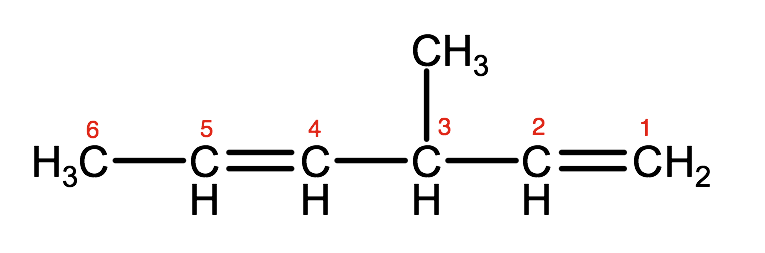
So for this example, we have double bonds on carbon 1 and 4. Therefore the last part of the name will be 1,4-hexadiene
Step 4. Using the same rules as we did for alkanes, name and number any substituent groups.
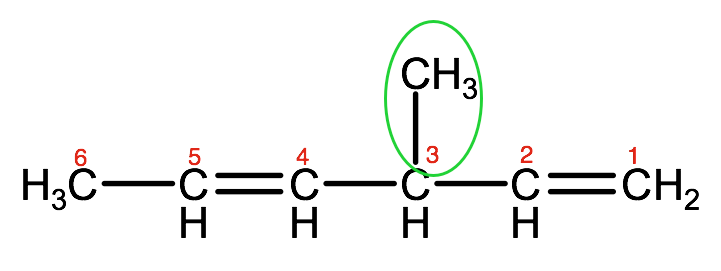
In this example, there is a methyl group on carbon 3, therefore we will identify this as 3-methyl
Put this all together and you have the full IUPAC name for this organic compound:
3-methyl-1,4-hexadiene
Check your understanding
2-1C-1: Write the names of the following compounds using IUPAC convention
a)
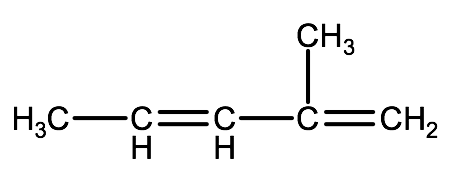
b)
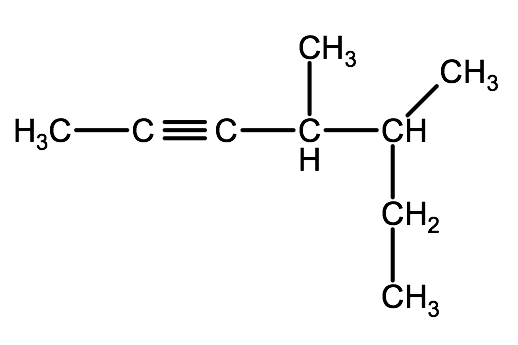
Check your answer: 2-1C:1
C. Naming Cyclic Alkanes & Alkenes
For cyclic alkenes and alkynes we follow the same convention as for alkanes. For the organic compound below, the IUPAC name would be 1-ethyl-1,3-cyclopentadiene
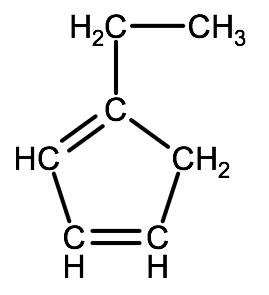
Check your understanding:
2-1C-2: Name the following hydrocarbon:
a)
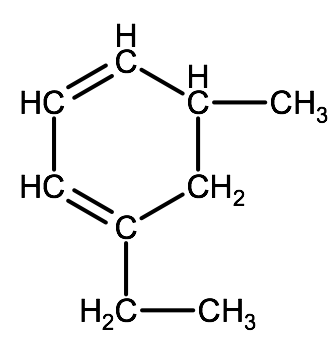
Check your answer: 2-1C:2
D. Drawing Alkenes & Alkynes
Drawing unsaturated hydrocarbons from IUPAC names is the same process as we learned for alkanes, watch the tutorial below for instructions followed by a step-by-step example.
Example: 4-methyl-1,2-pentadiene
Step 1. Draw the parent chain or ring from the last part of the compound name. In this example we start with pentadiene which implies a 5 carbon chain
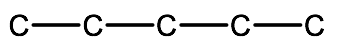
Step 2. Add in the multiple bonds to the appropriate carbons. In this example, we have 1,2-pentadiene which implies 2 double bonds (-diene) located on carbons 1 and 2
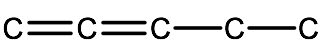
Step 3. Add any substituent groups followed by adding hydrogen atoms to any unfilled carbons.
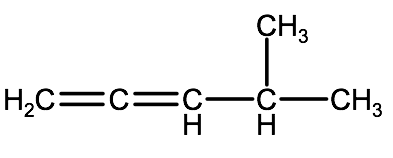
Check your understanding:
2-1C-3 Draw the structural formula's for the following IUPAC names:
a) 4-methyl-2-pentyne
b) 6-methyl-2,4-heptadiene
Check your answer: 2-1C:3
E. Summary
- Alkenes and alkynes are unsaturated hydrocarbons that have at least one double-bond (alkene) or triple bond (alkyne)
- IUPAC naming convention for alkenes and alkynes is the same as for alkanes
F. External Resources
If you would like to view some more examples with different method, please view the following below:
Cis-Trans and E-Z Naming Scheme for AlkenesAlkene Structure and Classification