SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-2C: Naming & Drawing Alcohols & Ethers
Description
In this activity you will learn the correct procedures for how to name and draw simple and complex alcohols and ethers.
Page content
A. Naming Alcohols
Alcohols are organic compounds that contain hydroxyl (-OH) groups. In IUPAC convention, all alcohols can be identified using the suffix -ol, such as metanol, propanol. Thus if you come across the name of any organic compound ending in -ol you know it must contain a hydroxyl group.
The naming system for alcohols is very similar to that for alkanes with the basic steps as follows:
- Identify the longest chain or ring (the parent chain). This will form the root name of the compound
- Count the number of -OH groups. If there is only one, the compound will have the suffix -ol.
- If there are two -OH groups the suffix will be diol, if three -OH groups the suffix will be triol
- Number the parent chain so that the -OH group is on the lowest numbered carbon atom
- Identify any substituents by name and the number of the carbon it branches from
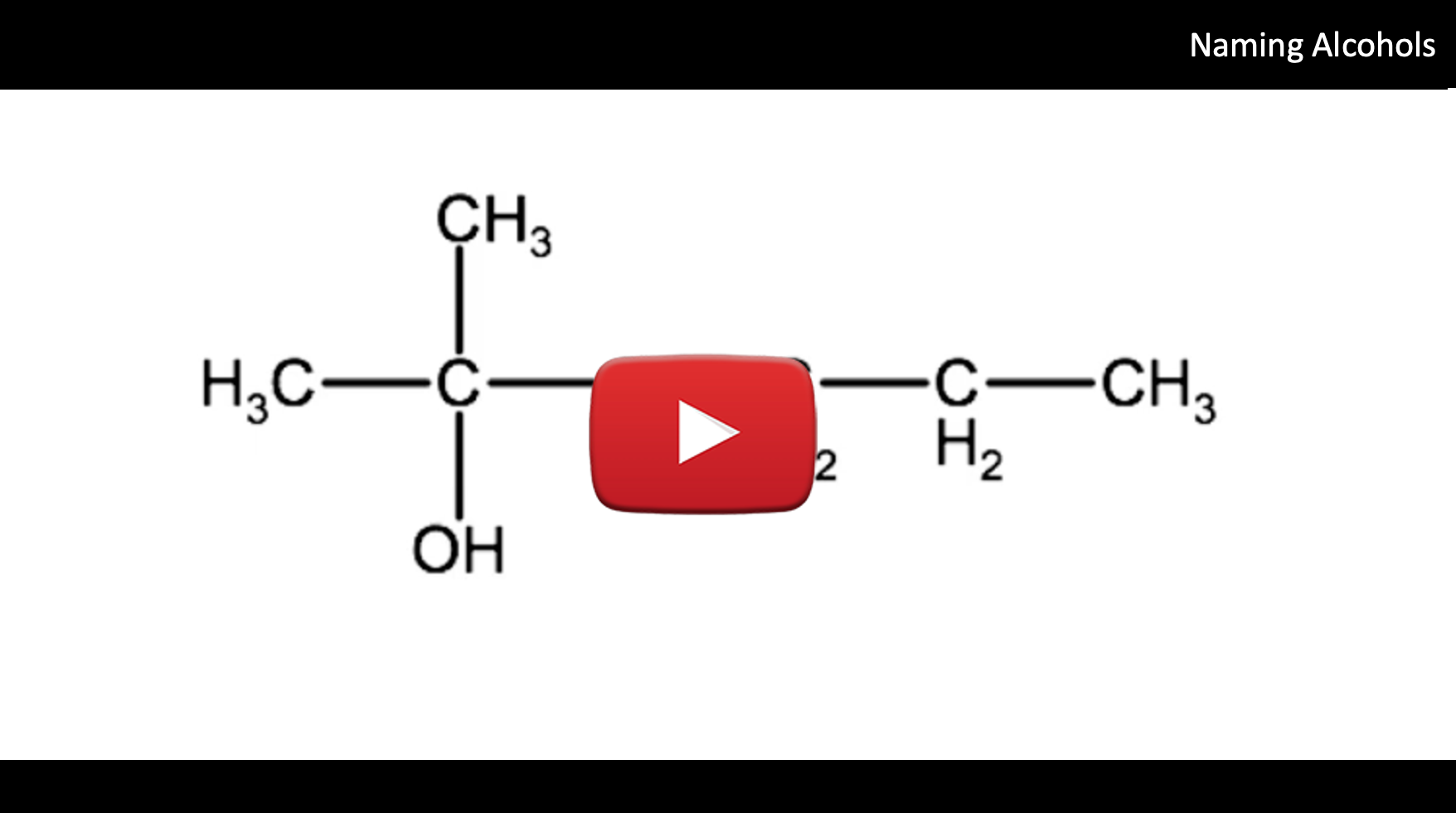
Watch the following video for a tutorial on how to name alcohols, followed by the step by step instructions:
