SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-2E: Amines & Amides
Description
In this activity you will learn the correct procedures for how to name and draw organic compounds containing nitrogen atoms known as amines and amides.
Page content
A. Naming & Drawing Amines
Amines and amides are two classes of organic compounds that contain nitrogen atoms. To derive the names of amides, you must know about the reactions necessary to produce them, which we will learn in a later lesson, for now you only need to know how to name and draw amines. Amines are compounds that are derived from ammonia (NH3) where one or more of the hydrogens of the ammonia atom are replaced by alkyl groups. Amines are classified as primary (1), secondary (2) or tertiary (3), depending on how many alkyl groups are attached to the nitrogen atom. The general IUPAC naming convention for primary amines is to take the root name of the single alkyl group that is bound to the nitrogen atom, remove the -e from the end of that name and add -amine.
Primary Amines
For example, a compound with the formula CH3NH2 and the structural formula:
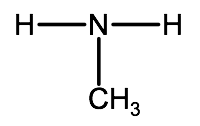
would simply be called methanamine.
Secondary Amines
For secondary and tertiary amines, we begin with the longest alkyl group bound to the nitrogen, this is our root name. For every additional alkyl group, we identify it with the capital letter N- as a prefix.
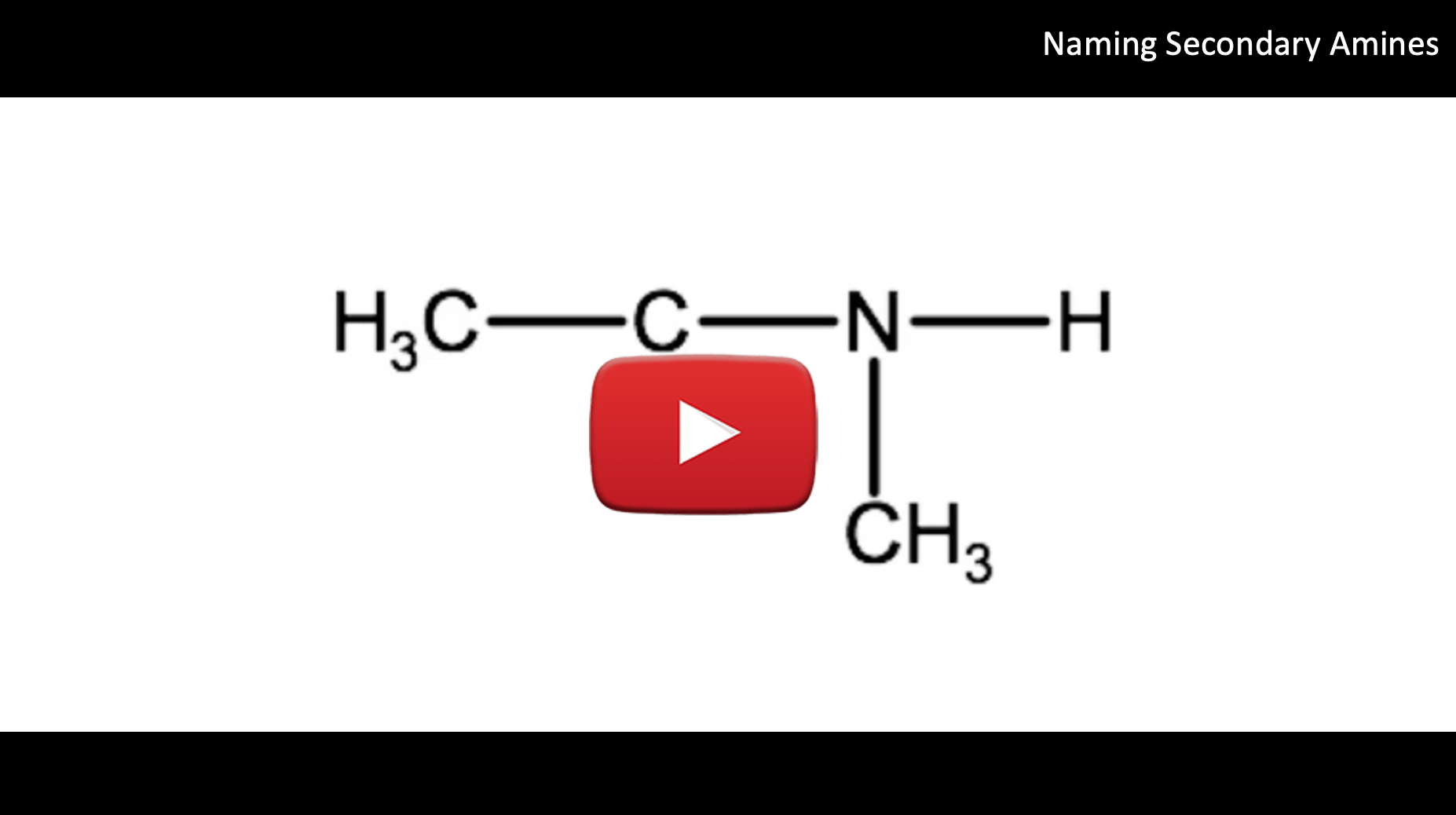
Watch the following video for a tutorial on how to name the secondary amine in the example below:
For example if you have the following molecule:
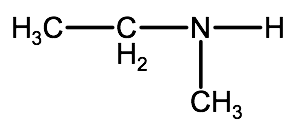
The longest alkyl group is an ethyl group, so our root name becomes ethanamine. We also have a methyl group and a hydrogen atom attached to the nitrogen, so this is a secondary amine. We identify the methyl group using the indicator N-methyl. Therefore when we combine the two parts of the name we end up with N-methylethanamine.
