SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-4B: Chemical Reactions: Hydrocarbons & Polymers
Description
In this activity you will explore chemical reaction that involve hydrocarbons in the products and reactants and discover how large molecules called polymers can form by linking multiple molecules of the same type called monomers.
Page content
A. Introduction
Click here for a pdf version of this lesson.
Organic compounds are involved in several different types of reactions both as the reactants and the products. Many of these chemical reactions are seen in our everyday lives. For example, alkanes undergo chemical reactions so that they can be used as fuel in everything from stovetops for heating up food, to rocket ships for sending them into space as you saw in the video above. The next few learning activities will examine some of the common chemical reactions that organic compounds are involved in both as the reactants and the products. This activity will explore combustion, substitution, addition and elimination reactions. We will begin by looking at reactions involving alkanes. Alkanes are made up of atoms that are held together exclusively by single bonds, which are very strong and stable. As a result, these compounds are generally unreactive with acids and bases for example, however they are involoved in one of the most important chemical reactions that is used every day on this planet and that is where this lesson will begin.

B. Addition Reactions - Alkenes & Alkynes
An addition reaction is a reaction in which 2 molecules react with each other and merge to form one molecule (the product) that is different from either reactant. The general equation for an addition reaction can be thought of as:
A + B → AB
For organic molecules, addition reactions are very common and there are several different subtypes that are relevant to the classes of organic compounds we have looked at so far. The video below describes 2 types of addition reactions: hydrogenation and halogenation followed by a summary in text:
