SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
5-1B: Oxidation Numbers
Description
In this activity you will be introduced the the concept of oxidation state, and learn how to determine the oxidation numbers of individual elements as well as the oxidation numbers of elements within a molecule.
Page content
A. Oxidation Numbers/Oxidation State
The oxidation number (or oxidation state) of an atom is a positive or negative number that is given to an atom to indicate the degree of oxidation or reduction in a redox reaction. Oxidation numbers are used to keep track of electrons during redox reactions, so that we can track which atoms are oxidized or reduced and to what extent.
A positive oxidation number means that an atom is in its oxidized state (i.e.that it has lost electrons), the higher the number, the greater the extent of the oxidation. A negative oxidation number means that an atom is in its reduced state (i.e. that it has gained electrons).
There are a set of rules for some specific atoms that we can follow to determine the oxidation number of a species in a redox reaction:
1. An oxidation number of zero is given to an atom in its elementary state.
Examples:
-
- Na(s) :oxidation number = 0,
- O2(g):oxidation number = 0
2. The oxidation number of a monoatomic ion is the same as its charge
Examples:
-
- Cl- : oxidation number = -1;
- Mg2+ : oxidation number = +2
3. The oxidation number for hydrogen is +1 in all compounds (except metal hydrides (metal + hydrogen) where it is -1)
Examples:
-
- H2O: Oxidation number for hydrogen is +1
- HCl: Oxidation number for hydrogen is +1
- LiH (metal hydride): Oxidation number for hydrogen is -1
4. The oxidation number for oxygen is -2 in all compounds (except peroxides, H2O2 in which it is -1)
Examples:
-
- H2O: the oxidation number for oxygen is -2
- CO2: the oxidation number for oxygen is -2
5. For Alkali metals (elements in the first column of the periodic table) the oxidation number is +1.
Examples:
-
- Na: Oxidation number is +1
- K: Oxidation number is +1
6. For Alkaline-earth metals (elements in the second column of the periodic table) the oxidation number is +2.
Examples:
-
- Be: Oxidation number is +2
- Ca: Oxidation number is +2
7. All other oxidation numbers are assigned so that the sum of the oxidation numbers equals the net charge on the molecule or ion. Therefore, for neutral molecules, the net sum of the oxidation numbers must equal zero; whereas for ions the net sum of the oxidation numbers must equal the charge of the ion. To determine the oxidation numbers for atoms in a molecule, we first apply rules #1-4 above than we apply rule #5 for all other atoms.
Examples: CO2 : We know that the oxidation number for oxygen is -2, and because there are 2 oxygen atoms in this molecules, the total contribution of oxidation numbers by oxygen is -4 (2 x -2). Since the CO2 molecule is neutral, the sum of the oxidation numbers must equal zero. Therefore the oxidation number for carbon in the CO2 molecule is +4
Sample Problem:
What is the oxidation number of chromium (Cr) in the polyatomic ion Cr2O72-
Step 1. Apply rules #1-4:
We know the oxidation number for oxygen is -2, since there are 7 total oxygen atoms in this ion, the total oxidation number contribution for oxygen is 7 x (-2) = -14.
Step 2. Assign oxidation numbers to any elements not covered by rules #1-4 to produce a net oxidation number equal to the charge of the ion:
We can apply a general formula here:
(Total Cr oxidation #) + (Total O oxidation #) = net charge
Because there are 2 Cr atoms and 7 O atoms in this molecule:
2(Cr Oxidation #) + 7(O Oxidation #) = -2
2(Cr Oxidation #) + (-14) = -2
2(Cr Oxidation #) = +12
Cr Oxidation # = +12/2 = +6
Therefore, Cr oxidation number is +6.
