SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-2D: Aldehydes, Ketones, Carboxylic Acids, and Esters
Description
In this activity you will learn the correct procedures for how to name and draw aldehydes, ketones, carboxylic acids, and esters.
Page content
A. Naming Aldehydes & Ketones, Carboxylic Acids, and Esters
Aldehydes and ketones are organic compounds that both contain a carbonyl group (-C=O) which is a carbon atom that is double bonded to an oxygen atom. In aldehydes, the carbonyl group is bound to at least one hydrogen atom and is found at the end of a carbon chain; whereas in ketones, the carbonyl group is located somewhere within the carbon chain. Naming these two classes of organic compounds follows many of the same steps we've already seen.
When naming aldehydes we replace the last letter -e from the name of the parent chain alkane with the suffix -al, whereas when naming ketones, the last letter e of the parent alkane is replaced with the suffix -one. With aldehydes, we don't need to indicate any carbon numbers as IUPAC convention states that the carbonyl group is always located on carbon #1. With ketones, the carbon number that the carbonyl group is located only needs to be indicated for chains that are 5 carbons or longer. When numbering the parent chain in this case, always number so that the carbonyl group is on the lowest number carbon possible. Once you have determined the characteristics of the carbonyl group, the next step is to name and identify any substituent groups if they are present.
Lets go through 2 examples, but try these out yourself based on the skills you've already learned before looking at the step by step.
Example 1:
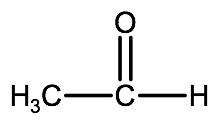
1. Since the carbonyl group here is bonded to a hydrogen atom we know that this is an aldehyde, therefore we will use the suffix -al.
2. The carbon chain is 2 carbons long, therefore the name of this compound will be: ethanal
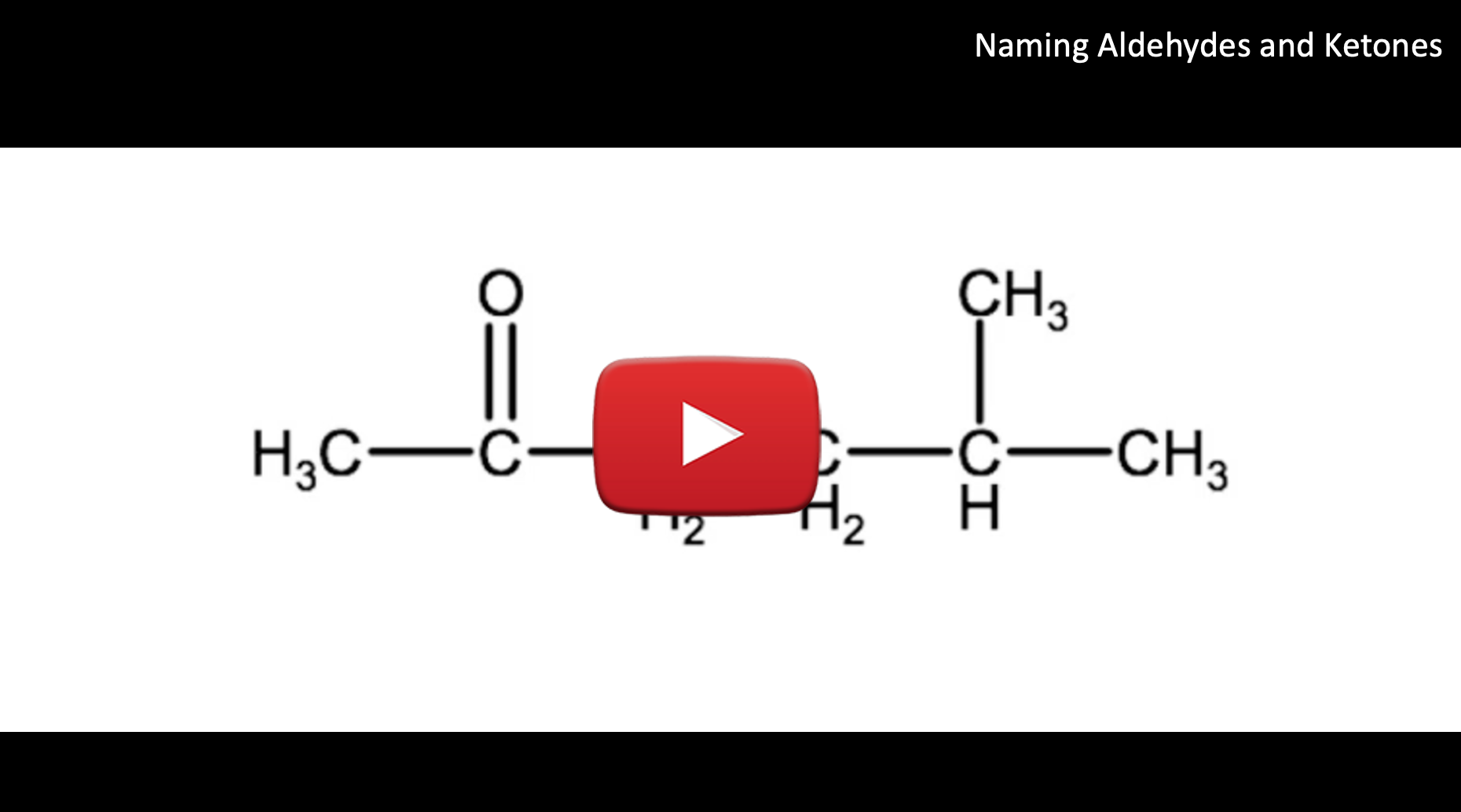
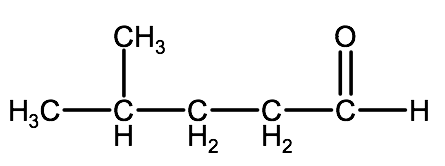
Example 2: Watch the following video for a tutorial on how to name the molecule below. Try to name it on your own before watching or reading the steps below and see if you can do it with the knowledge you have learned to this point.
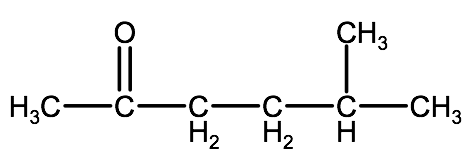
