SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
3-1A: Energy & Enthalpy
Description
In this interactive lesson you will be introduced to the concepts of energy and enthalpy as they relate to chemical reactions.
Page content
The following lesson will introduce you to the concepts of energy and enthalpy in chemical processes.
We are going to begin this unit by introducing some of the important terms and concepts that we will be using in the next few lessons. The focus of this unit is on energy changes and how they affect chemical reactions. We will be looking at different types of reactions and how they affect and are affected by their surroundings. By the end of this lesson, you should be familiar with terms and concepts including: system, surroundings, enthalpy, exothermic, and endothermic.
A. Energy
Potential & Kinetic Energy
All substances have a certain amount of energy associated with them. Either in the form of KINETIC ENERGY, which is the energy of motion, or POTENTIAL ENERGY, which the is energy that is stored within an object. Think about an unlit stick of dynamite, it is storing a tremendous amount of potential energy. This potential energy is stored within the chemical bonds of the atoms that make up the dynamite. When the stick is lit, this potential energy is converted into kinetic energy in the form of an explosion. HEAT that is given off from the explosion is a type of kinetic energy that is present in all substances.

Law of Conservation of Energy
In chemical processes, which include chemical reactions and changes of state, energy is released into the surroundings or absorbed from the surroundings. Energy can never be created or destroyed, only transformed from one form into another. This is known as the LAW OF CONSERVATION OF ENERGY: Energy cannot be created or destroyed. The total amount of energy in an isolated system remains constant.
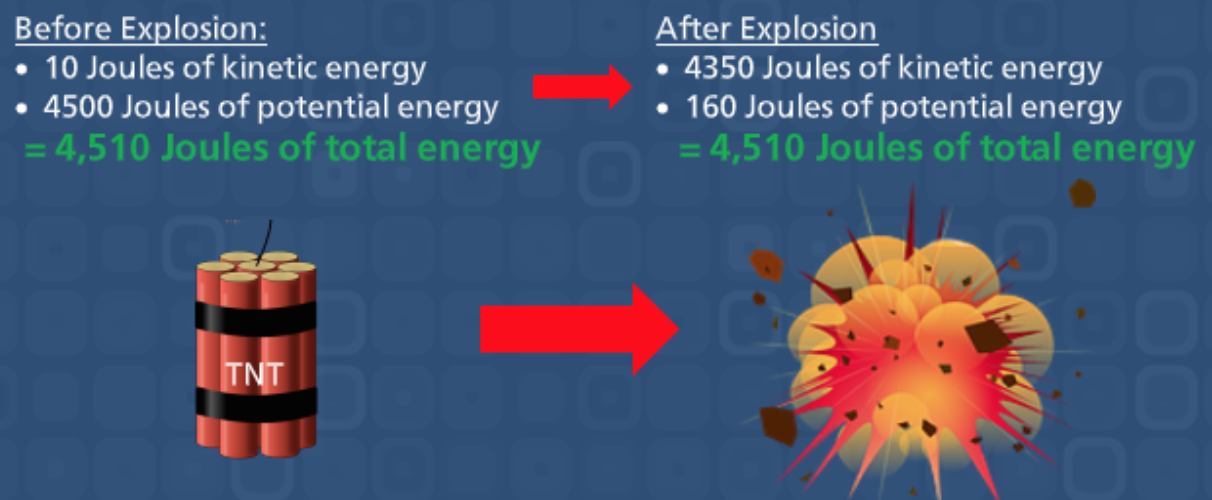
The total amount of energy remains the same before and after the explosion, only the type of energy has changed, from potential to kinetic.
System & Surroundings
Chemists are very interested in energy changes that occur during chemical processes, this includes energy that is released or absorbed during a reaction or change of state. To measure these changes, chemists need to define which objects are absorbing and which are releasing energy. The SYSTEM is defined as the sample you are examining, and the SURROUNDINGS are defined as everything else around the system. For example, if you are studying the energy changes that take place in a chemical reaction in water, the chemicals involved in the reaction would be the system and the water would be the surroundings. The products and reactants (system) would either absorb or release energy to or from the water (surroundings).

All substances have a certain amount of energy inside of them. Chemists are very interested in observing the changes of energy in a system during a chemical reaction or change of state. A system is the sample or reaction that a scientist is observing, while the surroundings are everything else that exists around the sample.