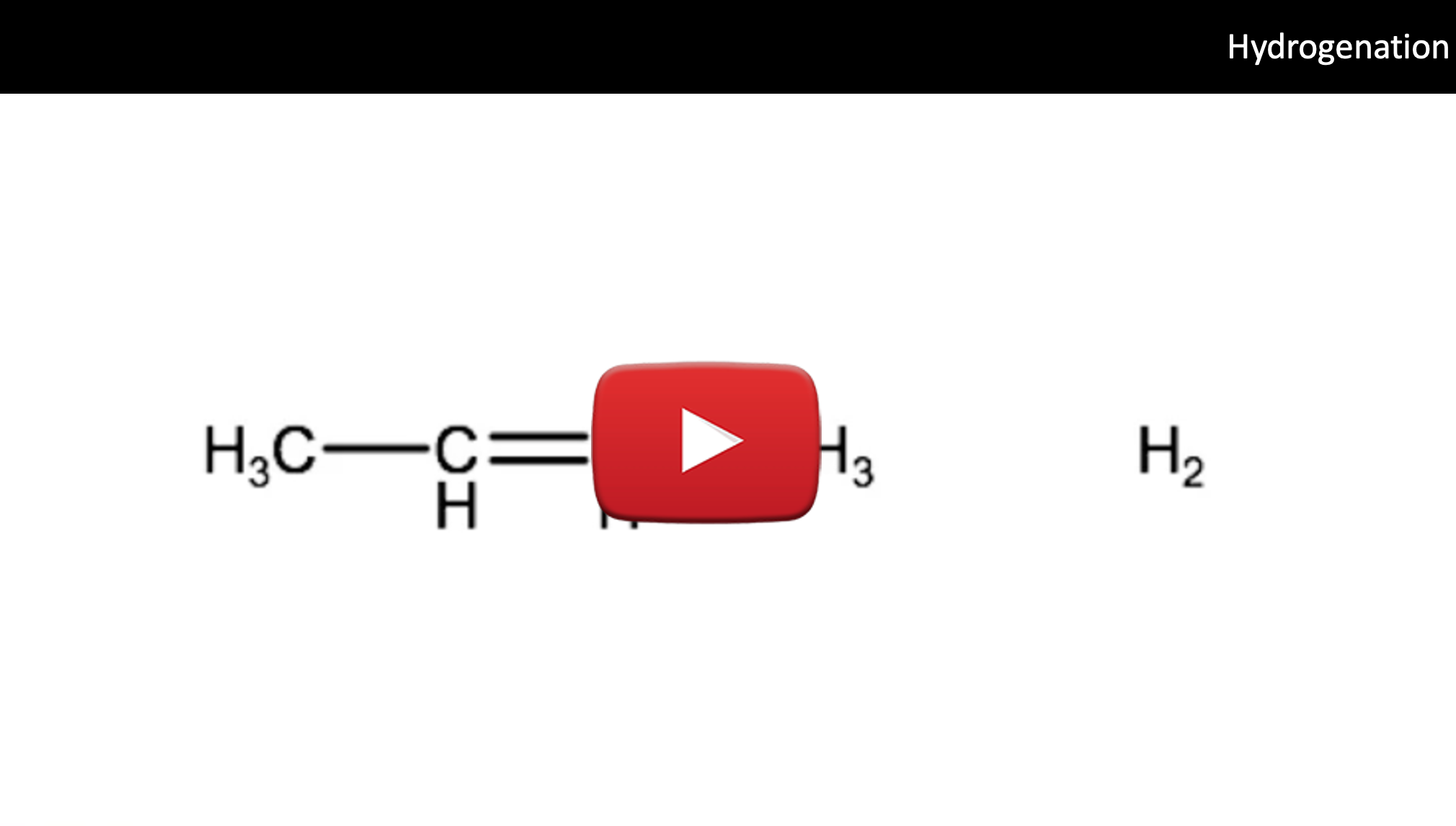
One type of addition reaction is known as hydrogenation, which can turn unsaturated hydrocarbons into saturated hydrocarbons. This type of reaction is often used in industry to convert alkenes which are somewhat reactive and can often be toxic into safer, and less reactive alkanes. Another common application of the hydrogenation reaction is to increase the saturation level of vegetable oil- an organic compound. Increasing the level of saturation by adding hydrogen atoms changes many of the physical properties of a molecule, in the case of vegetable oil, adding hydrogen atoms increases the melting point, allowing them to become solid at room temperature and therefore allowing for different commercial uses such as in margerine or peanut butter.
In a hydrogenation reaction, hydrogen (H2) molecules react with the multiple bonds of an alkene or an alkyne to produce a fully saturated molecule or an alkene, for example:
2-butene + hydrogen → butane
This can also be written as:
CH3-CH=CH-CH3 + H2 → CH3-CH2-CH2-CH3
These hydrogenation reactions can occur with alkenes, alkynes and cyclic alkenes.
Halogenation
Another type of addition reaction that unsaturated hydrocarbons undergo is called a halogenation reaction. Recall that the halogens are the elements that are found in the second last column of the periodic table (F, Cl, Br, etc). Similar to hydrogenation, in these addition reactions, halogen molecules in the form of either diatomic halogen molecules, (ie Cl2 or Br2) or hydrogen halides (ie HBr, HCl) react with the multiple bond of an alkene, alkyne or cycloalkane. Look at the first reaction below between butyne and bromine, based on what we just learned above about naming alkylhalides, can you predict the name of the product?
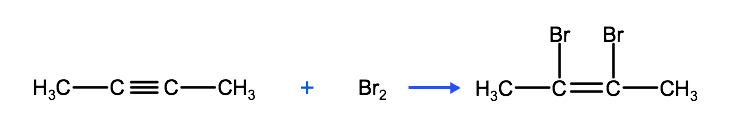
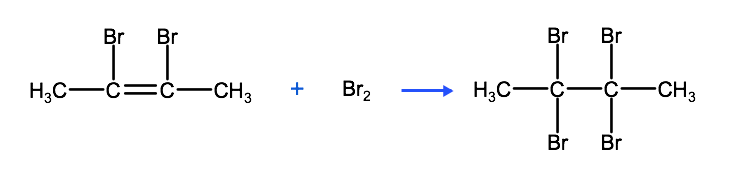
The reaction above produces the organic compound 2,3-dibromobutene, which can also react with bromine to produce 2,2,3,3-tetrabromobutane, as we see below.