Check your understanding:
What is the oxidation number of each element in Na2S2O3?
a) Na: +2; S: +2; O: -3
b) Na: +1; S: +2; O: -2
c) Na: +1; S: +1; O: -2
c) Na: +2; S: +1; O: -2
Click for the solution 5-1-B-1
B. Oxidation Numbers in Redox Reactions:
In a redox reaction, it is sometimes difficult to determine which reactants are oxidized and which are reduced just by looking at the chemical equation. To help with this, we can use the oxidation numbers directly in the reaction equation. It is important to note that in a redox reaction, if a species' oxidation number is lowered (or reduced) during the course of the reaction, it has been reduced. Conversely, if a species' oxidation number increases it has been oxidized. Let's use oxidation numbers in the redox reaction equation to determine what gets oxidized and what gets reduced:
Example: Determine which reactant is oxidized and which is reduced in the following reaction:
NiO(s) + C(s) → Ni(s) + CO(s)
Step 1. Determine the oxidation number of each species using our rules from the previous lesson.
a) NiO(s): Oxygen has an oxidation number of -2 and we can see from the equation that NiO has a neutral charge, therefore Ni must have an oxidation number of +2
b) C(s): this is an its elemental form, therefore the oxidation number is 0
c) Ni(s): this is its elemental form, therefore the oxidation number is 0
d) CO(s): Oxygen has an oxidation number of -2 and we can see from the equation that CO has a neutral charge, therefore C must have an oxidation number of +2
Step 2. Write the oxidation numbers over each species:
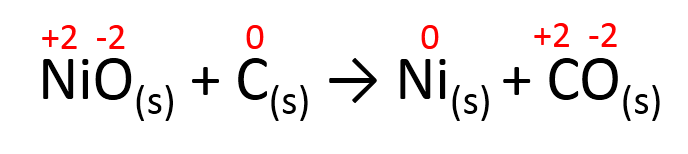
Identify which species saw their oxidation numbers change, if the oxidation number remained the same, no oxidation or reduction happened to that species.
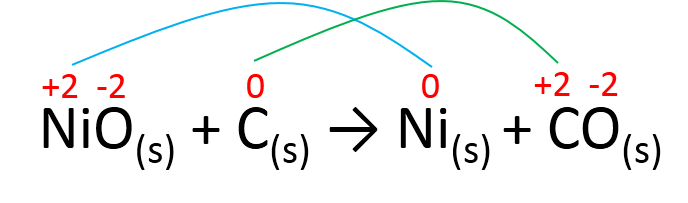
The oxidation number for Ni was lowered from +2 to 0, therefore, Ni was REDUCED. The oxidation number for C increased from 0 to +2 therefore, C was oxidized. As we said in the first learning activity, the substance that gains electrons is reduced, it is known as the OXIDIZING AGENT, whereas the substance that loses electrons is oxidized, it is known as the REDUCING AGENT.
Review Videos
The following videos from Khan academy will review some of the concepts regarding oxidation numbers:
Trends in Oxidation State:
Practice Determining Oxidation Number:
Source:
https://www.khanacademy.org/science/ap-chemistry/redox-reactions-and-electrochemistry-ap/redox-oxidation-reduction-tutorial-ap/v/practice-determining-oxidation-states
Summary
- Oxidation numbers are used to identify the degree of oxidation of a species in an oxidation-reduction reaction.
- The rules for determining oxidation numbers are as follows:
1. An oxidation number of zero is given to an atom in its elementary state.
2. The oxidation number of a monoatomic ion is the same as its charge.
3. The oxidation number for hydrogen is +1 in all compounds (except metal hydrides (metal + hydrogen) where it is -1).
4. The oxidation number for oxygen is -2 in all compounds (except peroxides, H2O2 in which it is -1).
5. All other oxidation numbers are assigned so that the sum of the oxidation numbers equals the net charge on the molecule or ion. Therefore, for neutral molecules, the net sum of the oxidation numbers must equal zero; whereas for ions the net sum of the oxidation numbers must equal the charge of the ion.
- To determine the oxidation numbers for atoms in a molecule, we first apply rules #1-4 above than we apply rule #5 for all other atoms.