SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
4-1B: Chemical Equilibrium
Description
In this activity you will explore how the equilibrium of a system relates to reversible chemical reactions when the concentration of products and reactants stabilizes.
Page content
A. Introduction:
Equilibrium is a term that refers to something that is at rest, or balanced. The word equilibrium has different meanings depending on the context. When a chemical system has reached equilibrium, the concentration of the reactants and products has stopped changing over time.
B. Chemical Equilibrium
Chemical equilibrium is reached when the concentration of the reactants and products in a reaction become constant. Remember that when we represent the concentration of a product or reactant, we use square brackets (eg: [H2O]). When a reaction begins, there is a high concentration of reactants and the concentration of the products is zero. Over time, as the reaction proceeds the concentration of the reactants goes down and the concentration of the products increases. At some point, the concentration of products and reactants stops changing. This is the point of chemical equilibrium. Chemical equilibrium can be reached when a reaction has stopped, but can also be achieved while reactions are still occurring as we will see below for dynamic equilibrium.
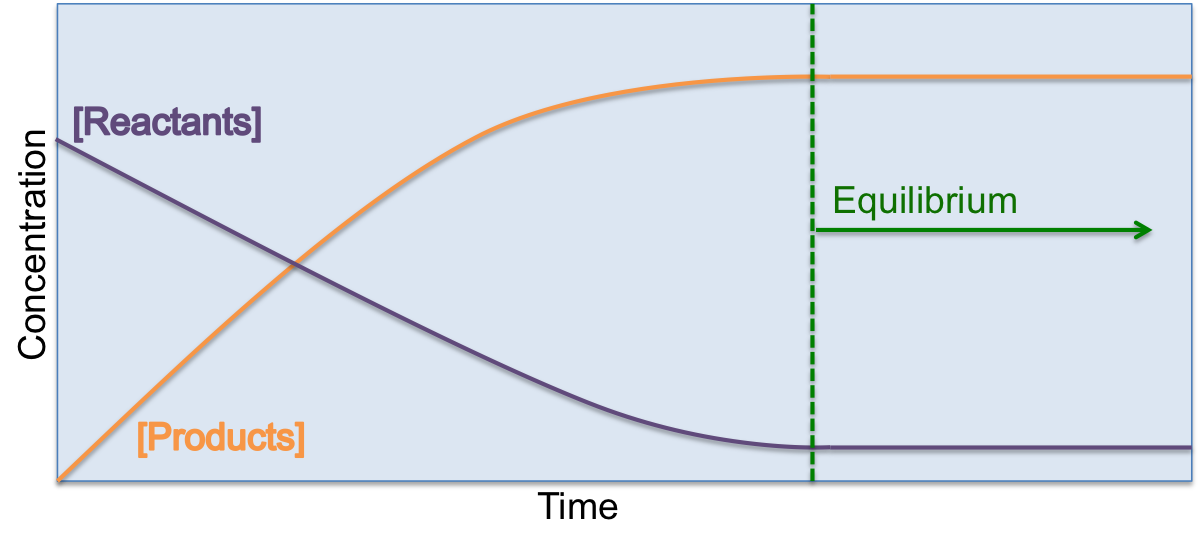
Chemical reactions can proceed in different ways depending on the nature of the reaction and the conditions it takes place in. Some reactions occur to completion where all of the reactant molecules are used up to form product. An example of this would be the complete combustion of methane, whereby all of the methane gas is converted to carbon dioxide and water:
CH4(g) + 2O2(g) → CO2(g) + 2H2O(g)
Other reactions proceed forward, but not all the way to completion. In this case, when the reaction stops, there will still be reactant molecules left over. For example consider the formation of dinitrogen tetroxide gas from nitrogen dioxide taking place in a sealed vial. The sealed vial implies that the reaction is occurring in a closed system, meaning that the system can exchange energy with the surroundings but no matter can escape or enter the system. Nitrogen dioxide has a brown colour, while dinitrogen tetroxide is colourless, therefore we can visualize the reaction taking place by the change in colour over time.