Hydration
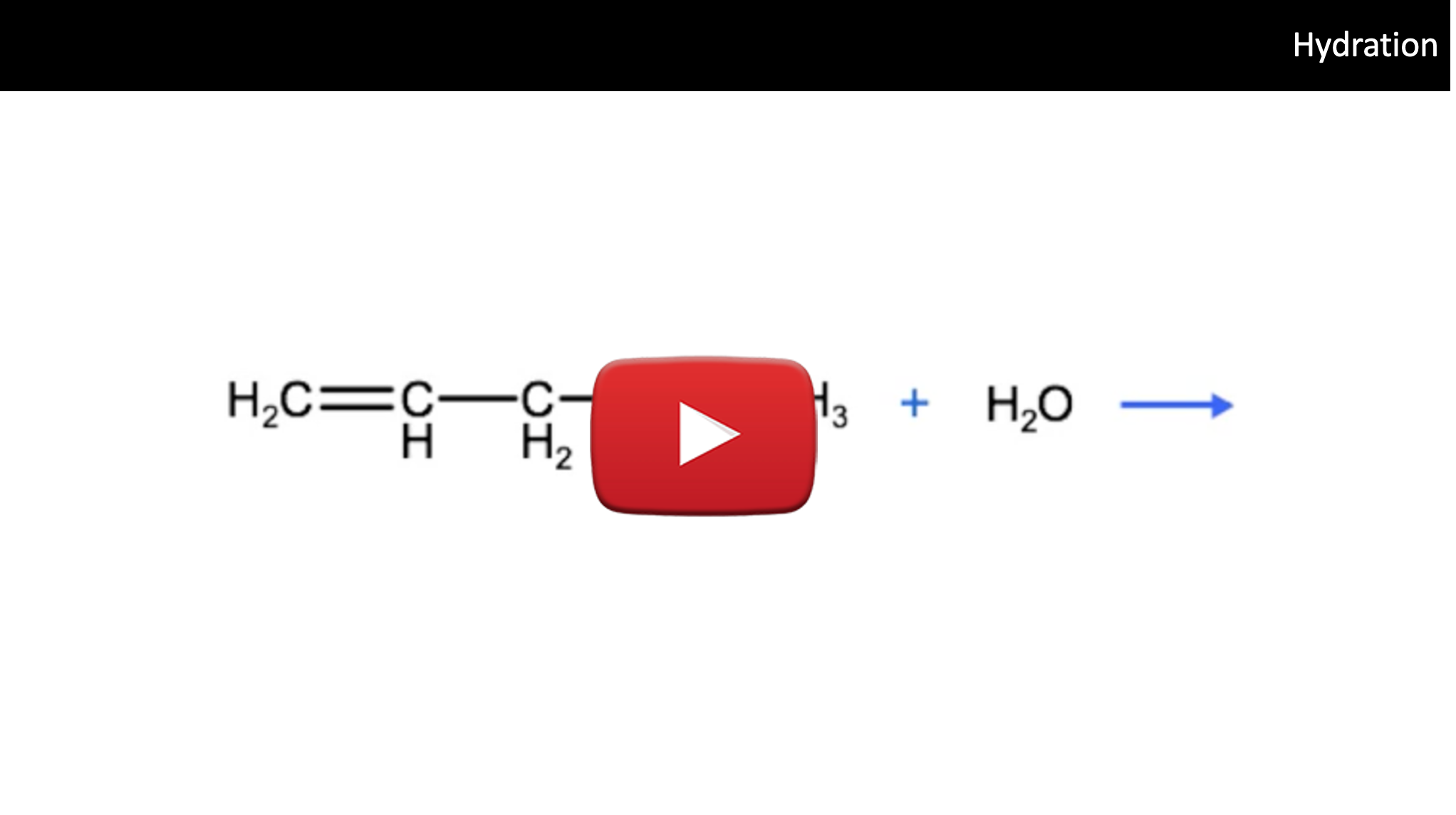
A third type of addition reaction is known as hydration. In this case, a molecule of H20 reacts with an unsaturated hydrocarbon in the presence of a catalyst and an acid to produce an organic compound that has a hydroxyl group bound to it. Do you remember what we call these? Take a look at the reaction below and name the product that forms:
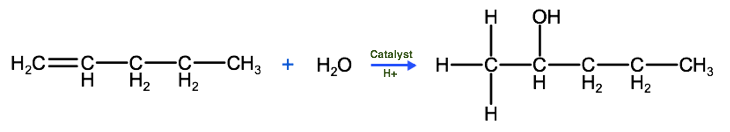
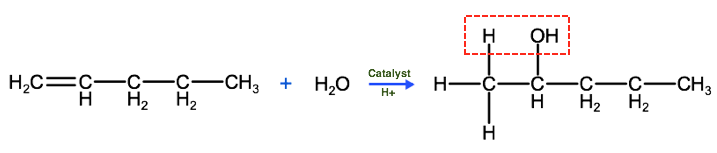
The correct IUPAC name for the product above is 2-pentanol, which as we know is in the class of organic compounds known as alcohols. So keep this in mind, that one way to produce alcohols is by the hydration of alkenes. One important thing to keep in mind, when considering hydration reactions and hydro halogen reactions is that we need to be able to predict which carbon the hydrogen atom will bond to and which atom the hydroxyl group (-OH) or halogen atom will bond to. To help predict this we can use Markovnikov's rule, which states that the hydrogen atom in a water molecule or hydrogen halide will tend to bond to the carbon atom that has the most hydrogen atoms bonded to it. So, as we see below, the hydrogen atom from the water molecule bonds to the terminal carbon, which was bound to 2 carbons before the reaction, while the hydroxyl group bonded to the second carbon, which had only 1 hydrogen atom bound to it before the reaction.
C. Combustion Reactions:
Alkanes are typically used as sources of fuel because there is a tremendous amount of energy stored within these molecules. When they are exposed to the proper conditions, this energy can be released and harnessed to help make cars move, power electrical generators and even help send rocket ships into outer space. Next time you see a car drive by or see a plane fly over head, realize that you are witnessing a chemical reaction occuring right in front of your eyes, and the type of chemical reaction you are seeing is called: COMBUSTION.

When you hear the word combustion what comes to mind? You may remember from previous chemistry courses, that combustion reactions involve a compound reacting with oxygen to produce carbon dioxide (CO2), water (H20) and thermal energy. When you see a fire burning you are witnessing combustion, and the heat you feel from it is the thermal energy being generated in the combustion reaction. The balanced reaction equation below represents the complete combustion of butane, which you should know, has the molecular formula C4H10, looks like:
2 C4H10 + 13 O2 → 8 CO2 + 10 H20 + thermal energy
It is this thermal energy that provides the power to move cars or heat up food on a stovetop, both of which contribute to the quality of life that we as humans have the opportunity to experience. However, the CO2 that is produced during the combustion of hydrocarbons is released into the air, where it floats up into the atmosphere and contributes to what is known as the greenhouse effect. The greenhouse effect helps to trap heat in our atmosphere and leads to the warming of our planet. This warming of our planet over time can have disasterous effects, impacting climate and affecting humans, plants, animals and the environment in a global manner. It is important for us as scientists and citizens of the world to consider the potential negative outcomes that are generated by the positive experiences of driving, flying or any other endeavours that rely on the combustion of hydrocarbons.
