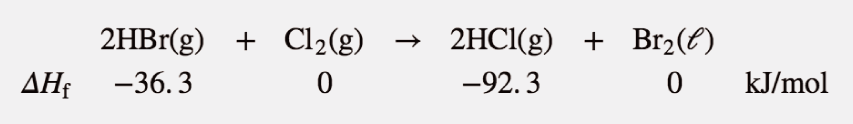SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
3-3A: Hess's Law
Description
In this activity you will use graphs, tables and problem solving skills to learn about Hess's Law and how to apply it to solve problems relating to reaction mechanisms.
Page content
A. Hess's Law
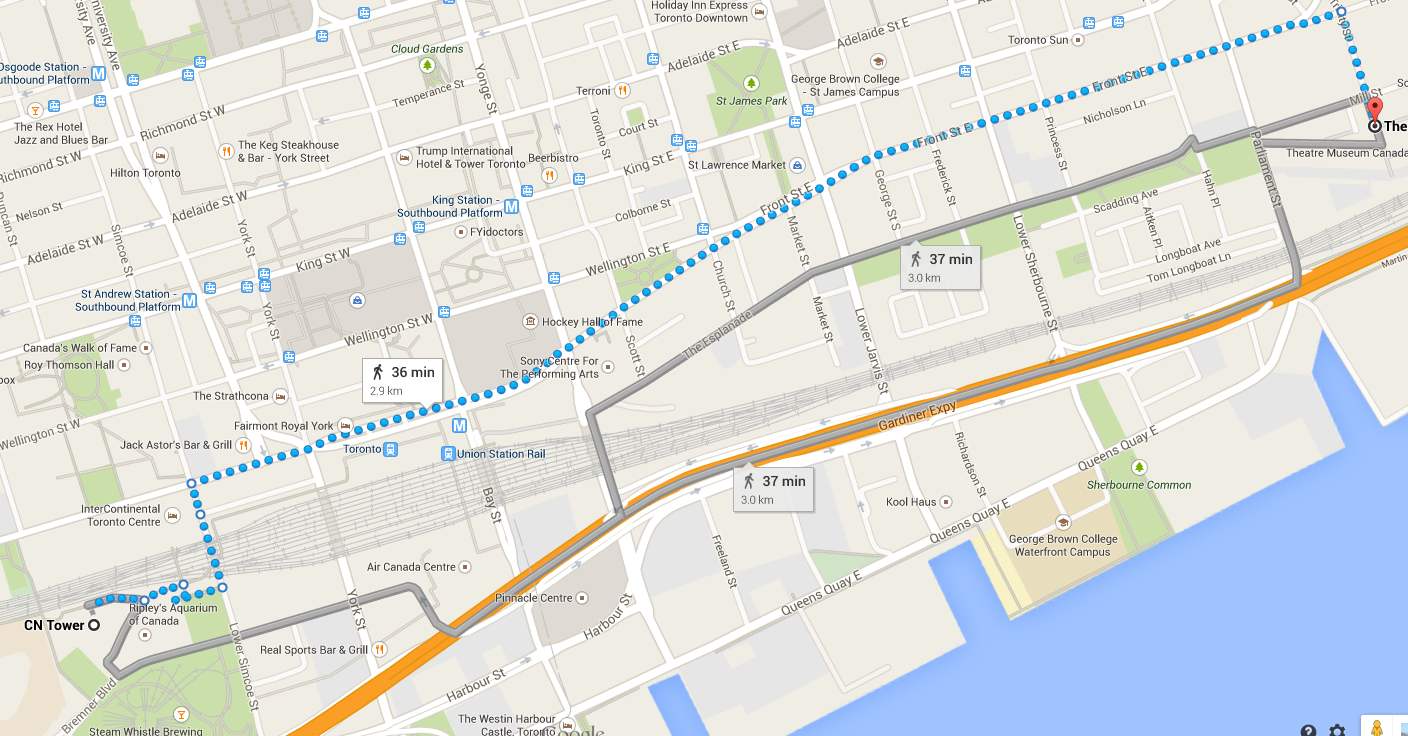
A chemical reaction involves one or more reactants undergoing a reaction to form one or more products with a resulitng change in enthalpy. By just looking at a chemical equation, it may seem as if a reaction is a one step process in which the reactants are converted to products but in reality, a chemical reaction may take one or more steps to ultimately form the final products. One way to think about this is to imagine you are in downtown Toronto and want to walk from the CN Tower to a restaurant in the Distillery District. There is more than one route you may take to get to the restaurant, some routes may be longer but have more interesting things to look at, while another route may be shorter but more boring to traverse. In either case, the starting point and the destination are the same, only the route to get there changes.
