SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
2-4D: Chemical Reactions: Aldehydes, Ketones, Carboxylic Acids & Esters
Description
In this activity you will explore chemical reaction that lead to the synthesis of aldehydes, ketones, carboxylic acids and esters.
Page content
A. Controlled Oxidation: Synthesizing Aldehydes
Aldehydes and Ketones are formed through a chemical reaction known as controlled oxidation. Controlled oxidation is very similar to the combustion reaction that we saw earlier that alkanes undergo, where an alkane and water react to produce O2, CO2 and thermal energy. In a controlled oxidation reaction of an alcohol, there is a limited quantity of oxygen present on the reactant side as a result, the products of the reaction are aldehydes or ketones depending on the nature of the alcohol that is oxidized.
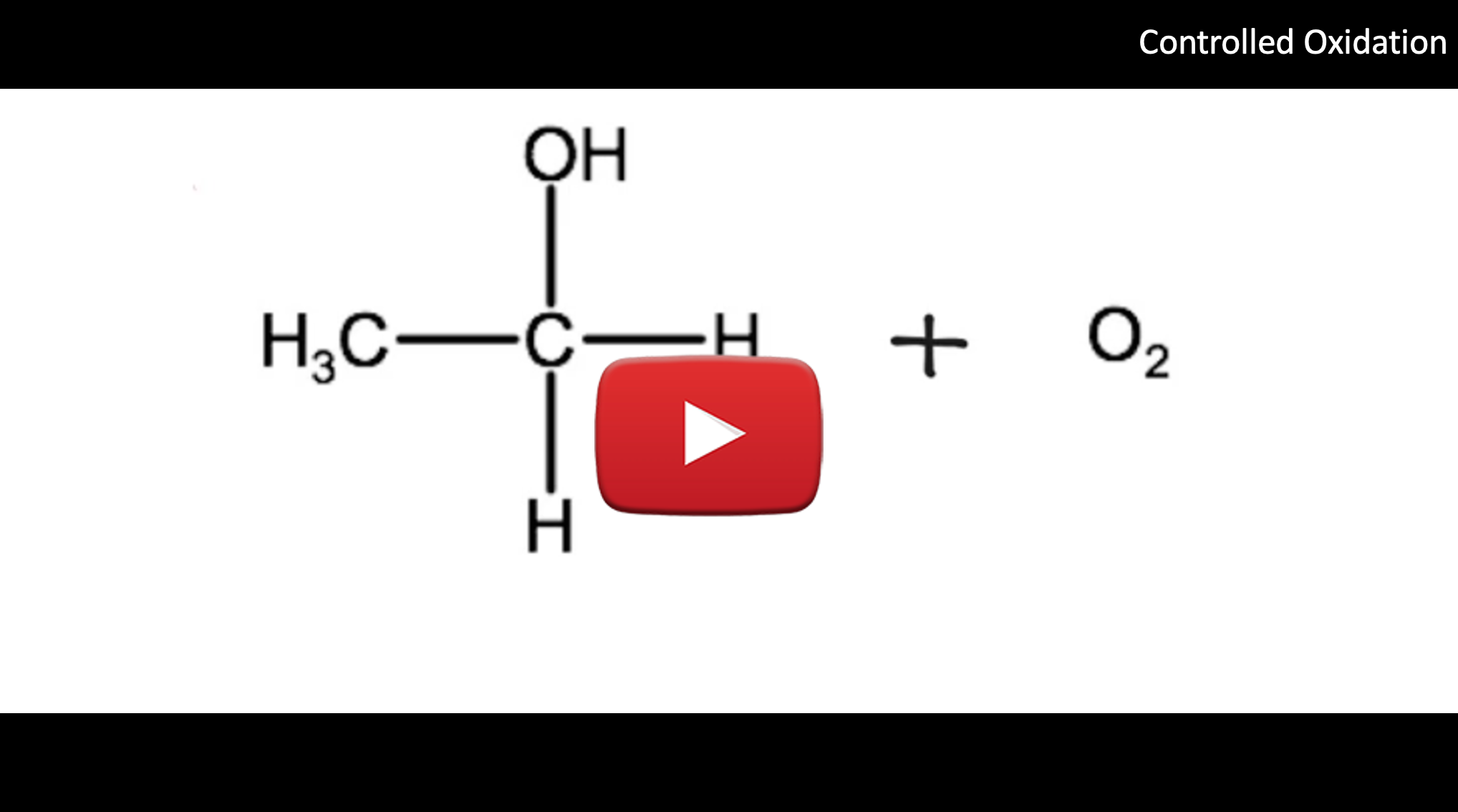
In a controlled oxidation reaction, the alcohol loses two hydrogen atoms which combine with an oxygen atom to form water, while the alcohol is transformed into an aldehyde or ketone. Below we see the controlled oxidation of ethanol using oxygen gas producing ethanal and water. Note that the equation below is not balanced, it is there to show you diagramatically which atoms of the ethanol molecule are involved.
