SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
3-4A: Rates of Reaction
Description
In this activity you will discover how to quantify rates of reaction and will learn about how different factors impact the rate of a reaction on a macro- and micro- level.
Page content
A. Rates of Reaction
The rate of a chemical reaction is the speed at which it takes place. In this case, when we say speed, we are referring to the change in concentration of a reactant or product over time. For example in a reaction such as: A + B → AB we can refer to the reaction rate by the equation:
| Δ[AB] |
| Δt |
Where Δ[AB] is the change in concentration of the product AB and Δt is the change in time.
There are different ways to observe reaction rates, including changes in concentration, colour, pH or mass. For example, in a reaction in which a colour change takes place, we can simply observe how long it takes for the colour of a solution to change once the reactants have been combined. In other cases we may want to measure the rate at which one of the products appears or one reactant disappears.
If you would like to explore how reaction rates in an animated environment, click on the simulator below. There will be an evaluation assignment at the end of this lesson that involves using the simulator to test different factors and how they affect reaction rates.
B. Collision Theory
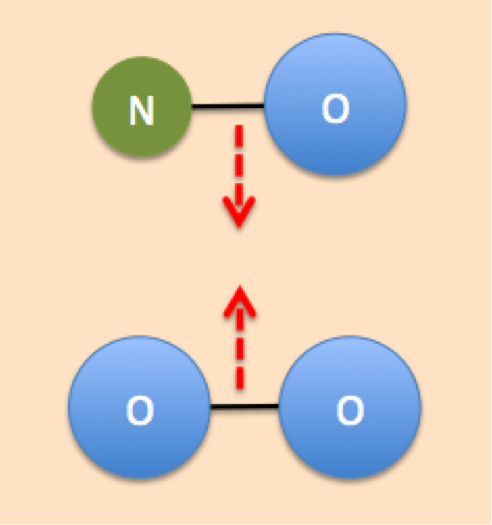
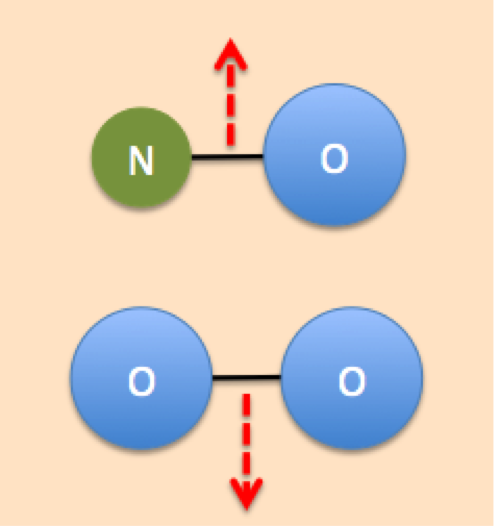
Collision theory states that chemical reactions can only occur if the reactant particles (atoms, molecules, or ions) collide with each other in the proper orientation and with enough kinetic energy. Collision theory helps us to explain how different factors affect the rate of a given reaction. The more collisions that occur between reactants, the faster the rate of reaction. Therefore, if we want to speed up the rate of a reaction, we need to find a way to increase the number of collisions that occur in the proper orientation and enough energy. Conversely, if we want to slow down the rate of reaction, we must reduce the number of collisions that occur.
| Collision = Reaction | No collision = No reaction |
 |
 |
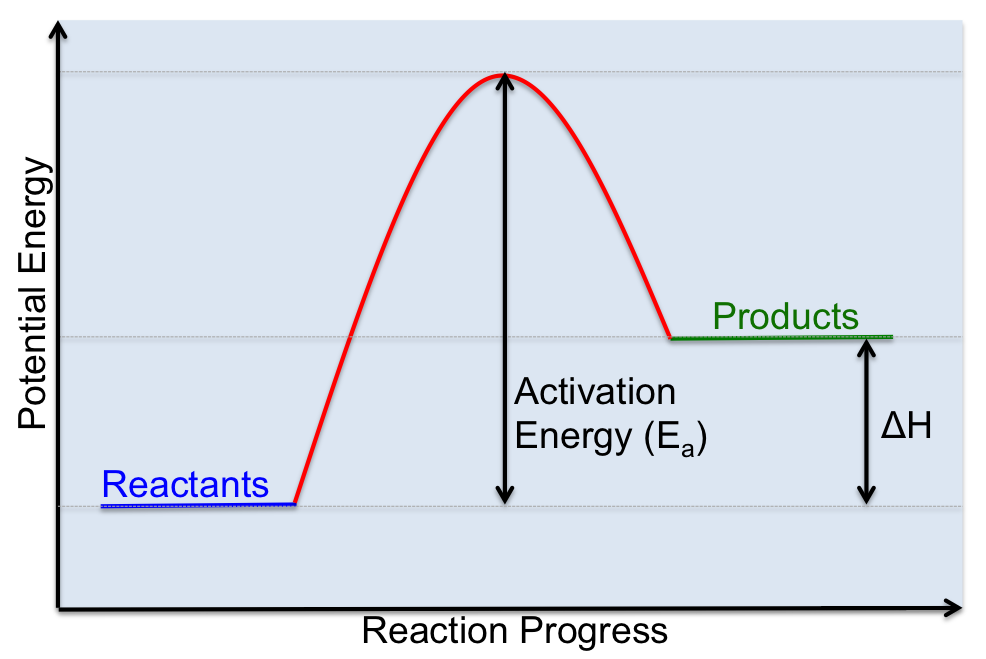
C. Activation Energy (Ea)