 |
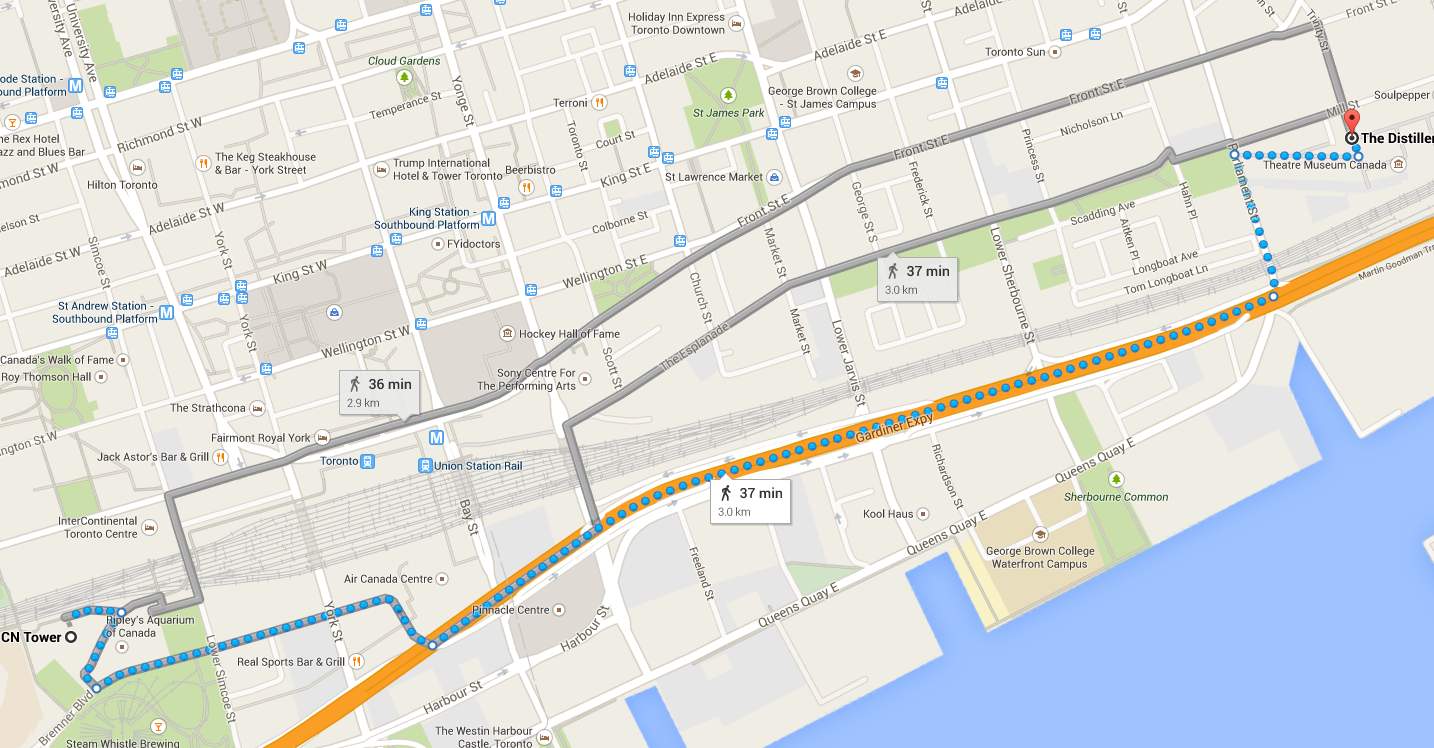 |
Interestingly, a chemical reaction follows the same logic, imagine the reactants are the starting point and the products are the destination. Although the reactants and the product are the same, the route to get from one to the other can be different. For example, the formation of nitrogen dioxide gas from nitrogen and oxygen gas can occur in one of two ways, a two-step process or a one-step process:
One-step process:
N2 (g) + 2O2 (g) → 2NO2 (g) ΔH = + 68 kJ/mol
Two-step process
1. N2 (g) + O2 (g) → 2NO (g) ΔH1 = +180 kJ/mol
2. 2NO (g) + O2 (g) → 2NO2 (g) ΔH2 = -112 kJ/mol
Each step in this 2-step process is called an elementary step.
