SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-2F: Summary
This page is part of the content downloaded from 2-2F: Summary on Monday, 3 March 2025, 9:20 AM. Note that some content and any files larger than 50 MB are not downloaded.
Description
2-2F: Summary
Page content
Summary
Alkanes
-
- Unsaturated hydrocarbons
- First part of name identifies number of carbons (meth-, eth-, prop-, but-, pent-, hex-, hept-, oct-, non-, dec-)
- Have the suffix -ane
- Alkyl groups are chains that branch off of root chains, these have the suffix -yl
Alcohols
-
- Alcohols, which are organic compounds with hydroxyl groups, end with the suffix -ol
- If there is more than one -OH group on the molecule, the suffix becomes -diol, -triol, etc.
- In alcohols, the parent chain is numbered so that the -OH group is on the lowest numbered carbon
Ethers
-
- Ethers are organic compounds with an oxygen atom bonded to 2 alkyl groups
- The longer of the 2 alkyl groups bound to the oxygen atom is used to form the root name, using the full alkane name for that alkyl group (e.g. propyl becomes propane)
- The suffix -oxy is added to the shorter of the 2 alkyl groups and this name comes in front of the root name (e.g. ethane becomes ethoxy)
Aldehydes
-
- Aldehydes are organic compounds with carbonyl (-C=O) groups at the terminal end of a molecule
- When naming aldehydes, the -e at the end of the root chain alkane name is replaced with -al
Ketones
-
- Ketones are organic compounds with carbonyl groups in the middle of the molecule
- When naming ketones, the -e at the end of the root chain alkane name is replaced with -one
Carboxylic Acids
-
- Carboxylic acids are organic compounds with carboxyl groups at the terminal end of the molecule
- When naming carboxylic acids, the -e at the end of the root chain alkane name is replaced with -oic acid
Amines
- Amines are organic compounds that are derived from ammonia in which a nitrogen atom is bonded to one or more alkyl groups
- Amines are classified as primary (1), secondary (2) or tertiary (3), depending on the number of alkyl groups they are bound to
- Primary amines have the suffix -amine added to the name of the alkyl groups alkane name (e.g. propanamine)
- Secondary amines have the root name of the longest alkyl group with the suffix -amine added. The shorter alkyl group is identified using the letter N-, if both alkyl groups are the same length, one is used as the root name and the other as the first name
- Tertiary amines take the root name of the longest alkyl group, the other two are each identified by N-, if 2 of the 3 groups are the same, the are identified as N,N-di-
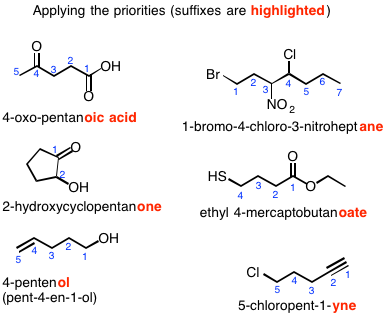
Naming Priority
When there are more than 1 functional group present on an organic compound, a priority scale must be applied when numbering the carbons for naming:

Here are some examples: