SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
4-1C: Le Chatelier's Principle
Description
In this activity you will navigate through an interactive activity to explore how systems at equilibrium react to changes in its system and surroundings.
Page content
A. Introduction
We have seen that over time, chemical reactions in a closed system tend to reach a point of dynamic equilibrium. We will now investigate what happens to these systems when a stress is added. In this case a stress just means a change in conditions, which can come in the form of a change in pressure, concentration, temperature, etc. What we are looking at here is how does a stress to the system affect the equilibrium. Does the reaction shift one way or the other? Recall that when a system is at equilibrium, the forward and reverse rates are equal. When the system is stressed the reaction will tend to shift in one direction or the other. For this section when we say the reaction shifts to the right, that means that the forward reaction occurs at a faster rate than the reverse, therefore the concenctration of product molecules will increase, when we say the reaction shifts to the left, we are implying that the reverse reaction occurs at a faster rate and the concentration of reactant molecules will increase. (Note the direction of the arrow)
| Equilibrium | Shift to the right | Shift to the left |
| 3H2(g) + N2(g) ↔ 2NH3(g) | 3H2(g) + N2(g) → 2NH3(g) | 3H2(g) + N2(g) ← 2NH3(g) |
B. Le Chatelier's Principle
In the last learning activity, we learned that over time, reversible chemical reactions tend to reach a point of dynamic equilibrium in which the rates of the forward and reverse reactions are the same.
In general, a system wants to be at the equilibrium point, and if that equilibrium is disturbed, the system will react in such a way so that it can regain a state of equilibrium.
The way that a system reacts when its equilibrium is disturbed can be explained by a concept that is known as Le Chaterlier's Principle which we will now explore in this learning activity.
When a system is at equilibrium the conditions are stable. At equilibrium, pressure, temperature and the concentrations of reactants and products remain constant. However, if any of these conditions change the system is thrown out of balance, and equilibrium is lost. The system wants to be in equilibrium, therefore if the equilibrium is disrupted, the system will act in such a way as to reach a new equilibrium by shifting the reaction in one direction the other. This concept is known as Le Chatelier's Principle.
Le Chatelier's Principle states that if a stress is applied to a system at equilibrium, the system will adjust in a way to oppose that change.
When we say stress in this case we mean a change in the conditions of the system, such as a change in pressure, temperature, concentration etc.
For example if the pressure is increased, the system will act to reduce the pressure, if the concentration of reactants increases the system will act to reduce that concentration and if the temperature is decreased, the system will act to increase the temperature. We will now explore how these different stresses affect systems and how they react to counter the stress.
One way to imagine Le Chatelier's Principle is to imagine you are walking up an escalator is moving down, you will remain in one place. In other words, you are at a state of equilibrium. (Rate walking up = rate of escalator down) If however, the escalator starts moving down at a faster rate (i.e. the system is stressed), you will start to move down the escalator. (Rate up<Rate down). To counteract the stress, you must increase the speed that you are walking up. Once you have increased your speed to the same speed of the escalator, you will once again remain in place, having established a new equilibrium at a different place. (Rate up = rate down)
C. Change in Concentration
Let's imagine we have a system at equilibrium, the concentration of the products and reactants is not changing, and the forward and reverse reactions are occurring at the same rate. What would happen if we all of a sudden added more reactants to the system? For this lesson we will use the reaction for the formation of ammonia gas from its constituents hydrogen and nitrogen gas, at equilibrium:
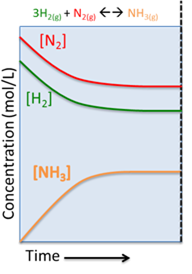
Lets consider an example where the system is stressed in the form of excess N2 added to the system:
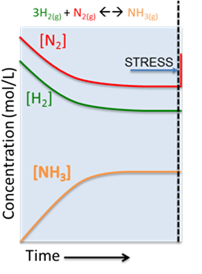
According to Le Chatelier's Principle, the system wants to counter this stress and reduce the concentration of N2. To accomplish this, the reaction must shift to the right consuming the excess N2. The reaction will proceed in this way until a new equilibrium is found .