If you add these two reaction equations, of the elementary steps, together, you get the overall reaction equation for the formation of nitrogen dioxide, which has the same ΔH value as the one step process.
N2 (g) + O2 (g) → 2NO (g) ΔH = +180 kJ/mol
2NO (g) + O2 (g) → 2NO2 (g) ΔH = -112 kJ/mol
N2 (g) + 2O2 (g) → 2NO2 (g) ΔH = +68 kJ/mol
When adding reaction equations together, we cancel out any molecules that appear on both sides of the reaction arrow, as long as they have the same coefficient. These are only reaction intermediates that will further react to produce the final chemical equation. For example, when adding the equations above, we cancel out the reaction intermediate 2NO that appears on both sides of the arrow. We also add any of the same molecules that appear on the same side of the arrow. Therefore the O2 (g) that appears in both equations are added together to become 2O2 (g) in the final equation.
Enthalpy Diagram
We can represent both possible reaction routes using an enthalpy diagram. Notice the total change in enthalpy for both.

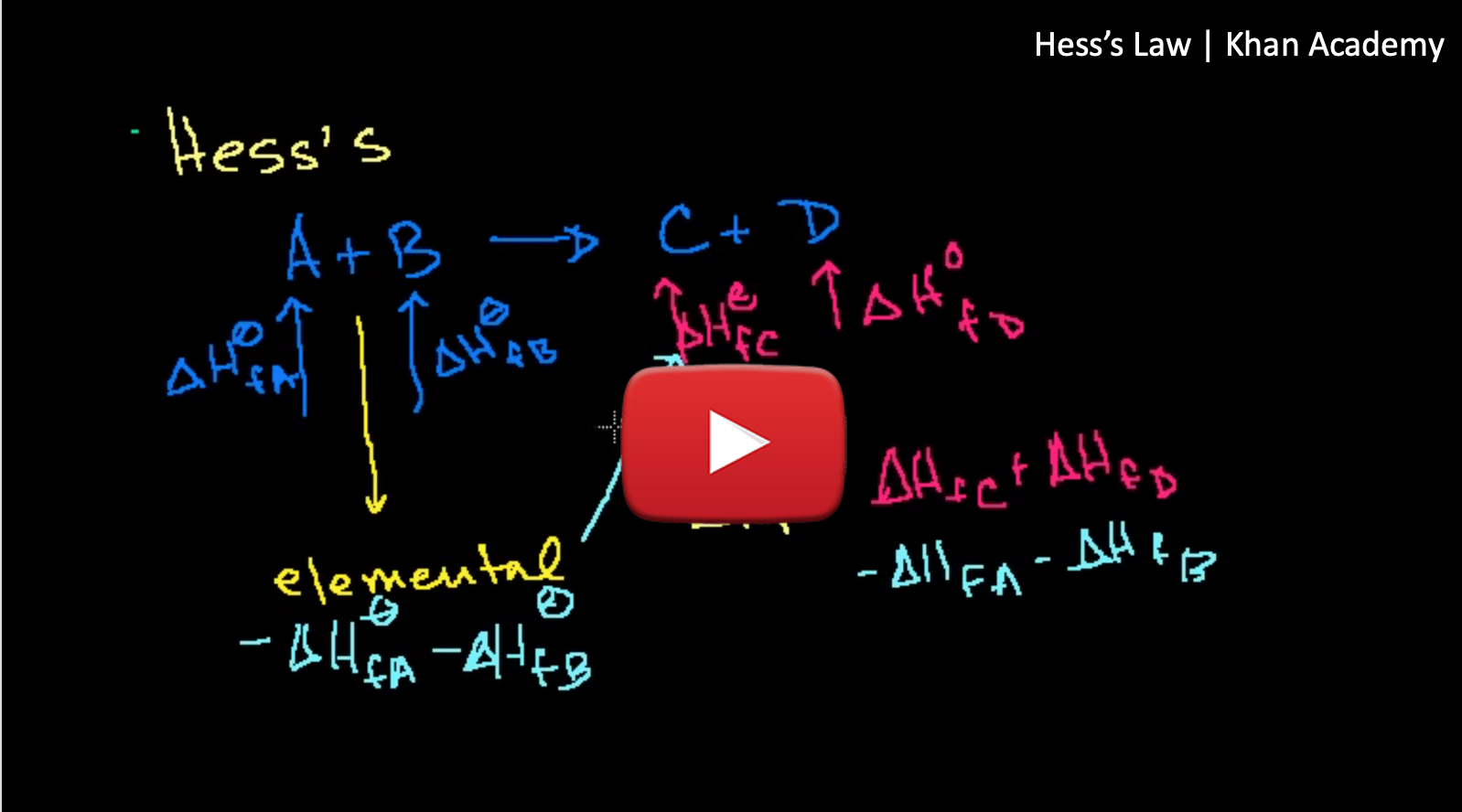
The total ΔH for the two-step process combines the ΔH for each step and is equal to the ΔH for the one-step process. The interesting thing about all chemical reactions is that no matter the path taken to go from products to reactants, the enthalpy change (ΔH) is the same. This concept is known as Hess's Law.
Hess's Law: The change in enthalpy for a chemical reaction going from reactants to products is the the same no matter the route taken to get there.
We can use Hess's law to calculate changes in enthalpy for reactions that can not be measured using a calorimeter. To do this we add together reactions with known ΔH values, these reactions are known as Standard Enthalpy of Formation (ΔH°f).
B. Standard Enthalpy of Formation (ΔH°f)