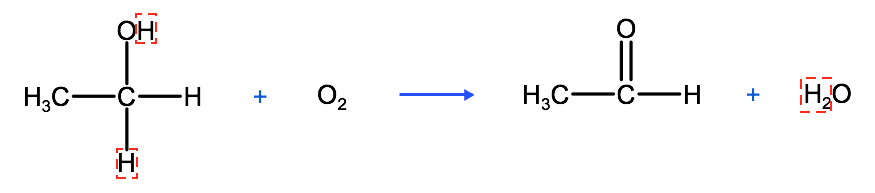
In an oxidation reaction, the oxygen can come from pure oxygen in the air or from compounds that are known as oxidizing agents, which are molecules that have oxygen within them. As we see in the example above, in a controlled oxidation, the carbon with the hydroxyl group loses 2 hydrogen atoms and gains a double bond. Depending on the placement of the hydroxyl group, an aldehyde or a ketone will form. Alcohols can be classified based on the carbon atom that the hydroxyl group is bound to within the alcohol. For example, if the hydroxyl group is bound to a terminal carbon (as we see in the example above), which by nature is only bound to one other carbon, it is classified as primary. If the hydroxyl group is bound to a carbon that is bound to two other carbon atoms, it is classified as a secondary alcohol. If the hydroxyl group is bound to a carbon that is bound to three other carbons, it is classified as a tertiary alcohol.
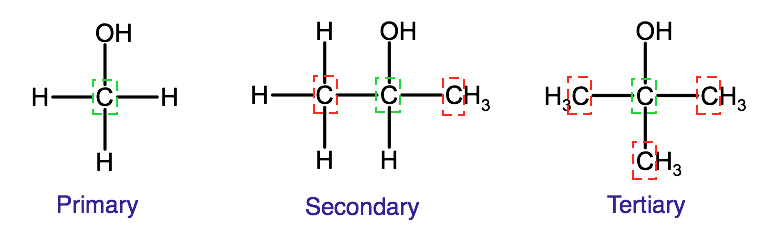
To produce an aldehyde, a controlled oxidation must occur with a primary alcohol, to produce a ketone, the controlled oxidation must occur with a secondary alcohol. Tertiary alcohols do not undergo controlled oxidation as there are no extra hydrogen atoms on the carbon that the hydroxyl group is bound to.