Example: Name the following organic compound
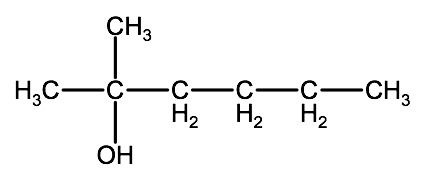
1. Identify the longest chain.
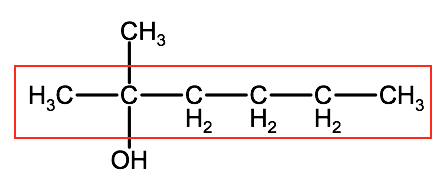
In this example the parent chain has 6 carbons, therefore the root name is hexane.
2. Count the number of -OH groups.
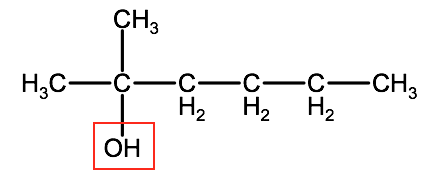
This molecule has only one -OH group, therefore this compound is a hexanol.
3. Number the parent chain so that the -OH group is on the lowest numbered carbon atom.
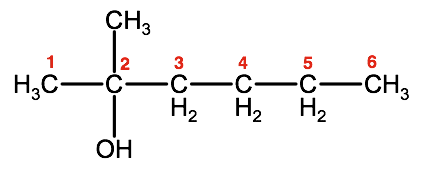
The -OH group in this molecule is on the #2 carbon. Therefore the -OH will be identified as 2-hexanol.
4. Identify any substituents by name and the number of the carbon it branches from
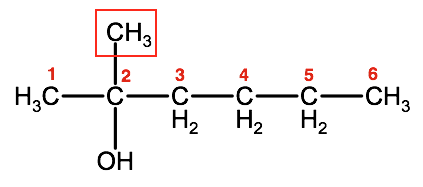
This molecule has a methyl group on the #2 carbon. This will be identified as 2-methyl
Combine all the elements and the IUPAC name for this alcohol is: 2-methyl-2-hexanol
Check your understanding:
Name the following molecules:
a)
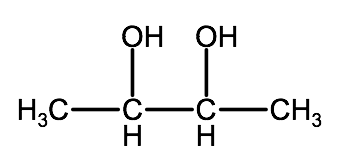
b)
Check your answers 2-2C:1
B. Drawing Alcohols
When drawing alcohols from IUPAC names we simply need to unpack the name piece by piece
Watch this tutorial video followed by the step by step instructions on how to name alcohols:
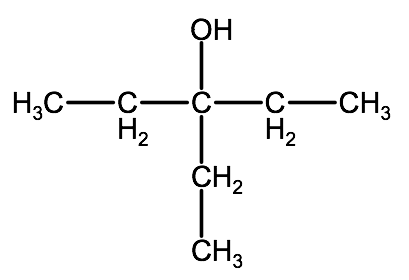
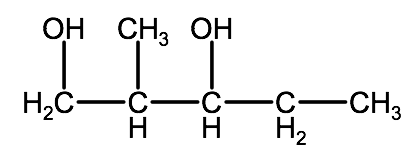
Example: 2-methyl-1,3-pentanediol
1. Determine the parent chain or ring:
2-methyl-1,3-pentanediol - pentane indicates a 5 carbon parent chain
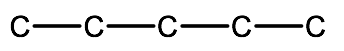
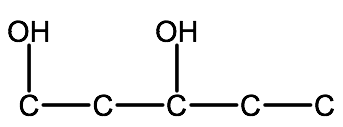
2. Determine the -OH groups:
2-methyl-1,3-pentanediol: -diol indicates there are two -OH groups, located on carbons 1 & 3
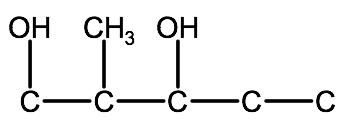
3. Determine the name and location of any substituent groups:
2-methyl-1,3-pentanediol: 2-methyl indicates a single methyl group branching from carbon 2
4. Fill up any unsaturated carbons with hydrogen atoms
Check your understanding:
Draw the following molecules:
a) 2,3-dimethyl-1-pentanol
b) 5-propyl-2,3-octadiol
Check your answers: 2-2C:2
C. Naming ethers
As we learned earlier, ethers are organic compounds in which an oxygen atom is bound on either side by carbon atoms within a carbon chain or ring. This essentially means that the oxygen atom is bordered by two alkyl groups. So to name ethers we must simply identify the two alkyl groups on either side of the oxygen atom. For IUPAC convention we simply take the name of the shorter alkyl group add the suffix -oxy followed by the name of the longer alkyl group as if it were an alkane. We then indicate the carbon number of the longer chain that the oxygen is bound to.
