SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
4-3D: pH, pOH and Buffers
Description
In this lesson you will learn about ways to quantify the acidity or alkinity of acids and bases as well as unique types of chemicals called buffers which can act to maintain a specific pH.
Page content
A. pH and pOH
|
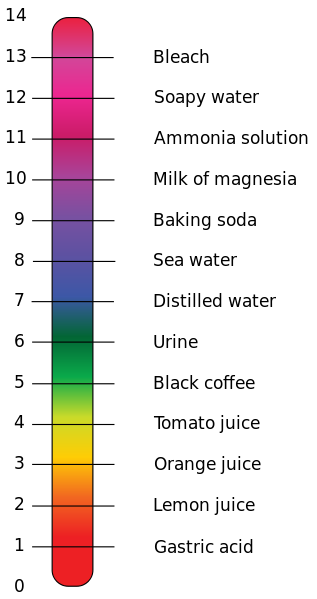
Acids and bases are characterized as weak or strong depending on their ability to ionize and form H+ or OH- ions. Chemists have developed a scale that allows us to quantitatively measure the strength of a given acid or base that is known as the pH scale. The pH scale is a measure of the concentration of the hydrogen ion in acidic and basic solutions and ranges in values from 0 to 14. Pure water, which is considered neutral has a pH of 7, acids have pH values below 7 with stronger acids having lower values, while bases have pH values above 7 with stronger bases having higher values. To calculate pH values we use the following formula: pH = -log[H+] this equation can be rearranged if you want to calculate the concentration of H+in a solution if given the pH value: [H+] = 10-pH For acid base reactions where we only know the [OH-] we can use the pOH scale which is simply calculated with the formula: pOH = -log[OH-] This equation can be rearranged to calculate for the [OH-]: [OH-] =10-pOH Once we know the value of either pH or pOH we can calculate the other value with the simple formula: pH + pOH = 14 As you can see the pH scale is logarithmic, meaning every time you increase a pH value by 1, you are really increasing by a factor of 10. In other words a pH of 4 is 10 times more acidic than a pH of 5 and a pH of 12 is 10 times more basic than a pH of 11 To refresh your memory on how logarithmic equations work, click this LINK Lets try a couple of sample problems using these equations: Calcluating pH and pOH from [H+] 1. Find the pH and pOH of a solution that has a [H+] of 4.2 x 10-4 M and determine whether it is acidic or basic: pH = -log[H+] pH = -log(4.2 x 10-4) pH = -(-3.38) pH = 3.38 pH + pOH = 14 pOH = 14 - pH pOH = 14 - 3.38 pOH = 10.12 Therefore the pH of the solution is 3.38 and is acidic. The pOH of the solution is 10.12 Calculating [H+] from pH 2. Calculate the [H+] in a solution that has a pH of 12.4 [H+] = 10-pH [H+] = 10-12.4 [H+] = 3.98 x 10-13 M Therefore the concentration of H+ ions in a solution with pH of 12.4 is 3.98 x 10-13 M. This is a very small value which makes sense because a pH of 12.4 indicates a very basic solution which should by definition have very little H+ |
 |
Calculating [H+] or [OH-] using Kw
3. A solution of HCl at 25 ºC has a hydrogen ion concentration of 1 x 10-3 M. Calculate the pOH and [OH] using Kw.
Kw = [H+]*[OH-]
We know that at 25 ºC, Kw = 1 x 10-14, therefore:
1 x 10-14 = [H+][OH-]
1 x 10-14 = (1 x 10-3)*[OH-]
[OH-] = (1 x 10-14) / (1 x 10-3)
[OH-] = 1 x 10-11 M
pOH = -log[OH-]
pOH = -log(1 x 10-11)
pOH = 11
Therefore, the solution has [OH-] of 1x10-11 M and a pOH of 11.
B. Buffers
Many chemical and biological processes require very specific pH levels to function properly. In order to maintain these specific pH levels, a type of solution called a buffer is used. A buffer is a solution that resists changes in pH by absorbing or releasing hydrogen or hydroxide ions in a solution. To achieve the goal of maintaining a constant pH, buffers are typically made up of a weak acid and its conjugate base or a weak base and its conjugate acid. For example, in the cells that make up our bodies, a phosphate buffering system is used in which the weak acid, H2PO4- is in equilibrium with its conjugate base, HPO42-
H2PO4- (aq) ↔ HPO42-(aq) + H+(aq)
This buffer will resist any pH changes by shifting its equilibrium according to Le Chatelier's Principle. For example, if H+ is added to the buffer solution, which would typically lower the pH, the equilibrium will shift to the left, to consume the excess H+ and maintain a constant pH. On the other hand, if OH- is added, which would typically raise the pH, the OH- ions will react with the H+ ions, forming H2O, this will cause the equilibrium to the right to make up for the reduced concentration of H+ and again, maintain a constant pH.
C. Summary
- pH and pOH are measures of the strength of an acid or base.
- Pure water has a pH of 7 is considered neutral
- Acids have pH values less than 7 while bases have pH values greater than 7
- The lower the pH value, the stronger the acid, the greater the pH value the stronger the base
- pH = -log[H+]
- [H+] = 10-pH
- pOH = -log[OH-]
- [OH-] = 10-pOH
- pH + pOH = 14
- Buffers are solutions that maintain a constant pH
- Buffers are made up of a weak base and its conjugate acid or a weak acid and its conjugate base