SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-4C: Chemical Reactions: Alcohols & Ethers
Description
In this activity you will explore chemical reaction that involve alcohols and ethers including hydration, dehydration and condensation reactions.
Page content
A. Chemical Reactions: Alcohols & Esters
Reactions with alcohols are one of the earliest ever forms of chemistry. People have been producing and drinking alcohol for centuries, even before they understood the chemistry behind it. Below we explore the reactions involved to produce alcohols followed by an explanation of the reactions used to produce ethers: condensation reactions.
B. Hydration Reactions & Alcohols
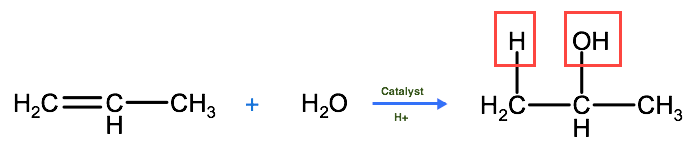
We saw in the previous learning activity, that alcohols are the product of hydration reactions involving alkenes. These reactions follow Markovnikov's rule, whereby the hydrogen atom is added to the carbon with the most hydrogen atoms bound to it before the reaction began. For example, 2-propanol (and not 1-propanol) is formed by the addition reaction of 1-propene with a water molecule because of Markovnikov's rule.
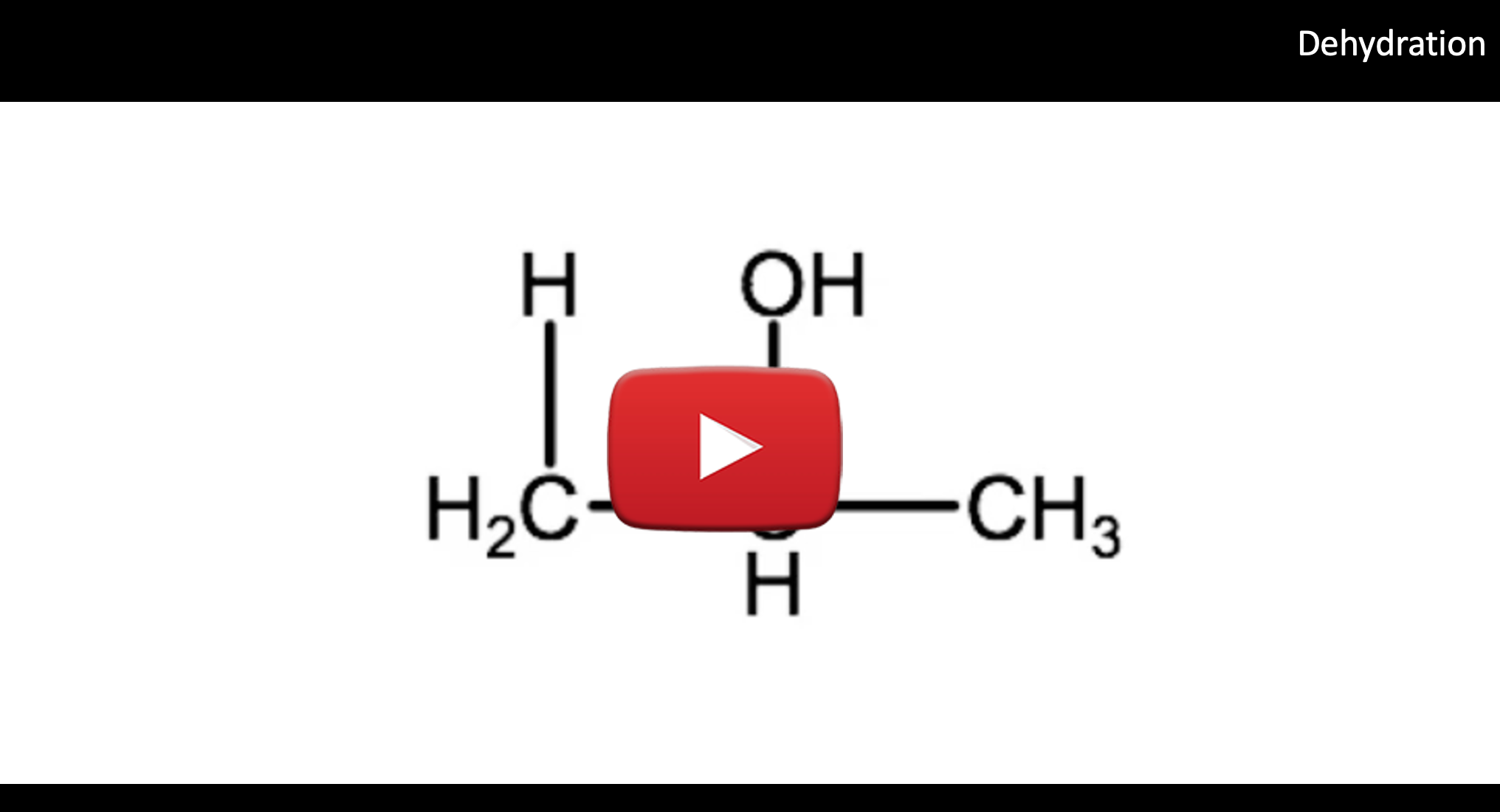
C. Elimination Reactions: Dehydration
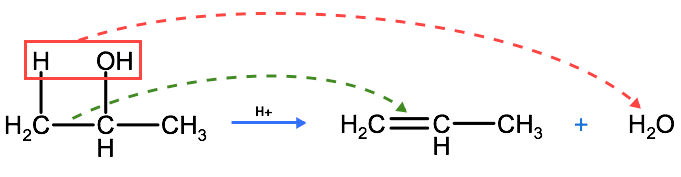