NO2(g) → N2O4(g)
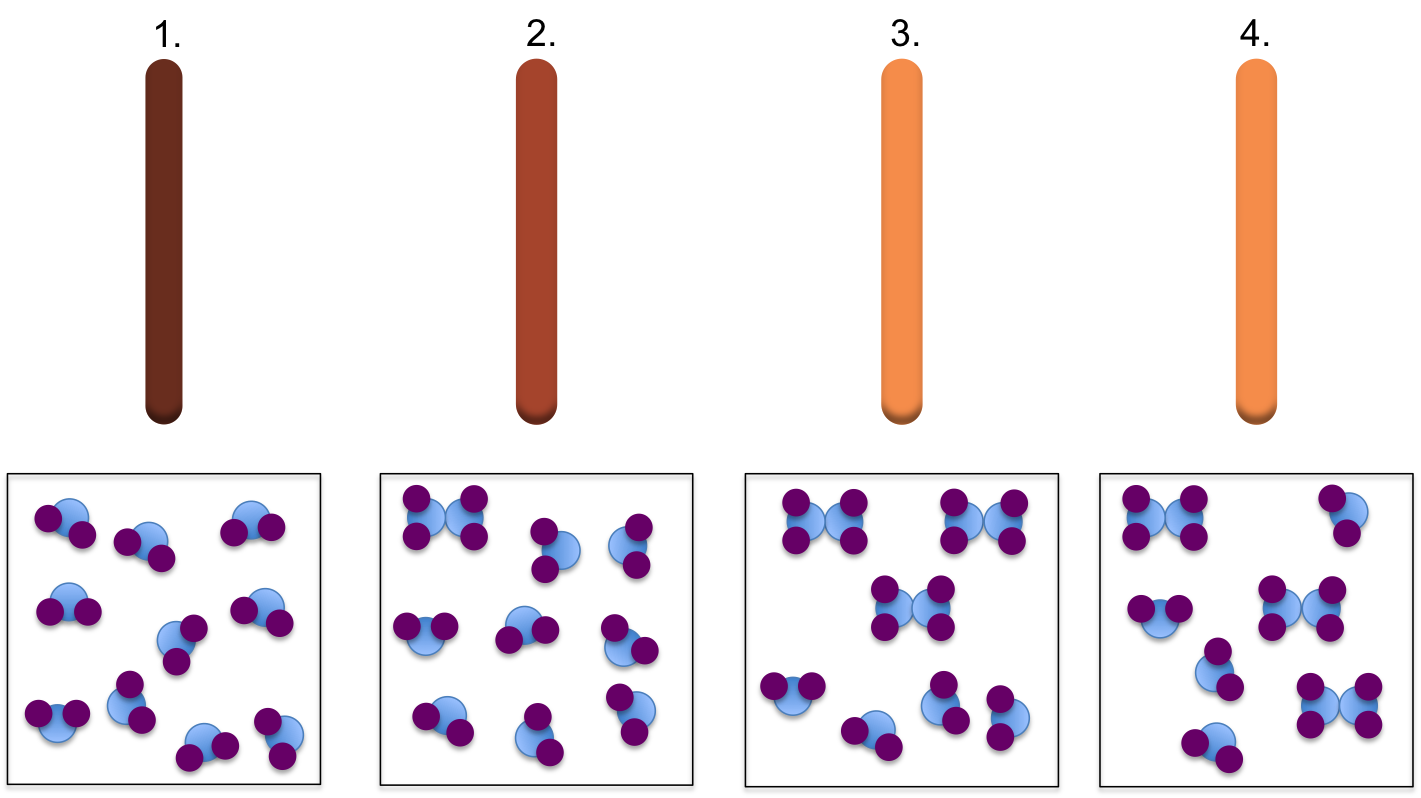
1. When the reaction begins there is only NO2 in the vial, and the colour is dark brown.
2. Over time, the NO2 begins to react, producing colourless N2O4, causing the brown colour to fade slightly.
3 & 4. As the reaction proceeds, more N2O4, is produced resulting in a further fading of the brown colour. Chemical equilibrium is reached when the concentration of reactant and product remains constant. At this point the colour remains constant.
