SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
5-3C: Cells & Batteries
Description
In this activity you will learn about how the electrical current produced in a Galvanic cell (or other types of cells) can be grouped together to form portable sources of electrical energy known as batteries.
Page content
Cells & Batteries
As you've seen, galvanic cells can be designed to produce an electrical current using the chemistry of redox reactions. Cells such as these that create an electrical current can be grouped together in a series to create what is called a battery.
A battery is simply two or more galvanic cells that are connected in a series. When cells are connected in a series, they are wired in such a way that the anode (negative terminal) of one cell is connected to the cathode (positive terminal) of the next cell. The electrons will only begin to flow through the cells when a device that can draw electrical energy is attached to one of the terminals. This prevents the battery from draining when it is not being used.
The following video from ed.ted.com explains how electrochemistry is used to provide transportable power in the form of batteries. Below the video is an interactive presentation that will explain how electrochemical cells can be grouped to form a battery.
Source: ed.ted.com
The following will explain how electrochemical cells can be grouped to form a battery.
Intro
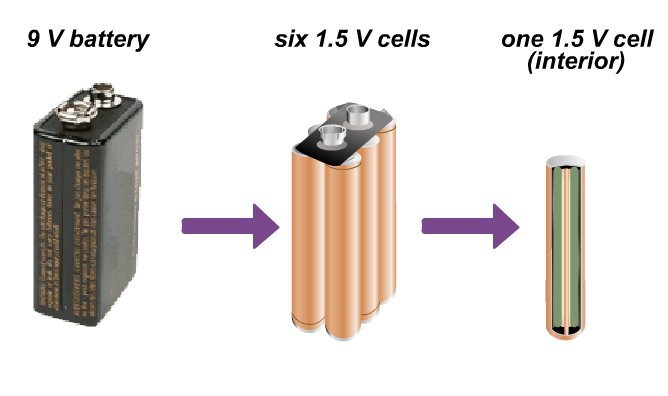
Technically, a battery is a group of two or more electrochemical cells that are connected as one unit. Cells are often wired together to increase the total voltage that is produced. For instance, a 9V (volt) smoke detector battery is really made up of six 1.5V cells that are connected together and then packed inside the familiar rectangular case.
Don't be confused by our everyday use of the word battery: common 1.5V cells (AAA, AA, C, and D sizes) are often called batteries, and they are sometimes labeled as such by the manufacturer, even thoguh they are really single cells.
What is an electrochemical cell?
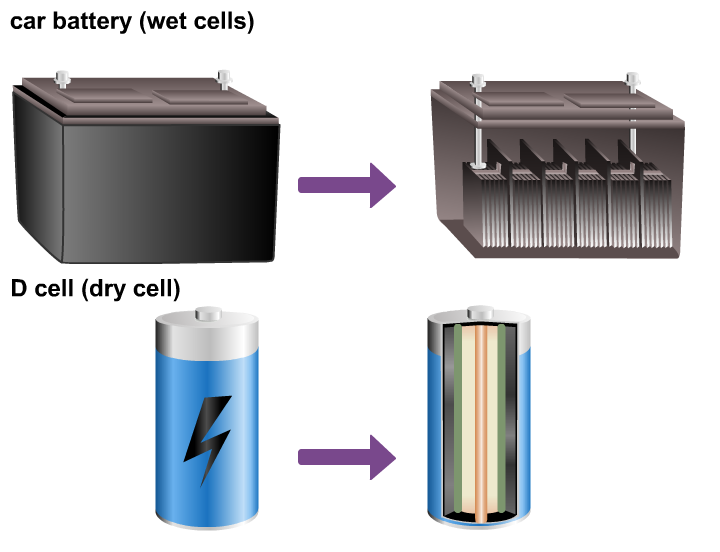
An electrochemical cell changes chemical energy to electrical energy. There are two types of cells: wet cells and dry cells.
A wet cell is sealed shut; the solution inside is a liquid that can leak or spill out. For example, car batteries are constructed of six connected wet cells, and they must be kept upright to avoid leakage of the corrosive acid solution inside.
Most other commercial cells and batteries are dry cells. A dry cell is sealed inside a container so that it can be used in any position without leaking. To make it even safer and more portable, the solution inside is made into a thick paste.
Review
Electrochemical cells are systems involving electrodes and electrolytes that provide portable electrical energy. This electrical energy is produced inside the cell when chemical energy is changed to electrical energy.
Copyright © 2005 Alberta Education
Explore Further
The following resource gives some basic information on how batteries work use the "page" arrows in the top left of the page to navigate, ending on page 1d.
Click here to access the resource directly from the source: schoolscience.co.uk