Standard Enthalpy of Formation (ΔH°f) is a reaction that produces one mole of a substance from its elements in their standard states. For example the ΔH°f for water is as follows is the reaction that produces 1 mole of water from its elements, hydrogen and oxygen:
![]()
The following link contains a table listing common Standard Enthalpies of Formation.
C. Calculating ΔH using Hess's Law
Lets use Hess's Law to calculate the ΔH for the following reaction:
![]()
Step 1. Using the standard enthalpies of formation table, find a reaction that has C(s) in it.

Therefore our first reaction is:
![]()
So, we've got the proper reactant, C(s), but we also have some compounds that are not in the reaction equation we are trying to solve for.
Step 2. Find a reaction that contains CO as a reactant to eliminate the unwanted CO product in the first reaction. When doing this, you should look for equations that contain compounds that you need for your final equation. In this case, we will look for a reaction that contains CO as a product and CO2 as a reactant.
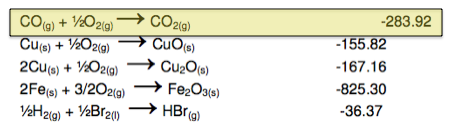
Therefore our second reaction is:
![]()
Step 3. We are now getting closer, but we now have some compounds that we need to eliminate. If we add these two reactions we end up with the equation:
C(s) + H2O(g) + CO(g) + 1/2O2 → CO(g) + H2(g) + CO2(g)
To produce our final equation: on the reactants side, we need to get rid of the H2O and we need to add another 1/2O2. On the products side we need to get rid of the H2. Because we have CO as both a reactant and a product these will cancel each other out. So we now will look for an equation that has 1/2O2 and H2 as reactants and H2O as a product. This will allow us to cancel out the necessary compounds and add the necessary 1/2O2.
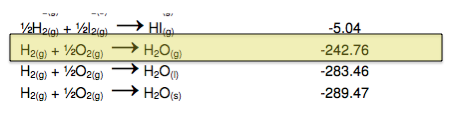
Therefore, our third reaction is:
![]()
Step 4. Add the equations together, cancel out any compounds that are on opposite sides of the equation, and add the ΔH values.