The following video outlines the controlled oxidation reactions that produce aldehydes and ketones from primary and secondary alcohols respectively:
B. Controlled Oxidation: Synthesizing Ketones
As we said above, to produce a ketone requires the controlled oxidation of a secondary alcohol (an alcohol where the hydroxyl group is bound to a carbon that is bound to two other carbons). In the example below we have the controlled oxidation of 3-pentanol producing the ketone: 3-pentanone. Again the equation below is unbalanced, but is used to show the mechanism of the controlled oxidation of a secondary alcohol
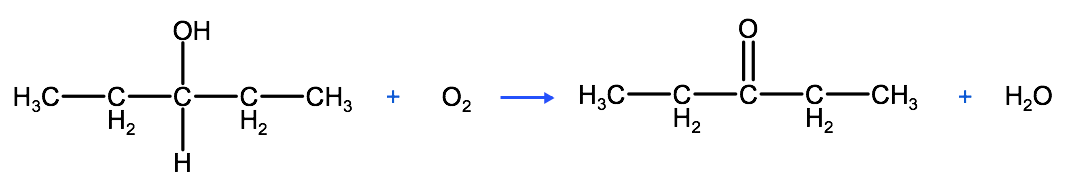
To pull this all together, to synthesize an aldehyde requires the controlled oxidation of a primary alcohol, whereas to synthesize a ketone requires the controlled oxidation of a secondary alcohol.
Check your understanding
C. Esterification
Esterification is the chemical process whereby esters are formed. Esters, are organic compounds formed from the reaction between a carboxylic acid and an alcohol in the presence of a strong acid catalyst that speeds up the reaction. Sulfuric acid
is the strong acid in the diagram below. Esters are formed through a condensation reaction that is classified as esterification. Recall that in a condensation reaction, one of the products is a water molecule. An esterification reaction involves
a condensation reaction between a carboxylic acid and an alcohol, producing an ester and water. Below we see an esterification reaction between ethanoic acid and 3-methylbutanol which produces the ester: 3-methylbutyl ethanoate
When naming esters, use the following steps:
1. Determine which alkyl group came from the carboxylic acid. The alkyl group that came from the carboxylic acid will be the one with a carbon double bonded to an oxygen.
2. Replace the -oic acid ending of the name of the parent acid with -oate.
3. The second akyl group which is attached to the oxygen comes from the alcohol. Name this alkyl group as usual and put it at the front of the name.
4. Put the two names together. Note that esters are named as two words.
The parent acid is butanoic acid and the alkyl group comes from ethanol. So this ester would be named ethyl butanoate.

D. Hydrolysis of Esters
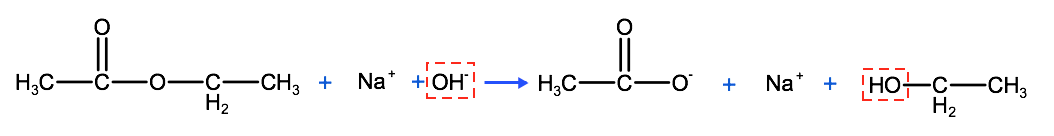
Esters can be broken down into their constituent carboxylic acid and alcohol precursors by a chemical reaction known as hydrolysis. In a hydrolysis reaction, a covalent bond is broken by the constituents of a water molecule, H and O. The constituents of the water molecule (H and O) in the hydrolysis of an ester come from the base sodium hydroxide (NaOH). The hydrolysis of an ester is essentially the reverse of esterification. Instead of a carboxylic acid and an alcohol reacting to form an ester, an ester reacts to form a carboxylic acid and an alcohol.
Below we have an example of the hydrolysis of ethyl ethanoate into ethanol and ethanoic acid. The hydroxyl group (-OH) from NaOH acts to lyse (cut) the single bond between the oxygen and the carbon of the ester and insert itself to create the alcohol.
E. Summary
- Alcohols can be classified as primary, secondary or tertiary based on the number of carbons that the carbon which is bound to the hydroxyl group is bound to
- Aldehydes can be synthesized through the controlled oxidation reaction of a primary alcohol
- Ketones can be synthesized through the controlled oxidation reaction of a secondary alcohol
- Esters are formed through a chemical process known as esterification
- In an esterification reaction, a carboxylic acid reacts with an alcohol producing an ester and water
- Esters can be broken down through a hydrolysis reaction that produces a carboxylic acid and an alcohol