SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
4-3F: Titration
Description
In this activity you will learn about an experimental technique that can be used to determine unknown pH levels by performing titrations.
Page content
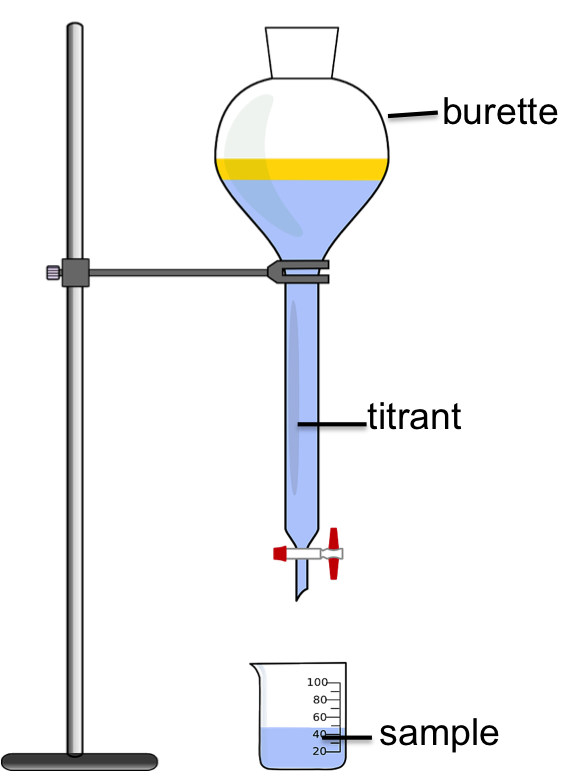
A. Titration
|
When an acid and base are mixed together, the hyrdogen and hydroxide ions combine to form water. This simple reaction can be used by chemists to determine concentrations and pH values for acids and bases using a laboratory technique known as titration. In a typical acid-base titration, a sample (either an acid or a base) is placed in an Erlenmeyer flask or beaker. An indicator is added to the sample that changes colour according to the pH of the sample. Drops of solution of a known concentration (acid if the sample is a base or vice-versa if the sample is a base), known as the titrant are then slowly added to the sample, drop-by-drop, from an apparatus known as a burette. When the sample has been completely neutralized (ie when all of the H+ or OH- in the sample has been consumed), the indicator will change colour. The point at which the indicator changes colour is known as the equivalence point. Once the equivalent point is reached, the volume of titrant used is recorded and from this information, we can calculate the concentration or pH of the sample. |
|
B. TItration: Weak Acids and Strong Bases
TItration is a technique that can be used to determine the concentration or pH of an unknown solution. In a titration between a weak acid and a strong base, the weak acid acts as the sample and the strong base as the titrant. Lets consider the titration between hydrofluoric acid (HF) which is a weak acid, and sodium hydroxide (NaOH) which is a strong base. Since HF is a weak acid, it will have a low Ka value and at equilibrium very little ionization will have occured, the following equilibrium will exist for HF in solution before the titration begins:
HF(aq) ↔ H+(aq) + F-(aq)
Since HF is a weak acid, the equilibrium position is far to the left, meaning that very little ionization has occured. On the other hand, the titrant, NaOH, is a strong base, which means that it is almost completely ionized:
NaOH(aq) → Na+(aq) + OH-(aq)
When the titrant is added, the OH- ions from the titrant will react with the H+ ions in the sample to produce H2O(l)
H+(aq) + OH-(aq) → H2O(l)
As titrant is added, H+ (product) is consumed. Recall from Le Chatelier's Principle, that when a system at equilibrium is stressed, the system responds to counteract that stress. Therefore, when the OH- from the titrant reacts with the H+ from the sample, the equilibrium of the sample will shift to the right to try to replace the H+ ions that are consumed. The EQUIVALENCE POINT is reached when all of the HF molecules have ionized to form H+ and F- ions, and the H+ ions have reacted with the OH- ions to produce H2O, in other words, at the equivalence point, the sample has been neutralized and the reaction has been pushed all the way to the left. In a titration of a weak acid with a strong base, at the equivalence point, the number of moles of OH- ions added is equal to the total number of moles of the weak acid before ionization. Using the concentration and the volume of titrant that was used to neutralize the sample we can calculate the concentration of the sample.
