SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-3A: Physical Properties of Matter
Description
In this activity you will review how to describe and categorize the different physical properties of matter.
Page content
Properties of Matter
All matter can be described by its physical properties. In this activity you will learn about these different properties and how matter can be described through its various qualitative and quantitative properties.

In the course of your studies, you have likely encountered the term matter
as it relates to science. In science, matter is the term used for any material
that has mass and takes up space. For something to be classified as matter
it must be made up of at least one subatomic particle such as a proton or
neutron. Other examples of matter include atoms, molecules, compounds,
plants, animals, etc. In this activity, you will learn about some of the
properties of matter and the physical changes that matter can undergo.
Examples of Matter
All matter can be described by its different properties. These properties
relate to how matter is structured, how it looks, how it behaves and many
other different descriptors. The properties of matter can be divided into
two groups, physical properties and chemical properties. To begin you will
learn about the physical properties of matter and the physical changes that
matter can undergo. Lets begin by discovering the various different physical
properties that can be used to describe matter.
A physical property is any property that can be observed or measured without changing the composition of matter. These properties can be used to describe matter. Examples of physical properties include: texture, colour, smell (odour), melting point (i.e. 10 ˚C), boiling point (i.e. 100 ˚C), state and many others.

The physical properties of matter can be described in two different ways:
qualitatively or quantitatively.
Quantitative physical properties are properties
that can be measured, and generally include
numbers such as temperature, mass, height,
boiling point, etc. For example “the mass of
that sample of copper is 10 grams” or “the
melting point of water is 0 ˚C.”
Qualitative physical properties are observations
that can be made without measurement. These
describe properties that can be observed with our 5
senses (sight, hearing, smell, touch, taste) without
measuring them. For example: “bacon has a
reddish colour and a strong odour” or “that sample
of water is in its gaseous state.”
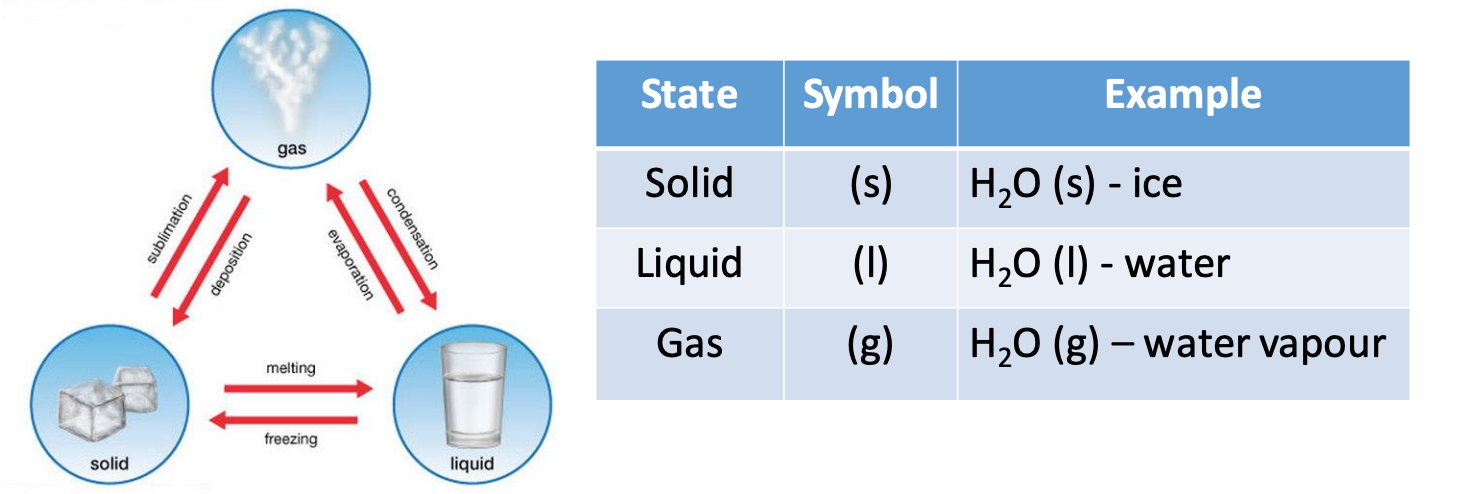
One very important qualitative property of matter is known as its state.
Matter can exist in one of 3 states: solid, liquid or gas. To indicate the
state of matter scientists typically use symbols in chemical formulae to
convey this important information as seen in the table below.
Summary
- Matter is any material that has mass and takes up space.
- Matter can be described by its qualitative and quantitative physical properties
- Quantitative properties are observations of matter that require measurement and include numbers such as boiling point.
- Qualitative properties are observations of matter that can be made without measurements such as appearance or state.
- State of matter refers to its form such as liquid: (l), solid: (s) or gas: (g).