An elimination reaction is one in which two substituents are removed from a molecule, and in organic chemistry this is typically accompanied by the formation of a double bond. One such elimination reaction that occurs with alcohols is called dehyration which is the reverse of a hydration reaction. In a dehydration reaction a hydrogen and hydroxyl group are removed from a reactant molecule, leading to the formation of a double bond. A water molecule is also formed as one of the products. Dehydration reactions require the presence of a catalyst to proceed, in this case an acid. The removal of both H and -OH creates a double bond between the carbons that were initially bound to the H and the -OH. Another way to think of a dehydration reaction is that a water molecule is removed from a reactant molecule, or the reactant molecule is de-hydrated. Dehydration reactions require acid to act as a catalyst to push these reactions forward. In the dehydration reaction below, we have the opposite of the reaction we just saw above. In our dehydration reaction we have 2-propanol undergoing a reaction to form 1-propene and water. Note the appearance of the double bond between carbon 1 and carbon 2.
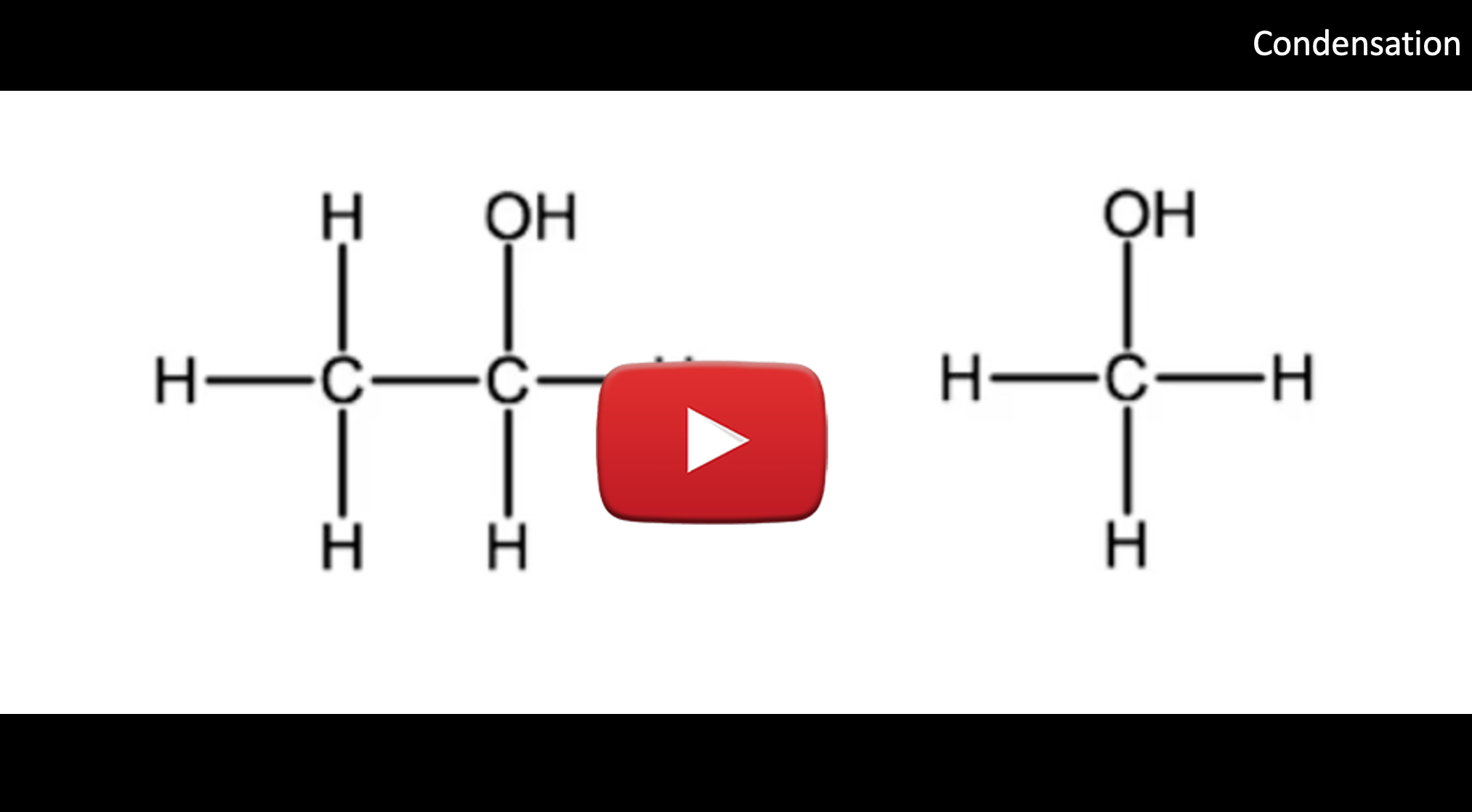
D. Condensation Reactions: Producing Ethers
A condensation reaction is one in which two molecules react with each other to produce one large molecule and one water molecule. These are similar to a dehydration reaction, in which one of the products is a water molecule, but remember, in a dehydration reaction, there was only