SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
5-2A: Balancing Redox Reaction Equations: Oxidation Numbers Method
Description
In this activity you will learn how to balance redox reactions using the oxidation method. Within the activity are embedded practice problems to test your understanding as you progress.
Page content
A. Introduction to Balancing Redox Reaction Equations
Redox reactions, like other chemical reactions must be properly balanced to accurately display the reaction occuring. Most simple redox reactions can be simply balanced by observation and trial and error. However for more complex redox reactions there are specific strategies we can use to help us balance them. For reactions that occur in acidic or basic solutions, the hydrogen (H+) and hydroxide (OH-) ions may participate so we will need to account for them as well which we will see in this lesson. Lets begin by learning the oxidation numbers method for balancing redox reaction equations for a simple equation.
B. Oxidation Numbers Method
The oxidation numbers method for balancing redox reaction equations follows these steps:
- Write the unbalanced chemical equation from the information you are given.
- Determine the oxidation numbers for each atom in the equation
- Identify which if any of the atoms have changes in their oxidation numbers and indicate how many electrons have been gained or lost for each.
- Make the total increase in oxidation number equal to the total decrease in oxidation number by using appropriate coefficients.
- Balance the rest of the equation by inspection. If necessary, add H2O to balance oxygen and balance hydrogen by add H+ or OH-.
- Check your answer and write the completed balanced equation
Problem 1: Balancing Redox Reactions in Neutral Conditions:
Use the oxidation numbers method to balance the following reaction: Fe2O3 + CO(g) → Fe(s) + CO2(g)
Step 1. Write the unbalanced chemical equation from the information you are given.
Fe2O3(s) + CO(g) → Fe(s) + CO2(g)
Step 2. Determine the oxidation numbers for each atom in the equation
Step 3. Identify which if any of the atoms have changes in their oxidation numbers and indicate how many electrons have been gained or lost for each.
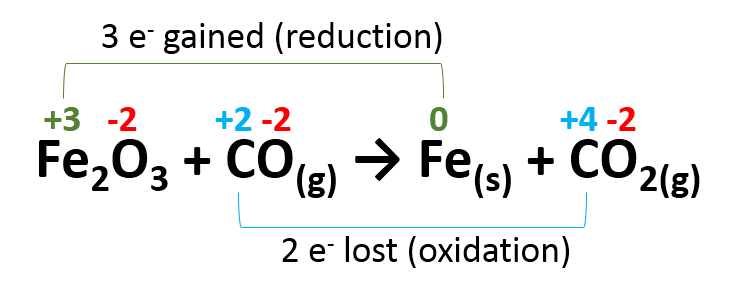
Note that the oxidation number is given per atom. So in Fe2O3, each Fe atom has an oxidation number of +3, but the combined charge from them will be +6. As we see with the green line above, Fe oxidation number changes from +3 to 0, this change in oxidation state is for each Fe atom in the Fe2O3 molecule.
In this reaction each carbon atom loses 2 electrons in an oxidation reaction and each Fe atom gains 3 electrons in reduction.
Step 4. Make the total increase in oxidation number equal to the total decrease in oxidation number by using appropriate coefficients.
In this example, the oxidation number increase should be multiplied by 3 ((+2) x 3 = 6) and the oxidation number decrease should be multiplied by 2 ((-3) x 2 = -6), this will give us a total increase of +6 and a total decrease of -6. To accomplish this, we will put the coefficient 3 in front of the carbons on both sides of the equation and the coefficient 2 in front of the Fe(s) on the right side of the equation. We don't need to put the coefficient 2 in front of Fe2O3 because the formula already tells us there are 2 Fe atoms.
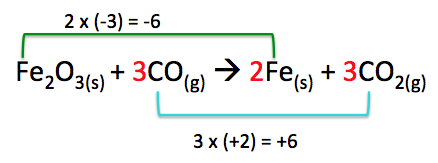
Step 5. Balance the rest of the equation by inspection.
At this point our equation is balanced so no further balancing is required. Therefore our final equation is:
Fe2O3(s) + 3CO(g) → 2Fe(s) + 3CO2(g)
Check your understanding:
Balance the following equation using the oxidation numbers method:
KMnO4(s) + HCl(aq) → MnCl2 + Cl2 + KCl + H2O
Click for the solution 5-2A-1.
D. Summary:
- When balancing redox reactions, the electrons being gained or lost must also be balanced
- The oxidation number method balances reaction equations by balancing the change in oxidation numbers on either side of the reaction arrow
- Steps for balancing redox reactions using the oxidation numbers method:
- Write the unbalanced chemical equation from the information you are given.
- Determine the oxidation numbers for each atom in the equation
- Identify which if any of the atoms have changes in their oxidation numbers and indicate how many electrons have been gained or lost for each.
- Make the total increase in oxidation number equal to the total decrease in oxidation number by using appropriate coefficients.
- Balance the rest of the equation by inspection. If necessary, add H2O to balance oxygen and balance hydrogen by add H+ or OH-.
- Check your answer and write the completed balanced equation