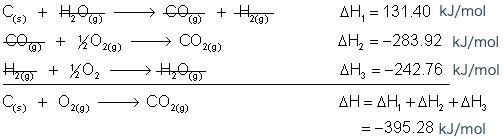
Now if you look on the Standard Enthalpies of Formation table, you will see that for our initial equation:
![]()
ΔH = -395.28 kJ/mol, which is the same value we got by taking multiple steps to get there.
D. Rules for Using Hess's Law
When selecting equations to use when solving problems using Hess's Law, we can manipulate the reaction equations to give us some of the variables we want. For example we can take a reaction from the table and simply reverse it to give us a product or reactant that we need. We can also change the coefficient in front of a compound, by simple multiplying the entire equation by a given coefficient. However, whenever solving using Hess's Law, we need to observe the following rules:
1. All equations must be balanced.
2. If a reaction is being reversed, the sign of ΔH must also be reversed. For example:
N2 (g) + O2 (g) → 2NO (g) ΔH = +180 kJ/mol
If we were to reverse this equation, we also reverse the ΔH value, therefore it becomes:
2NO (g) → N2 (g) + O2 (g) ΔH = -180 kJ/mol
3. The magnitude of ΔH is proportional to the number of moles of reactants and products in an equation. Therefore if you multiply an equation by a certain factor, you must also multiply the ΔH as well. For example, suppose you want to double the following reaction to be able to cancel one of the compounds in your equation:
N2 (g) + O2 (g) → 2NO (g) ΔH = +180 kJ/mol
You must also multiply the ΔH value by 2:
2N2 (g) + 2O2 (g) → 4NO(g) ΔH = +360 kJ/mol
Check your understanding:
Find the ΔH value for the combustion of CH4. Remember in a combustion reaction, a compound reacts with oxygen to produce CO2 and H2O, and the equation must be balanced. Therefore, our reaction equation that we are trying to solve looks like this:
CH4(g) + 2O2(g) → CO2(g) + 2H2O(g)
Standard Enthalpies of Formation Table
Try to solve this on your own before clicking on the solution.
E. Calculating Enthalpy of Formation of Any Reaction
Now that you understand that formation reactions are the primary thermochemical reactions involved, you can use a simple shortcut and formula to calculate the enthalpy of formation of any substance. You may have noticed in all our examples that we change the signs on all the enthalpies of formation of the reactants, but not the products. We also multiply the enthalpies of formation of any substance by its coefficient in the chemical equation (even though the coefficient is 1). This allows us to make the following statement: the enthalpy change of any chemical reaction is equal to the sum of the enthalpies of formation of the products minus the sum of the enthalpies of formation of the reactants. In mathematical terms,
ΔH°reaction = ∑npΔH°f,p − ∑ nrΔH°f,r
where np and nr are the number of moles of products and reactants, respectively (even if they are just 1 mol), and ΔHf,p and ΔHf,r are the enthalpies of formation of the product and reactant species, respectively. This ∑ (sum of) products-minus-reactants scheme is very useful in determining the enthalpy change of any chemical reaction, if the enthalpy of formation data are available. Because the mol units cancel when multiplying the amount by the enthalpy of formation, the enthalpy change of the chemical reaction has units of energy (joules or kilojoules) only.
Check your understanding:
Use the products-minus-reactants approach to determine the enthalpy of reaction for
E. Summary
- Reactions can occur in one step from reactants to products or in multiple steps.
- Hess's Law states that the change in enthalpy that occurs in a chemical reaction is the same no matter the path taken to get from reactants to products.
- Standard Enthalpy of Formation (ΔH°f ) is the reaction that produces one mole of a substance from its elements in their standard states.
- We can use the values from a table of standard enthalpies of formation to calculate the change in enthalpy for a reaction by piecing the net reaction together in steps.
- coandh.gif
- Enthalpy of Formation.mp4
- image10.gif
- image11.gif
- image9.gif
- Screen Shot 2014-11-17 at 10.31.37 AM.png
- Screen Shot 2014-11-17 at 10.31.53 AM.png
- Screen Shot 2014-11-17 at 10.34.27 AM.png
- Screen Shot 2014-11-17 at 10.34.38 AM.png
- Screen Shot 2014-11-17 at 10.37.30 AM.png
- Screen Shot 2014-11-17 at 10.37.39 AM.png
- Untitled.png
- waterformation.gif