SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
3-4E: Reaction Mechanisms
Description
In this activity you will discover how unseen steps in a chemical reaction impact the nature of the reaction and how the rate limiting step relates to reaction rates.
Page content
A. Reaction Mechanisms
When we write the balanced chemical equation for a reaction we are displaying the initial reactants and final products of the reaction. However, most reactions do not occur in a single step in which the reactants are directly converted into products, instead there are typically one or more intermediate species that form on the way to forming the final product. For example, consider the following reaction:
CO(g) + NO2(g) → CO2(g) + NO(g)
The balanced equation suggests that the CO reacts directly with the NO2 to form the products, but the reality is that this reaction occurs in steps. Each individual step that occurs during a reaction is called an ELEMENTARY STEP. In the first step, the NO2 reacts to form NO3 and NO, and in the second step, the CO reacts with the NO3 to produce the final products (CO2 + NO).
Step 1: 2NO2(g) → NO3(g) + NO(g)
Step 2: CO(g) + NO3(g) → CO2(g) + NO2(g)
Now you'll see if you add up these two reaction equations, you end up with our overall reaction equation:
The sequence of elementary steps that occurs as reactant molecules are converted into product molecules is called the REACTION MECHANISM. During the course of a reaction mechanism, different molecules form, including some that don't appear as products or reactants in the overall reaction equation. In the example above, Nitrogen Trioxide (NO3) appears in both steps 1 (product) and 2 (reactant) but is not present in the overall equation. When a molecule that is neither a reactant or product, but is formed and consumed during the reaction mechanism, it is known as a REACTION INTERMEDIATE.
B. Rates of Reaction
So, we've just discovered that reactions can occur in steps in which reaction intermediates can form, the next step is determining how these steps affect the rate of reaction. In the example above, we determined that the overall reaction actually occurs in two steps to produce the final products. It turns out that the two steps of the reaction mechanism occur at different rates, with one proceeding at a faster rate than the other:
Step 1: 2NO2(g) → NO3(g) + NO(g) SLOW
Step 2: CO(g) + NO3(g) → CO2(g) + NO2(g) FAST
Since the first step is the slowest step in the reaction mechanism, the overall reaction cannot occur any faster than this rate. This is similar to the bottleneck effect: when you are pouring out a liquid from a bottle, it is the narrowest part of the bottle that determines how fast the liquid comes out.
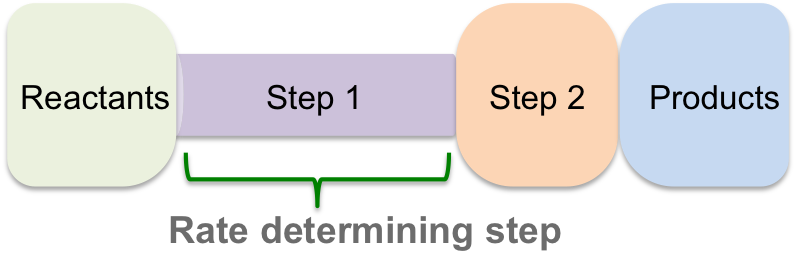
The slowest step in any reaction mechanism is called the RATE DETERMINING STEP. This is the case for any reaction mechanism, whether the slowest step is the first or last step, the overall reaction rate occurs at the slowest rate of any of the steps. Therefore, if you want to determine the rate of any complex reaction, you only need to determine the rate of the slowest step in the reaction mechanism and you will have the value for the overall rate of reaction.
Check your understanding:
A reaction has the following elementary steps,
Step 1: I2(g) → 2I(g) SLOW
Step 2: H2(g) + 2I(g) → 2HI(g) FAST
Determine its overall equation and identify any reaction intermediates and the rate determining step:
Answer 3-4B-1
C. Summary
- Most reaction do not occur as a single step going from reactants to products
- The individual steps that make up a reaction are called the elementary steps
- The sequence of elementary steps that comprise an overall reaction is called the reaction mechanism
- Some molecules that appear in the elementary steps do not appear in the overall equation, these molecules are known as reaction intermediates.
- The rate of a reaction is determined by the slowest elementary step in the reaction mechanism, this is called the rate determining step