SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
4-3C: pH Scale
Description
In this learning activity, we will describe how the pH scale is used to identify some compounds and learn about their properties. We will explore about acid-base indicators and will investigate acids and bases through interactive media.
Page content
1. Review:
You will recall that:
- Acids are substances that react with metals and carbonates, conduct electricity, turn blue litmus red, and neutralize bases. Acids are very useful in the food industry, for instance, they act as a preservative. For example, vinegar and lemon juice act as preservatives.
- Just like acids, bases in aqueous solutions conduct electricity and turn red litmus blue. Bases are electrolytes, meaning that their solutions are good conductors of electricity. For example, sodium hydroxide is an electrolyte because it completely separates into its ions as it dissolves in water.
2. Identifying Acids and Bases:
Since most acids and bases are clear, colourless liquids it is difficult to distinguish them from one another or even from water by using their appearance, alone. One way to identify acids from bases is through their different tastes. Acids such as vinegar taste sour while dilute bases, like quinine, taste bitter; however, since some acids and bases are very corrosive, tasting these substances is not a safe practice.
Another way to distinguish acids from bases is by using an acid base indicator. An acid base indicator is a chemical substance that changes colour as the concentration of hydrogen ions, or hydroxide ions in a solution, changes.
Two of the most common indicators are litmus and phenolphthalein.
Litmus is a plant extract that can be blue or red; it appears pink. Red litmus paper turns blue in the presence of a basic solutions and blue litmus paper turns red in the presence of an acidic solution. If the solution is neither acidic nor basic, it is said to be neutral. Water is a neutral solution and does not change the appearance of either red or blue litmus paper.

Phenolphthalein is a weak acid that is colourless; it turns bright pink in the presence of a basic solution. Instead of using chemical indicators, natural indicators such as red cabbage juice and blackberries, can also be used.

3. pH and the pH Scale:
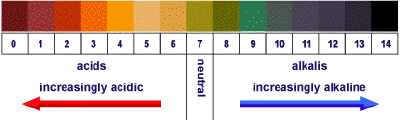
Chemists measure the acidity of a solution using a scale called "pH". pH is a measure of how acidic or basic a solution is. The pH scale is a numerical scale of all the possible numerical values of pH from 0 to 14.
In scientific terminology, pH is an abbreviation that stands for power (potential) of hydrogen. It is a measurement related to the number of hydrogen ions in a solution. A change in pH of one is really a 10 fold (10 times) change in how acidic, or basic, a solution is. An acid with a pH of 1, such as hydrochloric acid has a concentration of H+ ions of one tenth (1/10th) while an acid with a pH of 3, such as vinegar, has a concentration of H+ ions of one thousandth (1/103 or 1/1000th). In other words the concentration of H+ ions is 100 time higher in the hydrochloric acid than in the vinegar. Consequently, the concentration of H+ ions is higher in a solution with a pH of 1.0 than one with a pH of 3. Another example would be a solution with a pH of 3 is 10x more acidic than a solution with a pH of 4 and 100x more acidic than a solution with a pH of 5.
Now let's compare two basic solutions. A solution of sodium bicarbonate has a pH of approximately 8 while a solution of drain cleaner has a pH of about 13. This means that the concentration of hydrogen ions in the bicarbonate solution is higher than the drain cleaner solution. Since basic solutions measure the concentration of hydroxide ions rather than hydrogen ions in solution, the drain cleaner has a higher concentration of hydroxide ions and it has lower concentration of hydrogen ions in solution. For example, a solution with a pH of 13 is 10x more basic than a solution with a pH of 12 and 100x more basic than a solution with a pH of 11.
Therefore, the higher the pH value of a basic solution, the more basic it is.
The pH of a substance can only be determined when it is dissolved in water - an aqueous solution.
-
- A neutral substance is neither an acid nor a base and has a pH of 7 when it is in an aqueous solution.
- An acid is a substance that has a pH less than 7 when it is dissolved in water. The more acidic a substance is, the lower its pH value.
- A base is a substance that has a pH greater than 7. The more basic a substance is, the higher its pH value. The image below shows the pH values of some common substances and solutions.
The pH scale is a scale that indicates how acidic or how basic a solution is when it is measured. The pH scale provided illustrates some common household substances and solutions for which you may encounter and their pH value.

It is not possible to determine the pH of all solutions using just one indicator, such as litmus. A universal indicator is a mixture of chemicals that changes colour through a wide range of pH values. When pH paper is dipped into a solution one of these indicators will change colour and can then be compared to a standard colour chart. pH meters can also be used to more accurately determine pH values of aqueous solutions.
The following video will demonstrate the basics about the pH scale, universal indicator and litmus paper:
Source: Fuse School
4. Some Applications of pH:
(i). pH and Soil
The pH of soil can vary considerably, depending on the type of rock in the area, the kinds of plants growing there, and the materials that people have added. Different plants grow best in different conditions of soil acidity. For example, beans and clover grow in soil that is slightly basic (pH 7 to 10). Corn survives in mildly acidic soil (pH 5 to 6). Potatoes, however, prefer acidic soil that is below pH 5.
(ii). pH and Consumer Products
Many of your shampoo and skin care products were probably designed to have a pH that is close to neutral. However, many cleaning products (e.g. bleach) contain high concentrations of hydroxide ions. These cleaners are very corrosive, which make them effective at cleaning but also potentially quite damaging to skin and eyes.
(iii). pH and Swimming Pools
maintaining a backyard swimming pool or a hot tub require keeping a close watch on the pH of the water. Ideally, the pH of the water should be maintained within a narrow range of 7.2 to 7.8. The reason for that is microorganisms can’t grow at that pH range. If the pH falls below 7, then the water irritates the eyes. If it is above 8, then the water becomes cloudy and the chlorine compounds used to disinfect begin to lose their effectiveness.
