SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
4-3A: Acids & Bases Introduction
Description
In this activity you will review some of your previous knowledge of acids and bases and be introduced to new definitions and ways to quantify characteristics of these unique types of chemicals.
Page content
A. Acids & Bases Introduction
 |
Acids & bases are two of the most common classes of chemicals that we find in the world of chemistry. You should be familiar with them from previous chemistry courses. Acids and bases are found in everything from food, to cleaning products to inside our bodies. In this unit we will look at acids and bases in the context of equilibrium, but lets begin with a quick introduction into some of the early theories of acids and bases
B. Arrhenius Acids & Bases
Very early in the history of chemistry, it was recognized that there were classes of chemicals with distinctive properties called acids and bases. In 1887, the chemist Svante Arrhenius suggested that acids are hydrogen-containing compounds that ionize in aqueous solutions to produce hydrogen ions (H+) for example, the ionization of hydrochloric acid (HCl). The equation for the ionization of HCl in water can be written in 2 different ways:
Acid
| H2O | ||
| HCl(g) |
→ | H+(aq) + Cl-(aq) |
or
HCl(g) + H2O(l) → H3O+(aq) + Cl-(aq)
For the purpose of this course, H+ and H30+ are interchangeable and both represent the H+ ion in an acid-base reaction.
Base
Meanwhile, according to Arrhenius, bases are compounds that ionize in water to produce hydroxide ions (OH-) for example, the ionization of sodium hydroxide (NaOH) in water:
| H2O | ||
| NaOH(s) |
→ | Na+(aq) + OH-(aq) |
C. Brønsted-Lowry Acids and Bases
The Arrhenius system is good at identifying and classifying many acids and bases but it does not include all substances that exhibit the properties of acids and bases. So, a new theory was proposed by the chemists Johannes Brønsted and Thomas Lowry that defined acids and bases on their ability to donate or accept a proton (H+), known as the Brønsted-Lowry theory of acids and bases. The Brønsted-Lowry theory of acids and bases defines acids as proton donors and bases as proton recipients. (Keep in mind that a hydrogen ion (H+) is made up of 1 proton and zero neutrons and electrons, therefore, it is simply a proton).
Brønsted-Lowry Acid: In the reaction equation below, notice that the HF gas donates its H+ to a water molecule. Therefore, HF is an acid because it is a proton donor. Alternatively, notice that the H2O accepts the proton from the HF to form H3O+; therefore in this equation, water acts as a base as it is a proton acceptor.
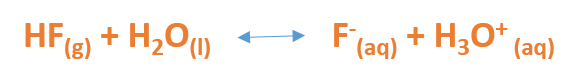
In the Bronsted-Lowry model, the substance that forms when a base accepts a hydrogen ion is called a CONJUGATE ACID. In the example above, the H3O+ is the conjugate acid. Meanwhile, the substance that forms when an acid loses its hydrogen ion is called a CONJUGATE BASE. In the reaction above, the F- is the conjugate base. An acid and its conjugate base are referred to as a CONJUGATE ACID-BASE PAIR. Therefore, HF and F- would be a conjugate acid-base pair, as would H2O and H3O+.
Brønsted-Lowry Base: In the reaction equation below, notice that the NH3 accepts a proton (H+) from the water molecule to form NH4+. Therefore, according to the Brønsted-Lowry theory, NH3 is a base because it is a proton acceptor. Alternatively, notice that the H2O donates the proton (H+) to become OH- therefore, in this reaction, H2O is an acid. This is one of the unique characteristics of water, its ability to act as both an acid and a base depending on the context.
![]()
In the above reaction, NH4+ is the conjugate acid, as it accepted the proton, while the OH- ion is the conjugate base as it lost its hydrogen ion. NH3 and NH4+ would be one conjugate acid-base pair, while H2O and OH- would be the other conjugate acid-base pair.
Remember: In this course, H3O+ and H+ are interchangeable, they both imply a proton from an acid-base reaction.
D. Strong and Weak Acids & Bases
Acids and bases differ in their strength based on the degree that they ionize in water. A strong acid or base is one that ionizes completely in water, fully dissociating into its constituent ions. In the reactions below, notice the reaction arrow indicating the reaction proceeding to completion.
Strong Acid: HCl(aq) → H+(aq) + Cl-(aq)
Strong Base: NaOH(aq) → Na(aq) + OH(aq)
On the other hand, a weak acid or weak base only ionizes slightly in aqueous solution at equilibrium. For example, ethanoic acid (CH3COOH) and ammonia (NH3) only ionizes slightly in water (note the reaction arrow, indicating a dynamic equilibrium between the forward and reverse reactions)
Weak acid: CH3COOH(aq) + H2O(l) ↔ H3O+(aq) + CH3COO-(aq)
Weak base: NH3(g) + H2O(l) ↔ NH4+(aq) + OH-(aq)
The strength of acids and bases can be measured using pH, pOH, Ka and Kb, all of which we will learn about in the next learning activity.