As we said above, for a reaction to occur molecules need to collide with each other, but molecules simply colliding doesn't guarantee that a reaction will occur. In fact molecules need to collide, but they must also collide with enough energy to break bonds and overcome any repulsive force between molecules. The minimum amount of energy that reactant molecules must possess to initiate a chemical reaction is called the ACTIVATION ENERGY, which is represented by the symbol: Ea . We can visualize the activation energy for a chemical reaction using a potential energy diagram, which is similar to the enthalpy diagrams that we have already seen. One way to think about activation energy is to imagine you are standing on one side of a wall, on the other side is a swimming pool. If you want to get to the swimming pool, you have to climb over the wall to get there. The energy you expend trying to get over the wall is similar to the activation energy required for a reaction to proceed. If you don't have enough energy to climb over the wall, you can't get to the pool. Similarly, for a chemical reaction, the reactants can't convert into the products unless there is enough energy is present to get over the activation barrier. This amount of energy is known as the activation energy.
| Endothermic Reaction | Exothermic Reaction |
 |
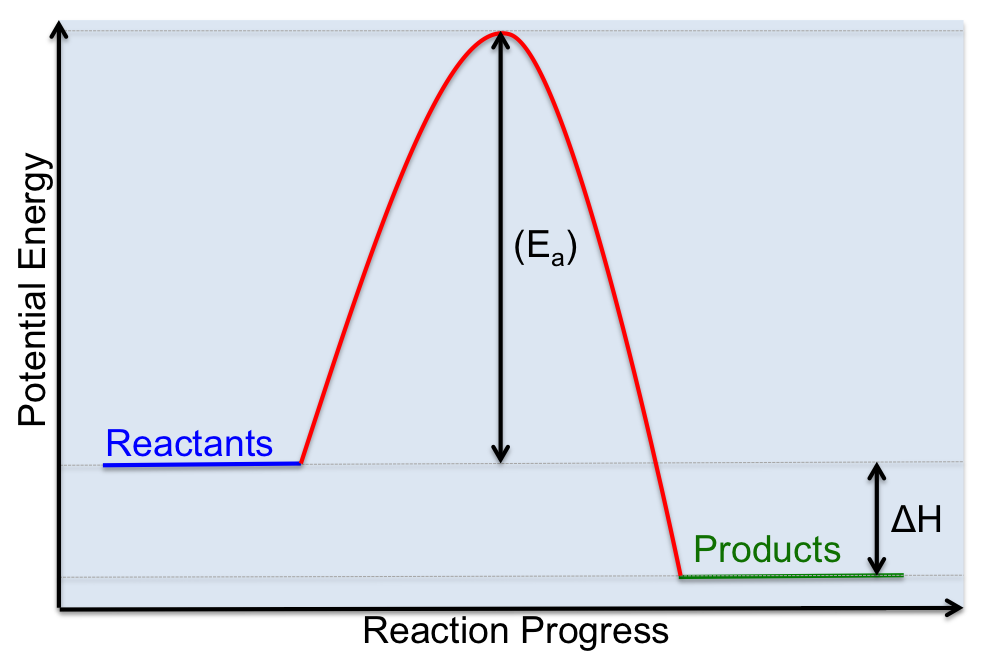 |
The activation energy of a given reaction can help us explain how rates of reaction differ. If we want to increase the rate of a reaction we need to find ways to reduce the activation energy.
Now that we have learned about how collision theory and activation energy affect reactions, we will now apply these concepts to different factors that affect the given rate of a reaction.
D. Factors Affecting Rate of Reaction:
Nature of the Reactants
The speed of a reaction is largely dependent on the properties of the reactants themselves. For example, ions in solution have a very high rate of reaction in precipitation reactions. Conversely, molecules with strong covalent bonds tend to have slow rates of reaction. This can be explained partially by applying the concept of activation energy. In reactions involving reactant molecules with strong covalent bonds, such as ethene, the activation energy for these reactions will be high because these bonds require the input of large amounts of energy to break apart.
