SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
3-1B: Change in Enthalpy: Reactions & Changes of State
Description
In this lesson you will navigate your way through an interactive activity as you explore how changes in enthalpy relate to chemical reactions and changes of state.
Page content
To quickly recap what we learned in the last lesson, chemists are very interested in observing whether a given system absorbs or releases heat during a chemical reaction or change of state. A system is the sample or reaction that a scientist is observing, while the surroundings are everything else that exists around the sample.
The heat content of a substance is also referred to as its enthalpy (ΔH). Chemists define chemical processes as either endothermic or exothermic depending on its change in enthalpy (ΔH). Endothermic reactions absorb heat from their surroundings and have an increase in enthalpy; while exothermic reactions release heat to their surroundings and have a decrease in enthalpy.
A. Change in Enthalpy (ΔH) in Changes of State
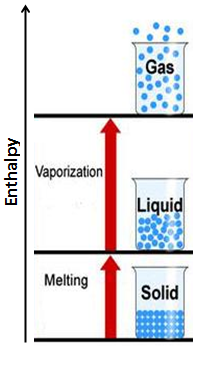
Melting & Vapourization: Solid to Liquid & Liquid to Gas
During a change of state, the molecules of a substance move closer together or farther apart from each other depending on the change. When a substance goes from a solid to a liquid (melting) or a liquid to a gas (vapourization), intermolecular bonds are broken and the molecules move farther apart from each other. The breaking of intermolecular bonds requires the absorption of heat from the surroundings, to overcome intermolecular forces. To remember this imagine boiling a pot of water, to do this you need to heat it up on a stove-top or over a fire. In other words you need to add heat to get the liquid to boil and vapourize into a gas. Therefore, in a change of state where intermolecular bonds are broken, the enthalpy of the system increases, and the ΔH is positive.
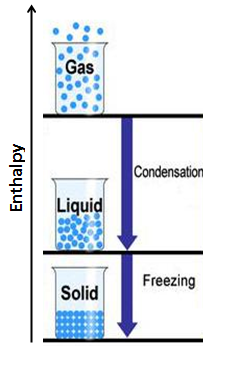
Condensation & Freezing: Gas to Liquid & Liquid to Solid
When a substance changes from a gas to a liquid (condensation) or a liquid to a solid (freezing), intermolecular bonds are formed and the molecules move closer to each other. When bonds form, whether they are intermolecular or intramolecular, energy, in the form of heat is released from the system into the surroundings. To remember this, think about making ice cubes in a freezer, to do this you would put liquid water into a cold environment, this removes heat from the water and allows it to freeze. Therefore in a change of state where intermolecular bonds form, the enthalpy of the system decreases and the ΔH is negative.
B. Changes in Enthalpy in Chemical Reactions
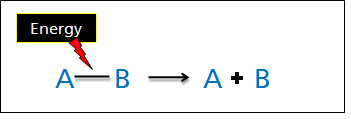
Breaking Bonds
In a chemical reaction, intramolecular bonds between atoms are broken and/or formed. Similar to changes of state, for chemical bonds to be broken, the system must absorb heat (energy) to break the bonds and the process is endothermic (ΔH is positive).
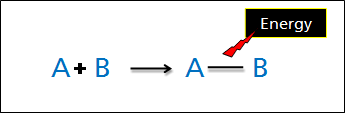
Forming Bonds
Conversely, when chemical bonds are formed between atoms, the system releases heat to the surroundings thus the process is exothermic (ΔH is negative)
C. Changes in Enthalpy in Nuclear Reactions
Nuclear reactions are processes that occur in the nucleus of atoms. There are 2 types of nuclear reactions: Fission and Fusion. Both of these processes are exothermic and release tremendous amounts of energy.
Nuclear Fission
In a nuclear fission reaction, a heavy nucleus splits into 2 lighter nuclei, when the nucelus splits a tremendous amount of energy is released from the system. We are able to harness the energy released from a nuclear fission reaction to produce electricity in nuclear power plants as well as for destructive purposes as when used in an atomic bomb.

Nuclear Fusion
Nuclear fusion is a process in which 2 nuclei combine (or fuse together) to form a single nucleus. When the nuclei combine, a tremendous amount of energy is released. Nuclear fusion is the exothermic process that occurs on the sun which produces the heat that we can feel all the way here on Earth.

D. Summary
- Changes of state, chemical reactions and nuclear reactions all have associated changes in enthalpy ΔH
- In changes of state, molecules move closer together or further apart
- For intermolecular bonds to be broken, heat must be added to the system, therefore the ΔH is positive
- When intermolecular bonds form, heat is released from the system, therefore the ΔH is negative
- Melting and vapourization require heat to be added to the system and are endothermic processes
- Condensation and freezing require heat to be removed from the system are exothermic processes
- In a chemical reaction, breaking bonds requires heat to be added to the system, therefore breaking bonds is endothermic
- In a chemical reaction, forming bonds releases energy from the system, therefore forming bonds is exothermic
- Nuclear reactions, which include fission and fusion are exothermic reactions that produce tremendous amounts of energy