1. Since the carbonyl group is in the middle of the carbon chain we know this is a ketone, therefore we will use the suffix -one.
2. The parent chain is 6 carbons long, therefore the root name will be hexanone.
3. Because the parent chain is more than 5 carbons, we need to number them, in this case the carbonyl group is on carbon 2, therefore we have 2-hexanone
4. Name and number the substituent groups. Here we have a methyl group on carbon 5. This will be identified as 5-methyl.
5. Combine the elements and we have 5-methyl-2-hexanone
Check your understanding:
Name the following compounds:
a)
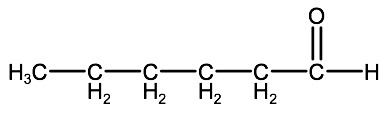
b)
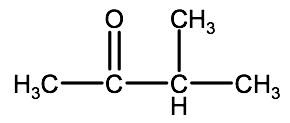
Check your answer: 2-2D:1
B. Drawing Aldehydes & Ketones
You should at this point be fairly comfortable taking an IUPAC name and converting it to a drawing, but let us go through one example each for an aldehyde and a ketone.
Watch the following video for a tutorial on how to draw the molecule 4-methylpentanal. Try drawing it out on your own first though.
How to draw 4-methylpentanal
1. The suffix -al tells us this is an aldehyde
2. The root name pentanal tells us the parent chain is 5 carbons long
3. 4-methyl indicates there is a methyl group attached to carbon number 4
Check your understanding:
Draw the following compounds:
a) 4-methyl-2-heptanone
b) 2-methylpropanal
Check your answers 2-2D:2
C. Naming Carboxylic Acids
By now you should be fairly comfortable with naming and drawing these organic molecules, so now we will move on to carboxylic acids and esters. As we learned earlier, carboxylic acids are organic compounds that contain a carboxyl group (-COOH), whereas esters are organic compounds that contain a functional group similar to a carboxyl group except that the hydrogen atom of the carboxyl group is replaced by an alkyl group. We will learn more about naming esters in a future lesson, after we have learned about the reactions that must occur to form an ester.
When naming carboxylic acids, we start with the name of the alkane of the parent chain, including the carbon atom of the carboxyl group, and we add the suffix -oic in place of the -e at the end of the name, followed by the word acid. For example in a 2 carbon chain with a carboxyl group, ethane becomes ethanoic acid. As we've seen before, once we determine the root name, we can go about identifying any substituent groups.