Concentration of the Reactants
As the concentration of reactants increases, the rate of reaction also increases, conversely as the concentration of reactants decreases the rate also decreases. This phenomena can be explained easily by collision theory. As we learned above, the greater the number of collisions per second that happen in a reaction, the faster the rate of reaction. When the concentration of a reactant increases, this means that there are a greater number of particles of the reactant in a given area, therefore the number of collisions per second increases.
| Low Concentration | High Concetration |
 |
|
Surface Area of the Reactants
Chemical reactions occur on the surface of reactants. For example, if you throw a log of wood into a burning fire, only the molecules that are on the surface of the log wood will burn (or undergo a combustion reaction) and the log would slowly continue to burn over time. However, if you were to take that same log, and make small shavings (or kindling) and throw those on to a fire, they would burn up much faster than the whole log. This is because by making shavings you are increasing the total surface area and therefore exposing more molecules to react.
| Small surface area = Slow rate of reaction | Large surface area = Fast rate of reaction |
|
|
|
Temperature
Temperature can affect the rate of a reaction in two ways. When the temperature of reactant molecules is increased, the reactant molecules move at faster speeds. This increase in speed increases the probability that two molecules will collide and it also causes the particles that do collide, to do so with more force. The increased force of the collision is associated with higher energy levels, therefore when reactant molecules do collide at higher temperature there is an increased likelihood that they will overcome the activation energy necessary for a reaction to occur. The opposite affect occurs when the temperature is lowered. Molecules move more slowly, collisions are less likely to occur and when they do occur it is less likely that they will have enough energy to overcome the activation energy barrier.
Catalysts
A catalyst is a substance that alters the rate of a chemical reaction without itself undergoing any permanent chemical change. Basically, catalysts are like assistants, that help a reaction proceed at a faster rate. They do this by lowering the activation energy required for a reaction to proceed as you can see in the diagram below.
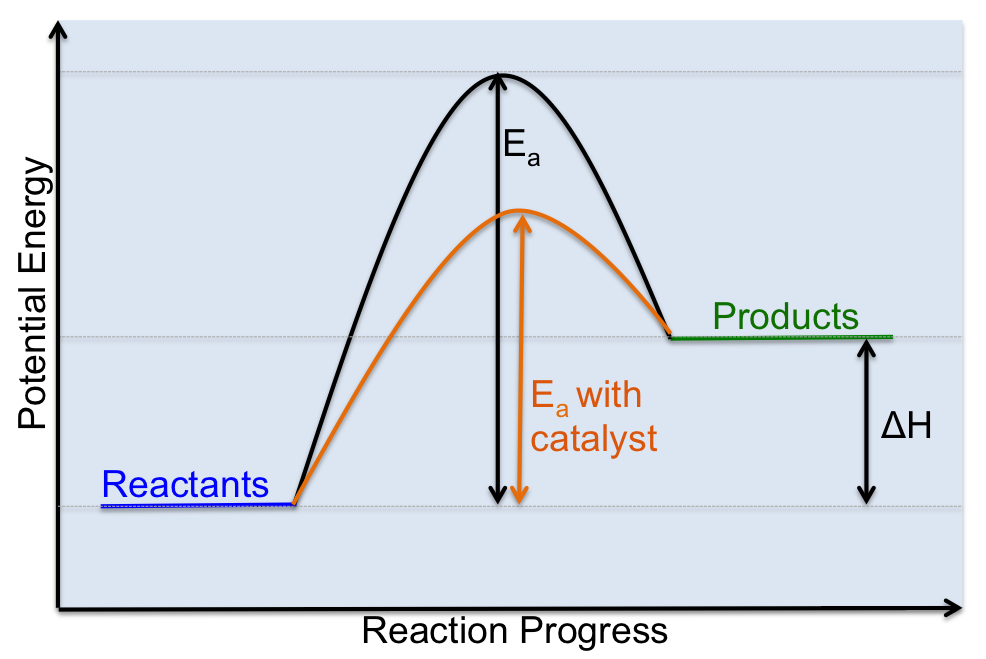
Click here to review some of these concepts relating to rates of reaction.
E. Summary
Rates of reaction
- The rate of a chemical reaction is the speed at which it takes place, measured as a change in concentration of a reactant or product over time.
| Δ[AB] |
| Δt |
- Collision theory states that chemical reaction can only occur if the reactant particles collide with each other in the proper orientation and with enough kinetic energy to overcome the activation energy threshold
- The activation energy (Ea) is the minimum amount of kinetic energy that reactant molecules must have to convert into product
- The rate of a reaction is affected by different factors including:
- the nature of the reactants: certain types of reactant molecules have higher rates of reaction (ions in solution) or lower rates of reaction (molecules with strong covalent bonds)
- concentration of the reactants: higher concentration = higher rate of reaction
- surface area of reactants: larger surface area = higher rate of reaction
- Temperature: higher temperature = higher rate of reaction
- Catalysts: lower the activation energy, increase rate of reaction