Note if you have 2 of the same alkyl groups on a secondary amine, you represent one of them in the root name and one of them in front with the prefix N. For example the following molecule
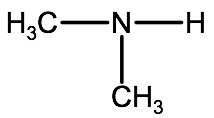
would have the IUPAC name N-methylmethanamine.
Tertiary Amines
Lets look at a tertiary amine to complete the picture now:
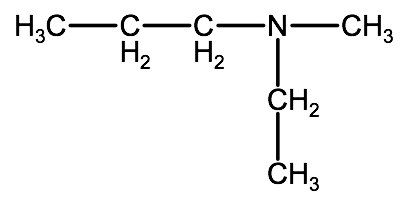
In this case, we have 3 alkyl groups bound to the nitrogen atom. The longest chain is a propyl group so our root name is propanamine. The other alkyl groups in this molecule are a methyl group and an ethyl group, again we will use the indicator N- for each of these so that we know this is an amine, giving us N-methyl and N-ethyl. When we combine everything we list the substituents alphabetically as we learned earlier for alkanes. So, combining our root name with our substituent identifiers, we end up with the following IUPAC name:
N-ethyl-N-methylpropanamine
Note if there are 3 of the same type of alkyl groups in a tertiary amine, we use the root name system for one of the substituents and we group the other 2 substituents together using the prefix di-, with N,N preceding it. For example in an amine with the following structure,
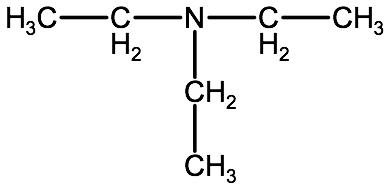
We would use the root name ethanamine, and we would group the other 2 ethyl groups as N,N-diethyl, combining them together to form the name N,N-diethylethanamine.
Check your understanding
Name the following compounds:
a)
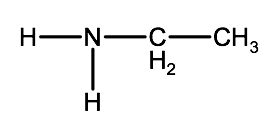
b)
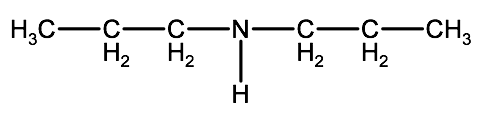
c)
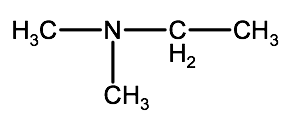
Check your answer 2-2E:1
B. Drawing Amines
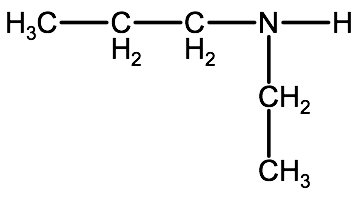
Example: Draw the following organic molecule: N-ethylpropanamine.
1. We can identify that it is an amine by the suffix -amine; furthermore, the presence of one N- in front of the name tells us it is a secondary amine.
2. Identify the alkyl groups. We know it is a secondary amine, meaning the structure will have a nitrogen atom bonded to 2 alkyl groups and one hydrogen atom. The propanamine in the name tells us there is a 3 carbon alkyl group and the N-ethyl tells us there is also a 2 carbon alkyl group.
3. Piece the elements together using the nitrogen atom as your base:
Check your understanding
Draw the following compound:
a) N,N-dimethylmethanamine
Check your answer 2-2E:2
C. Summary
- Amines are organic compounds that are derived from ammonia in which a nitrogen atom is bonded to one or more alkyl groups
- Amines are classified as primary (1), secondary (2) or tertiary (3), depending on the number of alkyl groups they are bound to
- Primary amines have the suffix -amine added to the name of the alkyl groups alkane name (e.g. propanamine)
- Secondary amines have the root name of the longest alkyl group with the suffix -amine added. The shorter alkyl group is identified using the letter N-, if both alkyl groups are the same length, one is used as the root name and the other as the first name
- Tertiary amines take the root name of the longest alkyl group, the other two are each identified by N-, if 2 of the 3 groups are the same, the are identified as N,N-di-
G. External Resources
If you would like to view some more examples with different method, please view the following below:
More Examples of Naming Amines
Nomenclature and Properties of Amides