Sample Problem:
25 mL of hydrofluoric acid is titrated with a 0.4M solution of NaOH. It takes 55 mL of NaOH for the indicator to change colour, indicating the equivalence point. Determine the concentration of the acid.
Step 1. Determine the amount of OH required to reach the equivalence point:
nNaOH = c x V
nNaOH = 0.4 mol/L x 0.055 L
nNaOH = 0.022 mol
Therefore, it took 0.022 moles of NaOH to neutralize the HF
Step 2. Determine the concentration of the sample.
Since each mole of NaOH neutralizes 1 mole of HF, there must be 0.022 moles of HF in the sample, and since we know that there are 25 mL (or 0.025 L) of sample we can calculate the concentration:cHF = n / V
cHF = 0.022 moles / 0.025 L
cHF = 0.88 moles/L
Therefore the concentration of hydrofluoric acid is 0.88 moles/L
C. Calculating Ka or Kb Using Titration
In a titration experiment, a sample of unknown concentration is titrated with a titrant of known concentration until it is neutralized. When the sample is neutralized, an indicator changes colour, indicating that the equivalence point has been reached. At the equivalent point, we can determine the initial concentration of the sample. If we also know the initial pH of the sample we can use a titration to calculate the Ka for a weak acid or Kb of a weak base.
Sample Problem:
25 mL of the weak acid, hydrofluoric acid, at a pH of 3, is titrated with a 0.4M solution of NaOH. It takes 55 mL of NaOH for the indicator to change colour, indicating the equivalence point. Calculate the Ka for HF.
What we need to calculate the Ka is the equilibrium concentrations of the products and reactants of the ionization of HF. To do this we will need to set up an ICE table. From the titration data we can determine the initial concentration of the weak acid, before equilibrium is reached. We can than use the pH to determine the concentration of the H+ and F- ions.
Step 1. Determine [HF]initial. From the titration, at the equivalence point we will find out [HF]initial
a) Determine the amount of OH- required to reach the equivalence point:
nNaOH = c x V
nNaOH = 0.4 mol/L x 0.055 L
nNaOH = 0.022 mol
Therefore, it took 0.022 moles of NaOH to neutralize the HF
b) Determine the initial concentration of the sample.
Since each mole of NaOH neutralizes 1 mole of HF, there must be 0.022 moles of HF in the sample, and since we know that there are 25 mL (or 0.025 L) of sample we can calculate the concentration: [HF] = n / V
[HF] = 0.022 moles / 0.025 L
[HF] = 0.88 moles/L
Therefore the [HF]initial is 0.88 moles/L
Step 2. Determine [H+]eq and [F-]eq.
We know that the pH at equilibrium is 3.
[H+]eq = 10-pH
[H+]eq = 10-3
[H+]eq = 0.001 M
From the equilibrium reaction equation for the ionization of HF:
HF(aq) ↔ H+(aq) + F-(aq)
we can see that for every mole of H+ produced, 1 mole of F- is produced. Therefore, [H+]eq = 0.001M and [F-]eq = 0.001M
Step 3. Write the equation for the Ka of HF:
Ka = [H+]eq*[F-]eq / [HF]eq
Step 4. Set up the ICE table:
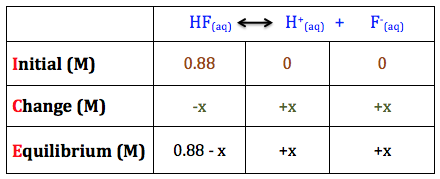
Step 5. Plug in the equilibrium values from the ICE table into the equation for Ka:
Ka = [H+]eq*[F-]eq / [HF]eq
Ka = (x)*(x) / (0.88 - x)
Ka = (0.001)(0.001) / (0.88-0.001)
Ka = (0.000001) / (0.879)
Ka = 1.1 x 10-6
Therefore the acid dissociation constant (Ka) for HF is 1.1 x 10-6
Summary
- Titration is a laboratory technique that can be used to determine the concentration of unknown acids & bases
- In a titration, an unknown sample is placed in an erlenmeyer flask and an indicator that changes colour with pH is added.
- The titrant is a solution with a known concentration that is added drop-by-drop into the unknown sample
- The equivalence point is the point at which the titrant has completely neutralized the unknown sample