Ka: Acid Dissociation Constant
As mentioned in an earlier lesson, when a weak acid is mixed with water, a small amount of ionization occurs and an equilibrium ultimately forms between the forward and reverse reactions. The Ka is the equilibrium constant that is specific to this ionization reaction for an acid and is represented by the following equation:
Ka = ([H3O+] * [A-]) / [HA]
The larger the Ka value for an acid, the stronger the acid is.
Kb: Base Ionization Constant
Similar to the Ka, the Kb is the equilibrium constant that is specific for the ionization of a weak base.
As we did with Ka, we do not include the concentration of liquid water in our equation for Kb, therefore our equation for Kb is:
Kb = ([BH+] * [OH-]) / [B]
The larger the Kb value, the stronger the base is.
E. Water: Acid or Base?
Water can actually be considered both an acid and a base by the Brønsted-Lowry theory. Water, on its own, in liquid form actually ionizes to a very limited extent through a process called auto-ionization which can be shown by the following reaction equation:
H2O(l) ↔ H+(aq) + OH-(aq)
So, you can see that one water molecule acted as an acid by donating its proton (H+) while the other water molecule acted as a base by accepting a proton. This reversible reaction reaches an equilibrium point that can be characterized by an equilibrium constant: Kw. This constant is referred to as the ionization constant of water and is calculated by the same formula we use for equilibrium constants. As we learned earlier, liquids and solids both have constant concentration values so we do not include them when calculating equilibrium constants. Therefore, our formula for Kw is:
Kw = [H+]*[OH-] = 1 x 10-14 at 25 ºC
As you can see above, experimental results have shown that at 25 ºC, Kw = 1 x 10-14. In pure water, for every H+ ion that dissociates, an OH- ion must also dissociate. Therefore, in pure water, [H+] = [OH-].
We can use this known value for Kw and apply it to other aqueous solutions of acids or bases. In solutions of acids or bases the concentration of the two ions is not equal, in an acidic solution, the [H+] is greater than the [OH-], while in basic solutions, the [OH-] is greater than the [H-]. We can use this relationship and the known value of Kw to find the concentration of either [H+] or [OH-].
For example:
A solution is made at 25 ºC by dissolving 0.45 mol of NaOH in water to make a 3.75 L solution. Determine the [OH-] and [H+].
Note: since we know that NaOH is a strong base, we can assume that it ionizes completely.
Step 1. Determine the concentration of [OH-]
[OH-] = 0.45 mol / 3.75 L
[OH-] = 0.12 M
Step 2. Substitute the value of [OH-] into the equation for Kw
Kw = [H+]*[OH-]
1x10-14 = [H+]*(0.12)
Step 3. Rearrange to solve for [H+]
[H+] = 1x10-14 / (0.12)
[H+] = 8.33 x 10-14
Step 4. Solution:
Therefore, at 25 ºC, when 0.45 mol of NaOH is dissolved in water, the [OH-] is 0.12 M and [H+] is 8.33 x 10-14. As you can see, the concentration of hydroxide ions is much much greater than the concentration of hydrogen ions. This makes sense since we know that NaOH is a strong base.
F. Summary
- Arrhenius acids & bases are classified based on their ability to ionize and form H+ or OH- ions
- Brønsted-Lowry classified acids and bases based on their ability to donate or accept a proton (hydrogen ion). According to Brønsted-Lowry an acid is classified as a proton donor while a base is classified as a proton acceptor
- A strong acid or base is one that ionizes completely in water
- A weak acid or base is one that ionize slightly in water, forming an equilibrium between the forward reaction in which ionization occurs and the reverse reaction in which the ions recombine
- The equilibrium constant of a generic weak acid (HA) is called the acid dissociation constant (Ka) and is calculated by the formula: Ka = ([H3O+] * [A-]) / [HA]
- The equilibrium constant of a generic weak base (B) is called the base dissociation constant (Kb) and is calculated by the formula: Kb = ([BH+] * [OH-]) / [B]
- Water can act as an acid or a base according to Brønsted-Lowry theory as H2O ionizes to a small degree to form H+ and OH- ions
- The equilibrium constant for the ionization of water (Kw) is calculated by the formula: Kw = [H+]*[OH-], which has a known value at 25 ºC of 1 x 10-14