SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
5-2B: Balancing Redox Reaction Equations: Half Reaction Method
Description
In this activity you will learn how to balance redox reactions in both acidic and basic solutions using the half-reaction method.
Page content
A. Introduction to Balancing Redox Reaction Equations
Redox reactions, like other chemical reactions must be properly balanced to accurately display the reaction occuring. Most simple redox reactions can be simply balanced by observation and trial and error. However for more complex redox reactions there are specific strategies we can use to help us balance them. For reactions that occur in acidic or basic solutions, the hydrogen (H+) and hydroxide (OH-) ions may participate so we will need to account for them as well which we will see in this lesson. In this activity we will learn how to balance redox reaction equations using the Half-Reaction Method.
B. Half-Reaction Method
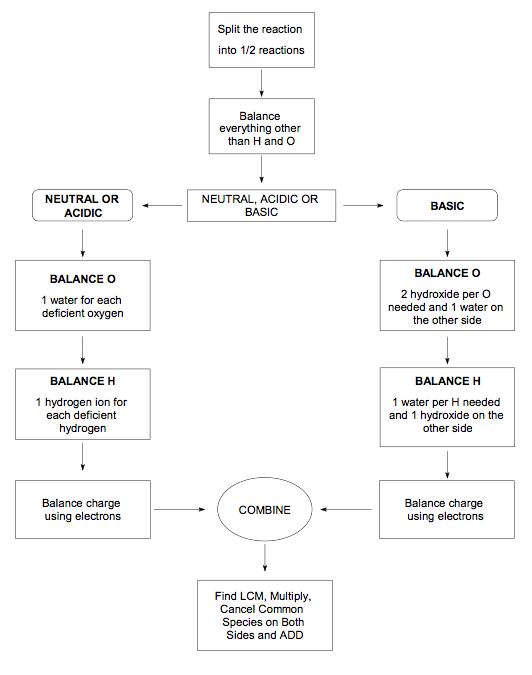
An alternative to the oxidation numbers method for balancing redox reaction equations is to use the half-reactions of the full equation. In this method, you balance each half-reaction separately and then combine them into the full balanced redox reaction equation.
- Write the unbalanced equation in ionic form
- Write the separate half-reactions for oxidation and reduction
- Balance elements other than hydrogen and oxygen by changing the coefficients in each half reaction.
- Balance the oxygen atoms by adding water molecules.
- Balance the hydrogen atoms by adding hydrogen ions (H+). If the reaction takes place in a basic solution, add an equal number of hydroxide ions (OH-) on each side of the reaction as the number of H+ ions that were added. The OH- and H+ that are on the same side of the equation will bond to form H2O.
- Balance the charges of each reaction by adding electrons.
- Balance the half-reactions so that each one has the same number of free electrons
- Add the 2 reactions together and cancel out any identical species on opposite sides of the arrow. Check your answer to see if you produced a fully balanced equation
Example:
Balance the following reaction, that takes place in an acidic solution, using the half-reaction method:
MnO4-(aq) + Fe2+(aq) → Mn2+(aq) + Fe3+(aq)
Step 1. Write the unbalanced equation in ionic form.
MnO4-(aq) + Fe2+(aq) → Mn2+(aq) + Fe3+(aq)
Step 2. Write the separate half-reactions for oxidation and reduction
MnO4-(aq) → Mn2+(aq) = Reduction (oxidation number of Mn reduced from +7 to +2)
Fe2+(aq) → Fe3+(aq) = Oxidation (oxidation number of Fe increased from +2 to +3)
Step 3. Balance the elements other than hydrogen and oxygen by changing the coefficients in each half reaction.
Both half reactions are balanced for elements other than oxygen or hydrogen.
Step 4. Balance the oxygen atoms by adding water molecules
MnO4-(aq) → Mn2+(aq) + 4H2O(l)
*Fe oxidation is already balanced
Fe2+(aq) → Fe3+(aq)
Step 5. Balance the hydrogen atoms by adding hydrogen ions (H+)
8H+(aq) + MnO4-(aq) → Mn2+(aq) + 4H2O(l)
*Fe oxidation is already balanced
Fe2+(aq) → Fe3+(aq)
Step 6. Balance the charges of each reaction by adding electrons.
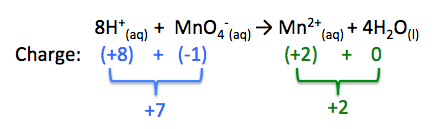
Looking at our half-reaction above, the left side of the equation has a net charge of +7, while the right side of the half-reaction has a net charge of +2. Therefore, to balance the charge we must add a total charge of -5 to the left side of the equation. Remember, each electron has a charge of -1, therefore we must add 5 electrons to the left side of the equation to balance the charge:
5e- + 8H+(aq) + MnO4-(aq) → Mn2+(aq) + 4H2O(l)
Now we balance the charges on the oxidation half-reaction:
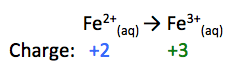
Looking at this half-reaction, we need to reduce the positive charge on the right by 1, therefore we will add 1 electron to the right side:
Fe2+(aq) → Fe3+(aq) + 1e-
Step 7. Balance the half-reactions so that each one has the same number of free electrons:
The reduction half-reaction has 5 electrons, while the oxidation half-reaction only has 1 electron. Therefore, we must multiply the entire oxidation half-reaction by 5 to balance out the number of electrons.
5Fe2+(aq) → 5Fe3+(aq) + 5e-
Step 8. Add the 2 reactions together and cancel out any identical species on opposite sides of the arrow. Check your answer to see if you produced the full balanced equation:
5e- + 8H+(aq) + MnO4-(aq) + 5Fe2+(aq) → Mn2+(aq) + 4H2O(l) + 5Fe3+(aq) + 5e-
We cancel out the 5 electrons on either side of the arrow giving us our fully balanced equation:
8H+(aq) + MnO4-(aq) + 5Fe2+(aq) → Mn2+(aq) + 4H2O(l) + 5Fe3+(aq)
Check your understanding:
Balance the following equation using the half-reaction method, that takes place in an acidic environment:
P(s) + Cu2+(aq) → Cu(s) + H2PO4-(aq)
For another view on how to balance redox reactions using the half-reaction method, click here.
The following videos review these methods for balancing redox reactions using the half-reaction method
Review
Balancing Redox Reactions Using the Half-reaction Method (in acidic solution)
Balancing Redox Reactions Using the Half-reaction Method (in basic solution):
D. Summary:
- When balancing redox reactions, the electrons being gained or lost must also be balanced.
- The half-reaction method of balancing equations breaks the full redox reaction down into its respective half-reactions and than balances each, followed by a balancing of the total number of electrons.
- Steps to balance redox reactions by the half-reaction method:
- Write the unbalanced equation in ionic form.
- Write the separate half-reactions for oxidation and reduction.
- Balance elements other than hydrogen and oxygen by changing the coefficients in each half reaction.
- Balance the oxygen atoms by adding water molecules.
- Balance the hydrogen atoms by adding hydrogen ions (H+). If the reaction takes place in a basic solution, add an equal number of hydroxide ions (OH-) on each side of the reaction as the number of H+ ions that were added. The OH- and H+ that are on the same side of the equation will bond to form H2O.
- Balance the charges of each reaction by adding electrons.
- Balance the half-reactions so that each one has the same number of free electrons.
- Add the 2 reactions together and cancel out any identical species on opposite sides of the arrow. Check your answer to see if you produced the full balanced equation.