one reactant, whereas in a condensation reaction we have two reactants. We previously learned that ethers are similar to alcohols in that they contain oxygen atoms that are single-bonded to carbon atoms, the difference being that in ethers, the oxygen atom is bound between 2 carbon atoms. To produce ethers, we need two alcohol molecules to react in a condensation reaction, producing an ether and a water molecule. For example, below we see the condensation reaction between ethanol and methanol to produce methoxyethane and water.
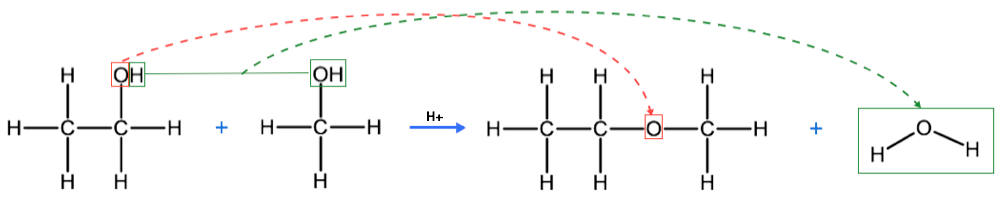
Notice that a condensation reaction requires the presence of two hydroxyl groups, one to contribute an oxygen atom to the ether and a hydrogen atom to the water molecule, and the other to contribute the full hydroxy group to the water molecule.
E. Summary
- Alcohols are produced from hydration reactions between an alkene and water
- Hydration reactions follow Markovnikov's rule
- Elimination reactions are those in which 2 substituent molecules are removed from a reactant molecule producing a double bond on the product molecule
- Dehydration reactions are an example of an elimination reaction
- In dehydration reactions, alcohols lose a hydrogen atom and a hydroxyl group, forming an alkene and water as the products
- Ethers are produced through condensation reactions where two alcohols react to produce an ether and a water molecule