Therefore, CHEMICAL EQUILIBRIUM occurs when all of the reactants and products have reached a constant concentration.
C. Dynamic Equilibrium
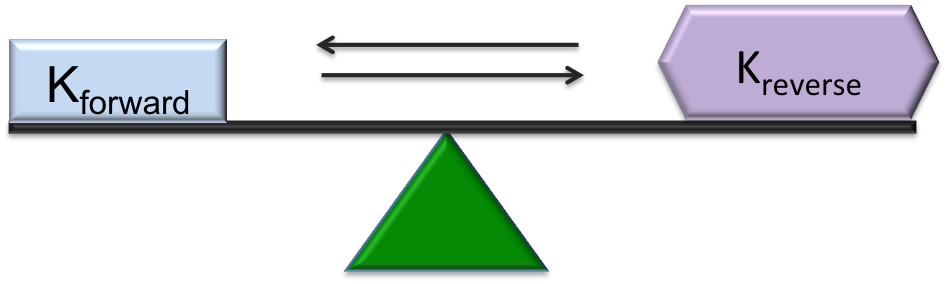
As mentioned above, some reactions are one directional and proceed forward until a state of chemical equilibrium is achieved. Other reactions are reversible, where the reaction can proceed both forwards and backwards. In a reversible reaction, dynamic equilibrium is achieved when the RATES of the forward and reverse reactions are equal. The word dynamic implies that things are changing. Thus, in a system at dynamic equilibrium, reactions are occurring at all times both forwards and backwards, but the rates of each reaction are equal. When a system is at dynamic equilibrium, no observable changes occur in terms of concentrations, colour, pressure etc. This concept of dynamic equilibrium can be applied to different types of reactions and chemical processes. Remember, the key to a dynamic equilibrium is that the forward and reverse rates are equal. We can represent reaction rates by the letter K.
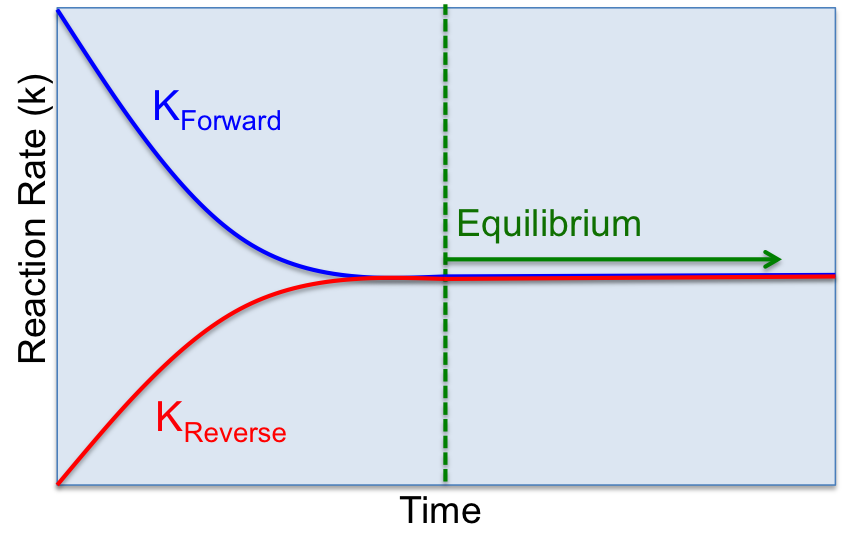
Dynamic Equilibrium: Reversible Chemical Reactions
Consider the following reversible reaction between hydrogen gas and nitrogen gas to produce ammonia gas, occuring in a closed system at high temperature and pressure:
3H2(g) + N2(g) ↔ 2NH3(g)
The forward reaction occurs when H2 and N2 combine to form ammonia (NH3), the reverse reaction occurs when NH3 dissociates into N2 and H2. When the hydrogen and nitrogen are first added, there is no ammonia present. Initially, the rate of the forward reaction is very fast and the rate of the reverse reaction is zero. Over time, as the two reactants react, their concentration goes down and at the same time the concentration of NH3 increases. As the concentration of NH3 builds up, the reverse reaction begins to occur, whereby the NH3 decomposes into H2 and N2. As more and more NH3 decomposes, its concentration goes down and, the forward reaction begins occuring again. There comes a point where the forward and reverse reactions occur at the same rate and the concentration of the products and reactants remains stable. This is the point of dyanamic equilibrium.
