SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
4-3B: Properties of Acids and Bases
Description
In this activity, we will review the properties of acids and bases. We will also learn how to identify and name different acids and bases.
Page content
Properties of Acids and Bases
Introduction
Acids and bases play a central role in much of the chemistry that affects our daily lives. Our body needs acids and bases to function properly. The creation of giant limestone structures and the different colours of flowers found in most gardens are a few of the products of the chemical reactions involving acids and bases.
Acids
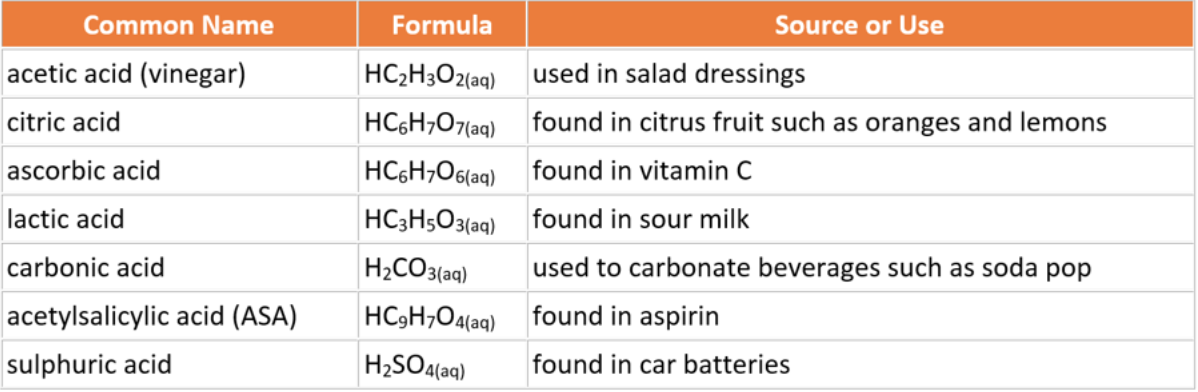
Acidic compounds give foods a sour taste. For example, vinegar gives a sour taste to salad dressing. Vinegar contains ethanoic acid, sometimes known as acetic acid.
Acids are ionic compounds that produce hydrogen ions when dissolved in water, H+(aq). Acids occur naturally in many fruits, as by-products of the metabolism, and are produced by bacteria that sour milk (lactic acid), and many insects.
Bases
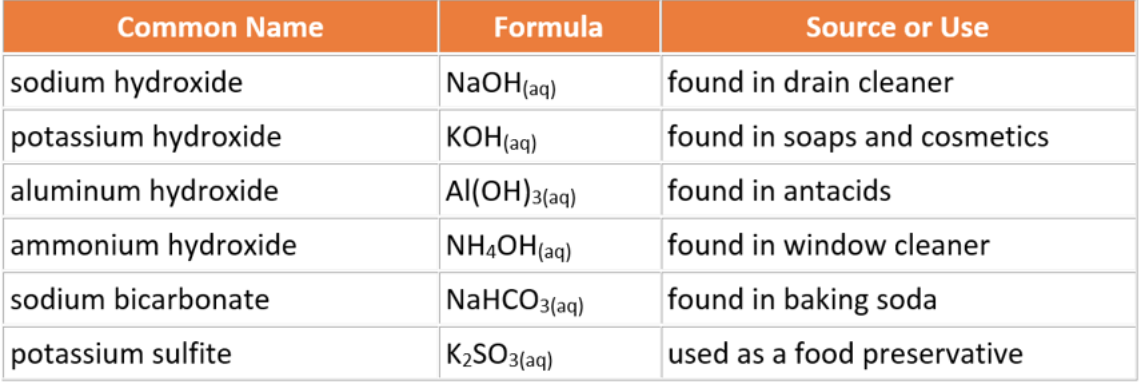
Bases have a bitter taste, but tasting most bases is hazardous. The slippery feel of soap in another property of bases. Another word for base is alkaline.
Bases are ionic compounds that dissociate in water producing hydroxide ions in solution, OH-(aq). Most household cleaning products found in your home, such as soaps, drain and window cleaners are bases. When the dentist freezes areas of your mouth before filling a cavity, a local anaesthetic, a base, is used for this purpose.
Concentration of Ions
Acids and bases range in their respective concentrations of hydrogen and hydroxide ions. Therefore some of them are harmless (low concentrations of H+ or OH- ions) while some of them can be quite dangerous (high concentrations of H+ or OH- ions). The tables list the most common acids and bases used in everyday life.
List of Common Acids
List of Common Bases
Naming Acids
When the chemical formula of an acid starts with H and has only one other non-metallic element, it is named by adding the prefix 'hydro' and the suffix 'ic' and the word 'acid'.
HCl --> Hydrochloric acid
HF --> Hydrofluoric acid
Acids that contain a polyatomic ion and an oxygen atom can be named as follows:
a) If the polyatomic ion ends in 'ate' add the suffix 'ic' and the word 'acid'.
SO42- is sulphate, so H2SO4 --> sulphuric acid
b) If the polyatomic ion ends in 'ite' add the suffix 'ous' and the word 'acid'.
SO32- is sulphite, so H2SO3 --> sulphurous acid
Naming Bases
The name of a base can be determined from the name of the positively charged metallic ion or polyatomic ion at the beginning and then adding the word 'hydroxide'.
NaOH --> Sodium hydroxide
NH4OH --> Ammonium hydroxide
Some bases are more difficult to recognize. Substances that contain bicarbonate/hydrogen carbonate, HCO3- are bases because they react with water to form hydroxide ions.
NaHCO3 --> Sodium bicarbonate
Summary - Acids
An acid is a compound that dissolves in water to produce hydrogen ions (H+) in solution.
For example: Hydrochloric acid (HCl) consists of hydrogen ions and chloride ions that separate when dissolved in water.
HCl(aq) ---> H+(aq) + Cl-(aq)
- Commonly contain H+
- Water-soluble (this can make them more dangerous)
- Sour-tasting
- Corrosive
- Conduct electricity
Summary - Bases
Bases are compounds that dissolve in water to produce hydroxide ions (OH-) in solution.
For example: Sodium hydroxide produces sodium ions and hydroxide ions when it dissolves in water.
NaOH(aq) ---> Na+(aq) + OH-(aq)
- Commonly contain OH-
- Water-soluble (this can make them more dangerous)
- Bitter-tasting
- Corrosive
- Conductive
- React with Protein (eg. those in your skin and eyes)