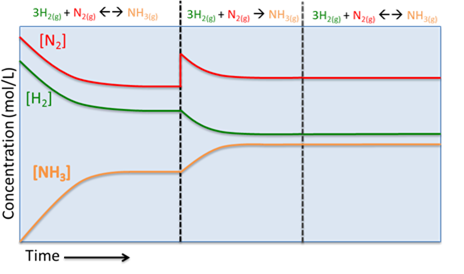
D. Change in Pressure
The pressure of a closed system is directly affected by the number of molecules in the system. If the number of molecules increases, so does the pressure and if the number of molecules decreases, the pressure is lowered as well. If a system at equilibrium is stressed by a change in pressure, the system will act to counter that change by increasing or decreasing the total number of molecules. Lets use our equation from above to see how the system will react to a change in pressure. In this example, each molecule represents one mole. When the system is at equilibrium, there are 18 moles total in a fixed volume.
| 3H2(g) + N2(g) ↔ 2NH3(g) |
| 18 moles total |
|
|
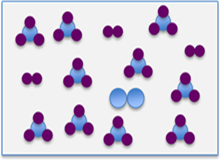
Now, if the pressure of the system were increased by reducing the total volume, we will now have a situation with the same total number of molecules in a smaller volume. The system will act to counter this stress by reducing the total number of molecules. If you look at the reaction equation, we can see that for every 4 moles of reactants (3H2 + 1N2), 2 moles of product are formed. In other words when the reaction proceeds forwards (or to the right) the total number of moles in the system is reduced by 2 (4 - 2 = 2). So, if the system needs to reduce the total number of molecules it will shift to the right and if it wants to increase the total number of molecules it will shift to the left. As you can see in the images below, when the pressure is increased, the reaction shifts to the right, reducing the total number of molecules from 18 moles to 14 moles.
| 3H2(g) + N2(g) ↔ 2NH3(g) | 3H2(g) + N2(g) → 2NH3(g) | 3H2(g) + N2(g) ↔ 2NH3(g) |
| 18 moles total | 18 moles total | 14 moles total |
|
|
 |
 |
E. Change in Temperature
When heat is added to a system at equilibrium, the system will react to try to remove that heat. Conversely when heat is removed from a system at equilibrium, the system will react to produce more heat. In our example above, the forward reaction is exothermic as we can see by the thermochemical equation:
3H2(g) + N2(g) → 2NH3(g) ΔH = - 92.58
or
3H2(g) + N2(g) → 2NH3(g) + Energy
Since the forward reaction is exothermic, this reaction releases heat. Another way to consider this is that energy (or heat) is one of the products. Therefore in an exothermic reaction, if heat is added, the reaction will shift to the left to try to consume this additional energy.
Recall that if a reaction is reversed, the sign of the ΔH is also reversed. Therefore the reverse of this reaction is endothermic:
2NH3(g) → 3H2(g) + N2(g) ΔH = + 92.58
or
2NH3 + Energy → 3H2 + N2
In this example if the temperature of the system is raised by the addition of heat the reaction will shift to the left, to try to counteract the increase in temperature. Remember that in an exothermic heat is released while in an endothermic reaction heat is absorbed, so in our case if the system shifts towards the endothermic reaction the heat that was added to the system will be absorbed.
F. Factors that Do Not Affect Equilibrium
Catalysts
Adding a catalyst does not shift equilibrium. Catalysts only work to speed up rate of reactions (forward/backwards). They can help the reaction achieve equilibrium faster, but they do not change the position.
Adding and removing pure solids and pure liquids
Adding a pure substance (solid or liquid) to a system at equilibrium does not cause a shift in equilibrium. This is because pure solids and pure have fixed concentration (They do not change when added to a reaction). As a result, they are not included in the equilibrium expression K, and have no effect on equilibrium.
G. Summary
- When a stress is added to a system, the equilibrium is disrupted
- A stress is any change in the conditions of a system (for example, changes in: temperature, concentration, pressure, etc.)
- A shift in equilibrium implies that the forward or reverse reaction starts occurring at a faster rate than the other, a shift to the right means that the forward reaction occurs faster than the reverse, while a shift to the left means that the reverse reaction occurs faster than the forward
- Le Chatelier's Principle states that if a stress is applied to a system at equilibrium, the system will adjust in a way to oppose that change.
- According to Le Chatelier's Principle:
- an increase in concentration of a reactant shifts the reaction to the right, while an increase in product concentration shifts the reaction to the left
- an increase in pressure shifts the reaction in the direction that produces fewer gas particles, while a reduction in pressure shifts the reaction in the direction that produces more gas particles
- an increase in the temperature of a system will shift the reaction in the direction that absorbs heat (ie the endothermic direction) while a decrease in the system temperature will shift the reaction in the direction that releases heat
- The addition/removal of solids and pure liquids do not affect equilibrium.
- The addition of catalysts do not affect equilibrium.