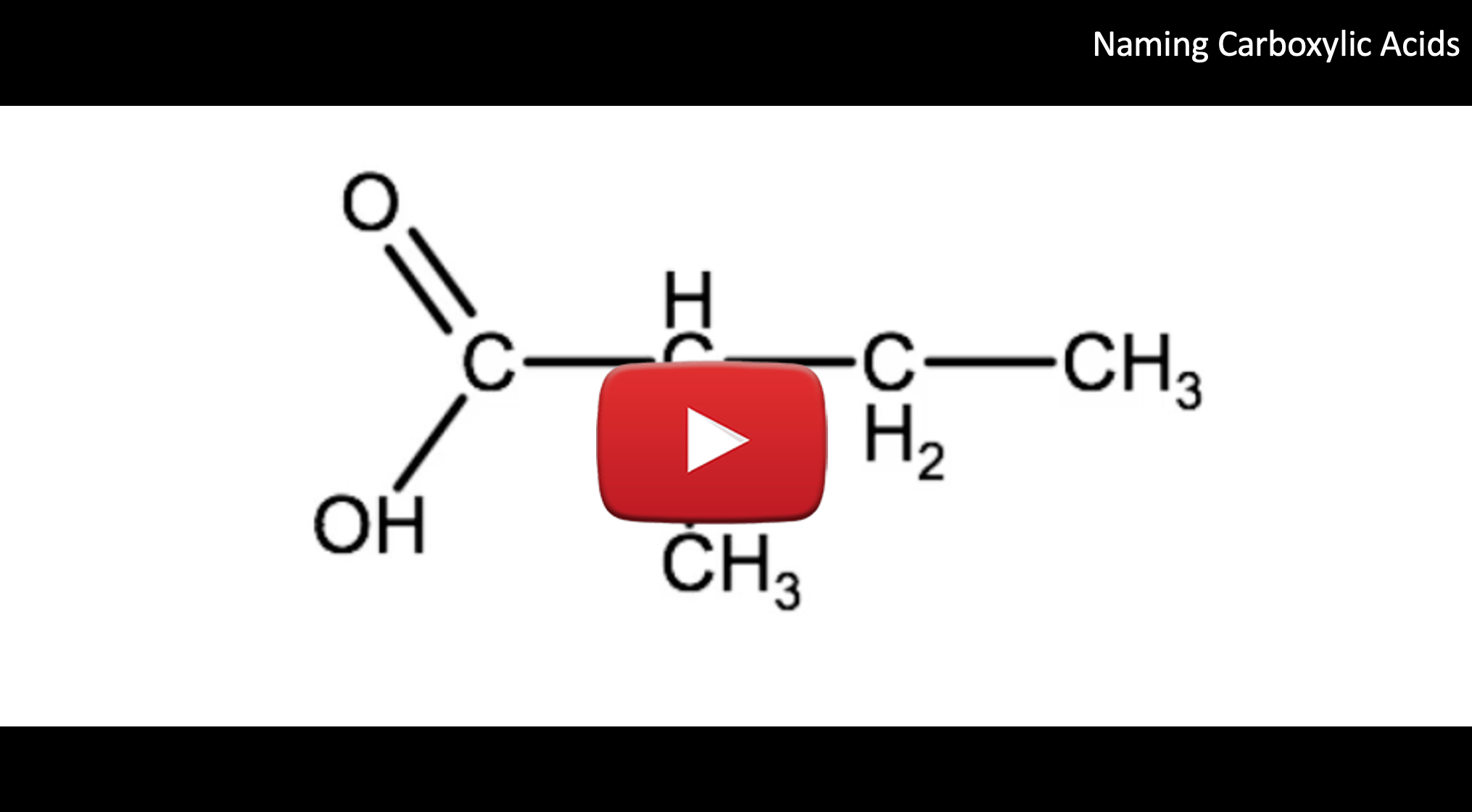
Watch the video below for a tutorial on how to name the carboxylic acid in example 1 below. Try naming this one on your own before watching the video.
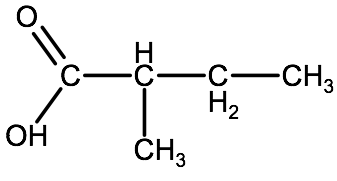
1. Identify the type of organic compound. In this case we see there is a carboxyl group on one end, so we know this is a carboxylic acid.
2. Identify the parent chain, including the carbon from the carboxyl group and add the suffix -oic acid. We have a 4 carbon parent chain (butane), therefore the root name becomes: butanoic acid
3. Identify any substituent groups. In this case we have a methyl group on carbon 2. Therefore we identify it as 2-methyl.
4. Combining everything we end up with 2-methylbutanoic acid.
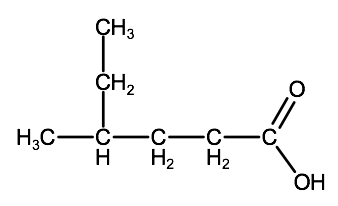
Check your understanding
Name the following compound:
a)
Check your answer: 2-2D:3
D. Drawing Carboxylic Acids
Watch the video below for a tutorial on how to name the carboxylic acid in example 2. Try drawing this one on your own before watching the video and see how you do.