B. Heat & Enthalpy
What do you think of when you hear the word heat? Warmth? Temperature? Fire? Heat is a form of kinetic energy that is related to the movement of the molecules that make up a substance. When a substance is heated up, its molecules move faster and faster and when it is cooled down they move more slowly. If enough heat is added to a substance, its molecules can move fast enough to pull apart from each other and this is what causes a change of state from a solid to a liquid for example. Heat can be transferred between substances, and it always moves from a hotter to a cooler object. When heat is added to a substance, its temperature increases and when it is removed its temperature decreases.
The heat content of a substance is called its ENTHALPY, which is represented by the symbol: H. Chemical reactions and changes of state involve transfer of heat from the sample to the surroundings or vice-versa. For example, if you leave a cup of hot water on a table, the water transfers heat to the surrounding air as well as the cup and the table. This transfer of heat causes the water to cool off and the cup, table and air to heat up. This transfer of heat causes a change in enthalpy which is represented by the symbol: ΔH (this is pronounced: "delta-H). In chemical processes we are very interested in measuring this change in enthalpy (ΔH).
If you leave a beaker of hot water (the system) on a desktop, it will begin to cool. In other words, the water loses heat to the surroundings. This loss of heat from the system to the surroundings is called enthalpy change ΔH
C. Endothermic & Exothermic Reactions
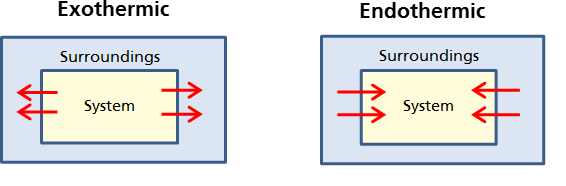
Chemical reactions can be defined based on whether they absorb or release heat, or in other words, their change in enthalpy. When a system releases heat to its surroundings during a reaction or change of state, the process is described as EXOTHERMIC. The prefix "exo-" means out of, so in an exothermic process, heat moves out of the system.
When a system absorbs heat from its surroundings, the process is defined as endothermic. Endo- means inside, so in an endothermic reaction heat moves inside the system. A good memory tool to remember the difference
between endo- and exo-thermic is to pronounce exothermic as ex-ho-thermic, where the -ho- stands for heat-out.
When distinguishing between exothermic and endothermic, remember we are always considering whether the system is absorbing (endothermic) or releasing (exothermic) heat
D. Endothermic Reactions & Enthalpy
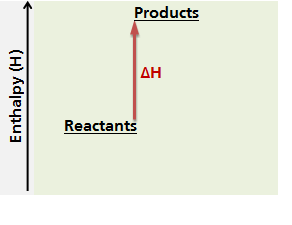
In an endothermic reaction, the system gains (or absorbs) heat from its surroundings during the course of the reaction, therefore its enthalpy changes. We can represent the change in enthalpy for a reaction using an energy diagram. The diagram below represents the change in enthalpy (ΔH) of an endothermic chemical reaction. In an endothermic reaction, the products, which are formed after the reaction, have a higher enthalpy value than the reactants because the system has absorbed heat from the surroundings. Therefore, in an endothermic reaction, the change in enthalpy, ΔH, is positive.
E. Exothermic Reactions & Enthalpy
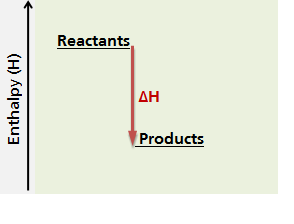
In an exothermic reaction, the system releases heat to its surroundings during the course of the reaction, therefore its enthalpy changes. The diagram below represents the change in enthalpy (ΔH) of an exothernmic chemical reaction. In an exothermic reaction, the products, which are formed after the reaction, have a lower enthalpy value than the reactants because the system has released heat to its surroundings. Therefore, in an exothermic reaction, the change in enthalpy, ΔH, is negative.
F. Summary
- All substances contain a certain amount of energy within them in the form of kinetic and potential energy
- The law of conservation of energy states that energy cannot be created or destroyed
- The system is the object or sample that one is investigating
- The surroundings is everything else around the system
- Chemists are interested in the transfer of energy that takes place between system and surroundings
- Heat is a form of energy that can be transferred, causing temperature to rise or fall.
- The heat content of a substance is known as its enthalpy (H)
- The change in heat that occurs in a chemical process is known as its change in enthalpy (ΔH)
- Endothermic reaction: when the system absorbs heat from its surroundings, its temperature increases and ΔH is positive
- Exothermic reaction: when the system releases heat to its surroundings, its temperature decreases and ΔH is negative