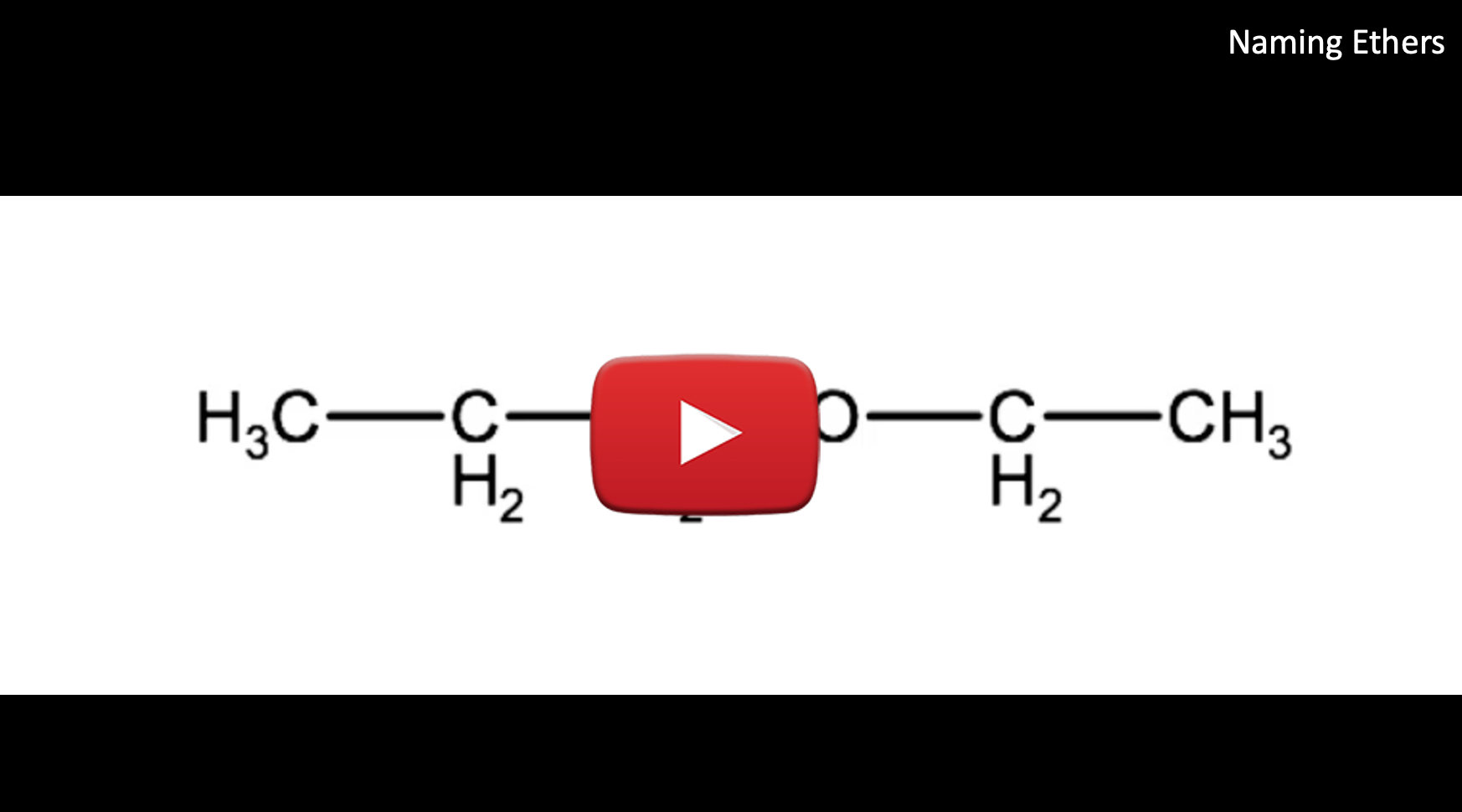
Look at the following molecule and try to determine its name before watching the video and looking at the step by step instructions.
