SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
5-2C: Summary
This page is part of the content downloaded from 5-2C: Summary on Monday, 3 March 2025, 9:20 AM. Note that some content and any files larger than 50 MB are not downloaded.
Description
Summary
Balncing Redox reactions using the oxidation numbers method
- When balancing redox reactions, the electrons being gained or lost must also be balanced
- The oxidation number method balances reaction equations by balancing the change in oxidation numbers on either side of the reaction arrow
- Steps for balancing redox reactions using the oxidation numbers method:
- Write the unbalanced chemical equation from the information you are given.
- Determine the oxidation numbers for each atom in the equation
- Identify which if any of the atoms have changes in their oxidation numbers and indicate how many electrons have been gained or lost for each.
- Make the total increase in oxidation number equal to the total decrease in oxidation number by using appropriate coefficients.
- Balance the rest of the equation by inspection. If necessary, add H2O to balance oxygen and balance hydrogen by add H+ or OH-.
- Check your answer and write the completed balanced equation
Page content
Summary
Balncing Redox reactions using the oxidation numbers method
- When balancing redox reactions, the electrons being gained or lost must also be balanced
- The oxidation number method balances reaction equations by balancing the change in oxidation numbers on either side of the reaction arrow
- Steps for balancing redox reactions using the oxidation numbers method:
- Write the unbalanced chemical equation from the information you are given.
- Determine the oxidation numbers for each atom in the equation
- Identify which if any of the atoms have changes in their oxidation numbers and indicate how many electrons have been gained or lost for each.
- Make the total increase in oxidation number equal to the total decrease in oxidation number by using appropriate coefficients.
- Balance the rest of the equation by inspection. If necessary, add H2O to balance oxygen and balance hydrogen by add H+ or OH-.
- Check your answer and write the completed balanced equation
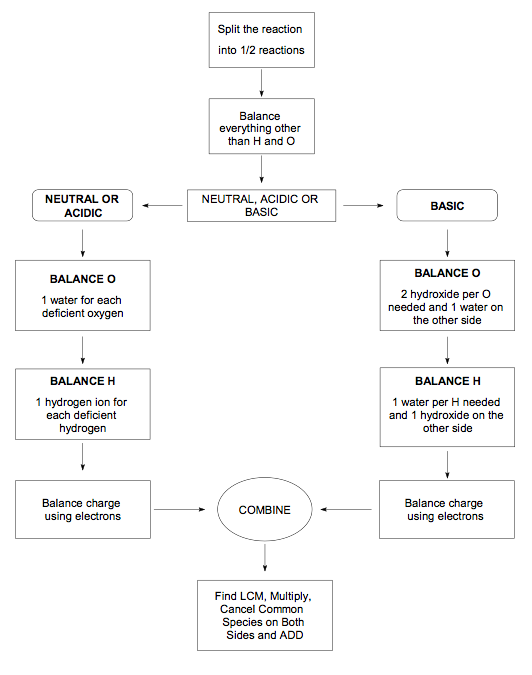
Balncing Redox reactions using the half-reaction method
- When balancing redox reactions, the electrons being gained or lost must also be balanced
- The half-reaction method of balancing equations breaks the full redox reaction down into its respective half-reactions and than balances each, followed by a balancing of the total number of electrons.
- Steps to balance redox reactions by the half-reaction method
- Write the unbalanced equation in ionic form
- Write the separate half-reactions for oxidation and reduction
- Balance the oxygen atoms by adding water molecules
- Balance the hydrogen atoms by adding hydrogen ions (H+). If the reaction takes place in a basic solution, add an equal number of hydroxide ions (OH-) on each side of the reaction as the number of H+ ions that were added. The OH- and H+ that are on the same side of the equation will bond to form H2O.
- Balance the charges of each reaction by adding electrons.
- Balance the half-reactions so that each one has the same number of free electrons
- Add the 2 reactions together and cancel out any identical species on opposite sides of the arrow. Check your answer to see if you produced the full balanced equation