D. Substitution Reactions
In a substitution reaction, 2 molecules react with each other and atoms from each are exchanged and substituted for each other, creating 2 products that are unique from the reactants. A general formula for a substitution reaction can be represented as:
AB + CD → AD + BC
One such substitution reaction that alkanes undergo produces a compound that is known as an alkyl halide, which is an alkane that has one or more halogen atoms bound to it in place of a hydrogen atom. Recall that halogens are the elements that are found in the second last column of the periodic table (F, Cl, Br, etc). In substitution reactions with alkanes, halogen molecules are in the form of dI atomic molecules, for example Cl2, Br2, and the products are alkyl halides and hydrochloric acid. For example, in the reaction below, methane and chlorine react to produce chloromethane
CH4 + Cl2 → CH3Cl + HCl
Naming Alkyl Halides
When naming alkyl halides, we take the root name of the halogen and add the letter –o, for example bromo or chloro, we put this in front of the name of the alkyl group. In the reaction above the products would therefore be bromomethane and hydrochloric acid. For alkanes with more than one carbon, we follow the same rules we already learned and indicate the carbon number that the halide is bound to, if there are more than one halogen atoms bound to the alkyl halide, we use the prefix di, tri, or tetra as well as the numbers of the carbon they are bound to. For example the molecule CH2Br-CHBr-CH3 would be named 1,2-dibromopropane.
E. Polymers
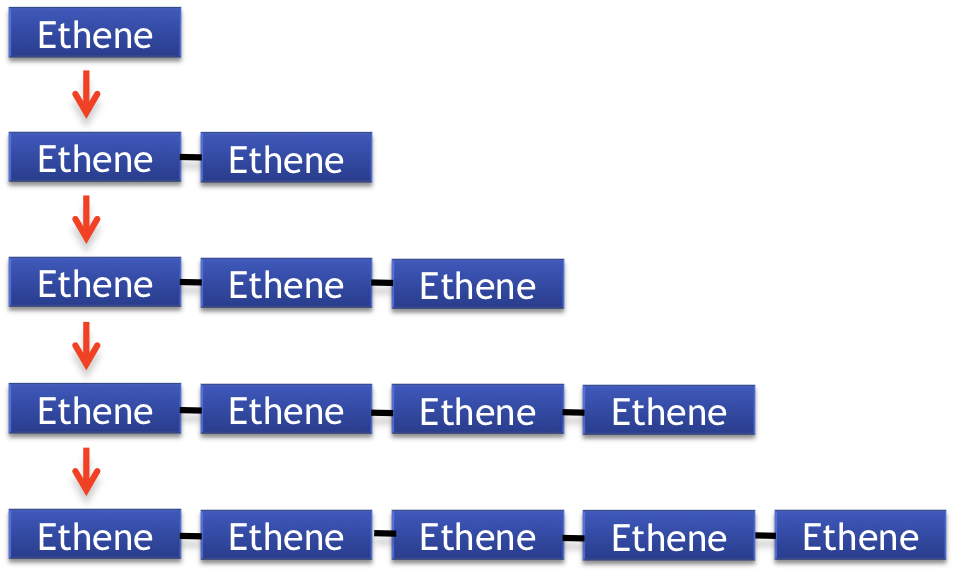
Polymers are large molecules that are made up of individual units called monomers. These monomers can be joined together to form mononers in both addition and condensation reactions. Polymers are found in many everyday products from nylon to plastic. For example polyethene (which is commonly referred to as polyethylene) is a polymer that is the most common type of plastic. It is formed by the addition reaction of multiple monomers of ethene to form a very large polymer molecule.
F. Summary
- Combustion reactions with alkanes produces CO2, H2O and thermal energy
- Alkanes can undergo substitution reactions to produce alkyl halides
- Alkenes and Alkynes are more reactive due to their multiple bonds
- Hydrogenation: addition reaction that adds hydrogen atoms to unsaturated hydrocarbons, produces alkenes and alkanes
- Halogenation: addition of halogen atoms to hydrocarbons
- Hydration: reactions with water molecules whereby a hydrogen atom and hydroxyl (-OH) group are added to reactant molecule
- Hydration reactions produce alcohols
- Hydration and halogenation reactions follow Markovnikov's rule, whereby the hydrogen atom is added to the carbon that has the most hydrogen atoms bound to it prior to the reaction
- Polymers are large molecules made up of individual units called monomers, these polymers can be formed by addition or condensation reactions