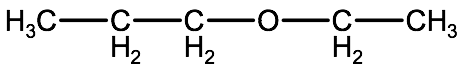
Watch this video for a tutorial on how to name ethers, followed by the step by step instructions:
1. Identify the alkyl groups the oxygen is bound to:
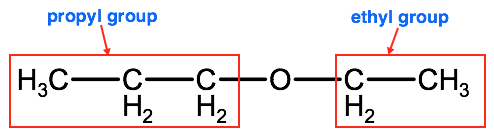
2. Take the shorter alkyl group and add the suffix -oxy, add this name to the longer chain written as an alkane. In this example the shorter group is an ethyl so we will start the name with ethoxy and combine it with propane, which is the full alkane name for the alkyl group propyl. This gives us ethoxypropane
3. Indicate the carbon number that the oxygen is bound to. in this case it is carbon 1 of the propyl group. This may sound confusing but if we rearrange the configuration slightly you will see why this is carbon 1:
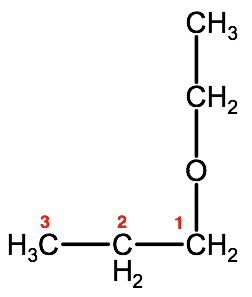
Therefore this ether would have the IUPAC name: 1-ethoxypropane
Check your understanding:
Name the following molecule:
a)
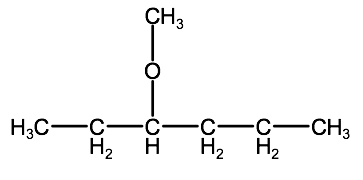
Check your answer:
2-2C:3
D. Drawing Ethers
We go through the same process as drawing alcohols from names, working backwards. For example: 3-ethoxypentane.
Watch this video for a tutorial on how to draw ethers, followed by the step by step instructions:
1. Determine the two alkyl groups on either side of the oxygen atom.
3-ethoxypentane: pentyl is the longer alkyl and ethyl is the shorter one.
2. Determine the location of the oxygen atom.
3-ethoxypentane: the ethyl group branches off the pentyl group at carbon 3. Therefore our molecule, 3-ethoxypentane, looks like:
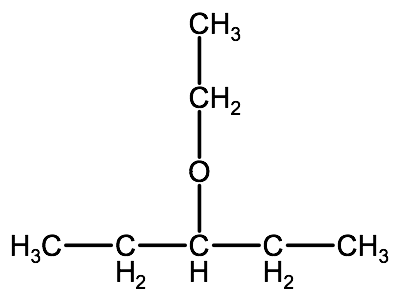
Check your understanding:
Draw the following molecule:
a) 4-methoxyheptane
Check your answer: 2-2C:4
E. Summary
- Alcohols, which are organic compounds with hydroxyl groups, end with the suffix -ol
- If there is more than one -OH group on the molecule, the suffix becomes -diol, -triol, etc.
- In alcohols, the parent chain is numbered so that the -OH group is on the lowest numbered carbon
- Ethers are organic compounds with an oxygen atom bonded to 2 alkyl groups
- The longer of the 2 alkyl groups bound to the oxygen atom is used to form the root name, using the full alkane name for that alkyl group (e.g. propyl becomes propane)
- The suffix -oxy is added to the shorter of the 2 alkyl groups and this name comes in front of the root name (e.g. ethane becomes ethoxy)
G. External Resources
If you would like to view some more examples with different method, please view the following below:
Triple Bond Cause Linear Configurations (related to the above video)
Alcohol Nomenclature (Classification and naming)