| Kforward > Kreverse | Kforward < Kreverse | Dynamic Equilibrium: Kforward = Kreverse |
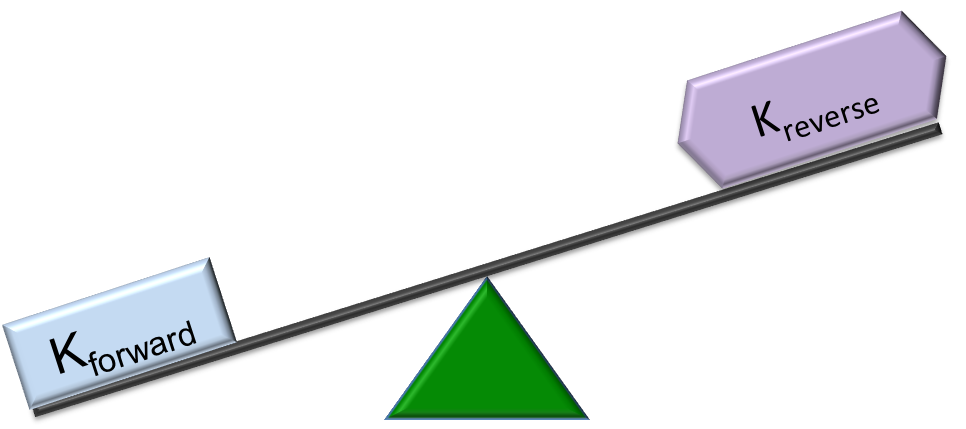 |
 |
 |
Dynamic Equilibrium: Weak Electrolytes in Solution
The concept of dynamic equilibrium can also be applied to a solution of weak electrolytes. For example, the ionization of acetic acid (CH3COOH), in which the acetic acid molecule dissociates into individual ions (H+ & CH3COO-) can also occur in the reverse direction, where the ions recombine to form the acetic acid molecule:
CH3COOH(aq) ↔ H+(aq) + CH3COOH-(aq)
When the rate of the forward reaction (the dissociation of acetic acid) is equal to the rate of recombination of the ions, a dynamic equilibrium is formed.
Dynamic Equilibrium: Liquid Vapour Equilibrium
A dynamic equilibrium can also form for changes of state, for example from liquid to gas and the reverse of that process from gas to liquid. A liquid that is contained in a sealed container will form a dynamic equilibrium with its own vapour. The equation for this process, using water as an example would be:
H2O(l) ↔ H2O(g)
Basically, what is happening in this form of dynamic equilibrium is that some liquid water molecules are evapourating in gaseous water molecules, while at the same time some of the gaseous water molecules are condensing into liquid water when the rate of vapourization is equal to the rate of condensation, dynamic equilibrium has been reached.
Check your understanding:
Which of the following represents chemical equilibrium:
a) [products] = [reactants]
b)The concentration of products and reactants is constant
c) [products] > [reactants]
d) The rate of the forward reaction begins to slow
Answer 4-1-1
D. Summary
- Chemical equilibrium is the point at which the concentration of the reactants and products in a reaction become constant.
- Dynamic equilibrium is the point at which the rates of the forward and reverse reactions are equal.
- Dynamic equilibrium can apply to chemical reactions, the ionization of weak electrolytes in solution as well as transitions between liquid and vapour states.