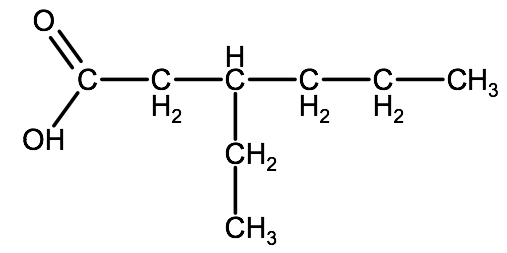
Example 2: Drawing the following molecule: 3-ethylhexanoic acid
1. Draw the root structure: 3-ethylhexane
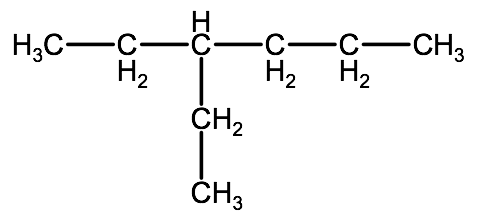
2. Change one of the carbons on the end of the chain to a carboxyl group. Note that since we have an alkyl group as a substituent and we want this to always be the lowest possible number, we draw the carboxyl group at the terminal carbon that is closest to the alkyl group.
Check your understanding
Draw the following compound:
a) 2-methylpropanoic acid
Check your answer 2-2D:4
E. Naming Esters
Esters are compounds that are responsible for the very distinctive odours produced by many fruits and vegetables. Esters are synthesized from a condensation reaction between a carboxylic acid and an alcohol. Synthetic esters are used to provide flavouring to many food and perfumes. Some smells are very pleasant like ethyl butyrate (Strawberry) or octyl acetate (orange), but some are not so nice such as ethyl acetate (nail polisher remover) .
Esters can be thought of as a carboxylic acid derivative
![]()
The R' is an alkyl or aryl (benzene-like) group (replacing the H of the acid), where the rest of the molecule is the carboxylic acid portion.
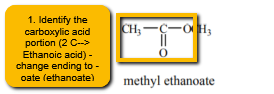
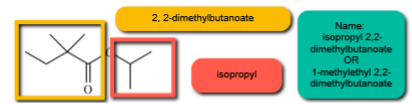
1. Locate the carboxylic acid portion of the molecule and name this as the parent carboxylic acid. Since it is an ester, you will now change the ending “anoic/ic acid' to 'oate/ate'. Ex. acetic -> acetate, propanoic -> propanoate
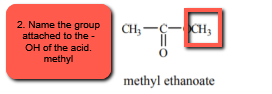
2. Name the rest of the molecule that is attached to the carboxylate oxygen (oxygen that would be the -OH group) using normal naming conventions. NOTE: carbon 1 is the Carbon attached to the oxygen. (e.g., methyl, ethyl, isopropyl, etc.).
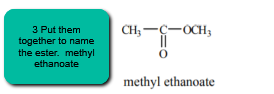
3. Put them together! The name of the group attached to the carboxylate oxygen is first! The name of the carboxylic acid portion comes second.
 |
 |
 |
4. If there are side chains: use normal naming conventions. The carbonyl carbon of the acid portion is numbered 1!
F. Drawing Esters
1. The -oate in the name tells you the makeup of the parent chain. The parent chain/ carboxylic acid is the portion that contains the carbonyl. Draw the carboxylic acid structure for this.
2. Drop the -H on the -OH group of the carboxyl and add in the alkyl group to the carboxylate oxygen.
3. Add any substituents on the appropriate carbons of the parent chain or other group. Remember the carbonyl C is numbered 1 on the carboxylic side. On the alkyl group, C1 is the C attached to the O.
Check your understanding:
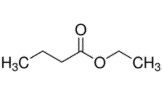
Name the following compounds:
a)
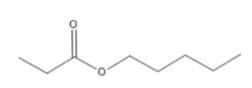
b)
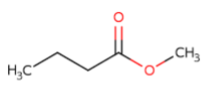
|
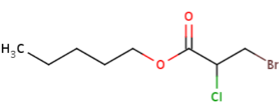
Try drawing the following:
a) ethyl butanoate
b) pentyl 3-bromo-2-chloropropanoate
|
G. Summary
- When naming aldehydes, the -e at the end of the root chain alkane name is replaced with -al
- When naming ketones, the -e at the end of the root chain alkane name is replaced with -one
- When naming carboxylic acids, the -e at the end of the root chain alkane name is replaced with -oic acid
G. External Resources
If you would like to view some more examples with different method, please view the following below: