SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
**Graphing the Periodic Table Group Activity 1-3
This page is part of the content downloaded from **Graphing the Periodic Table Group Activity 1-3 on Monday, 3 March 2025, 9:20 AM. Note that some content and any files larger than 50 MB are not downloaded.
Page content
Graphing the Periodic Table Group Activity
- In groups have students answer the following questions:
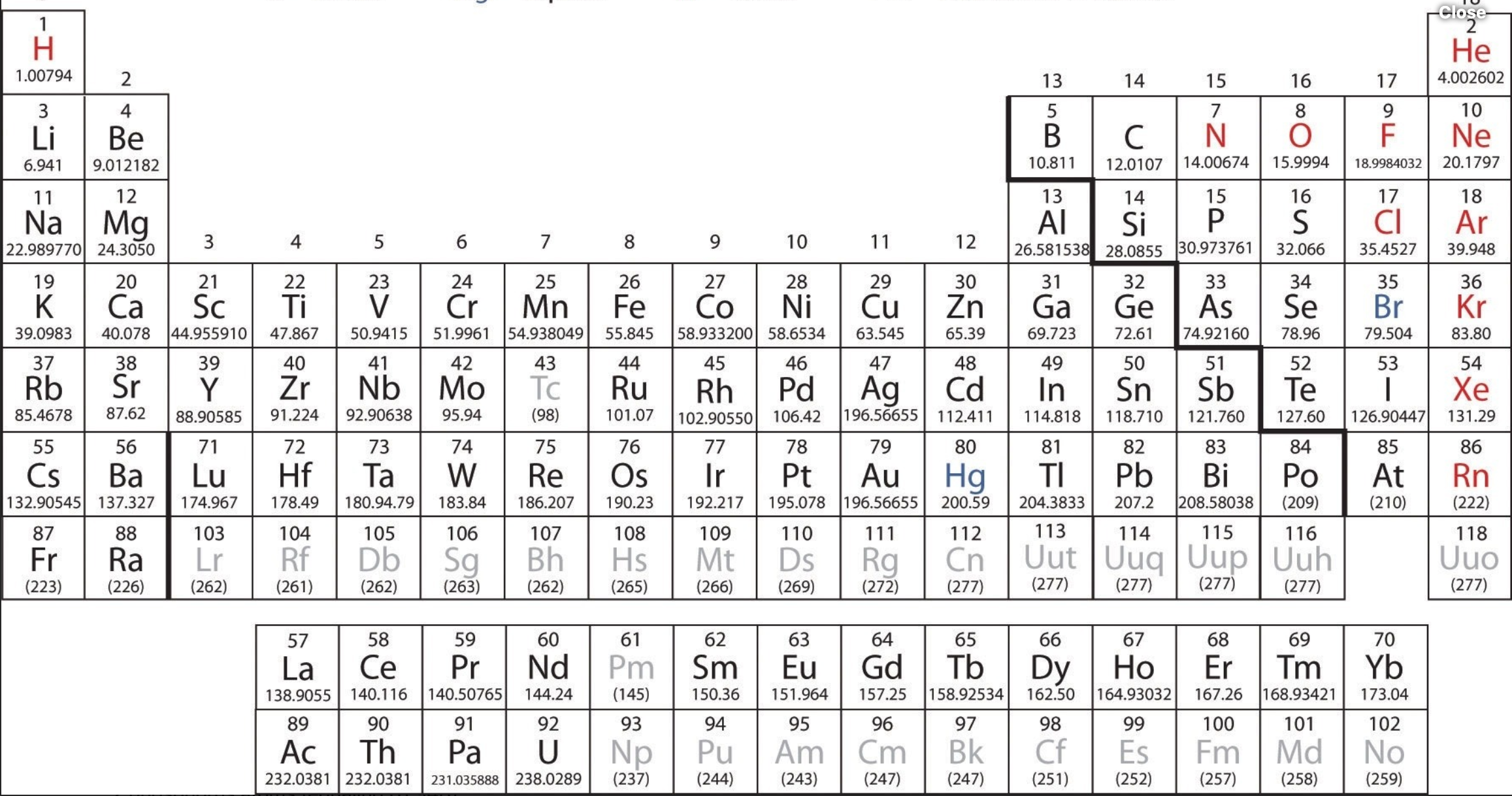
- Explore molar mass versus atomic number. Why do the elements become heavier as the atomic number increases?
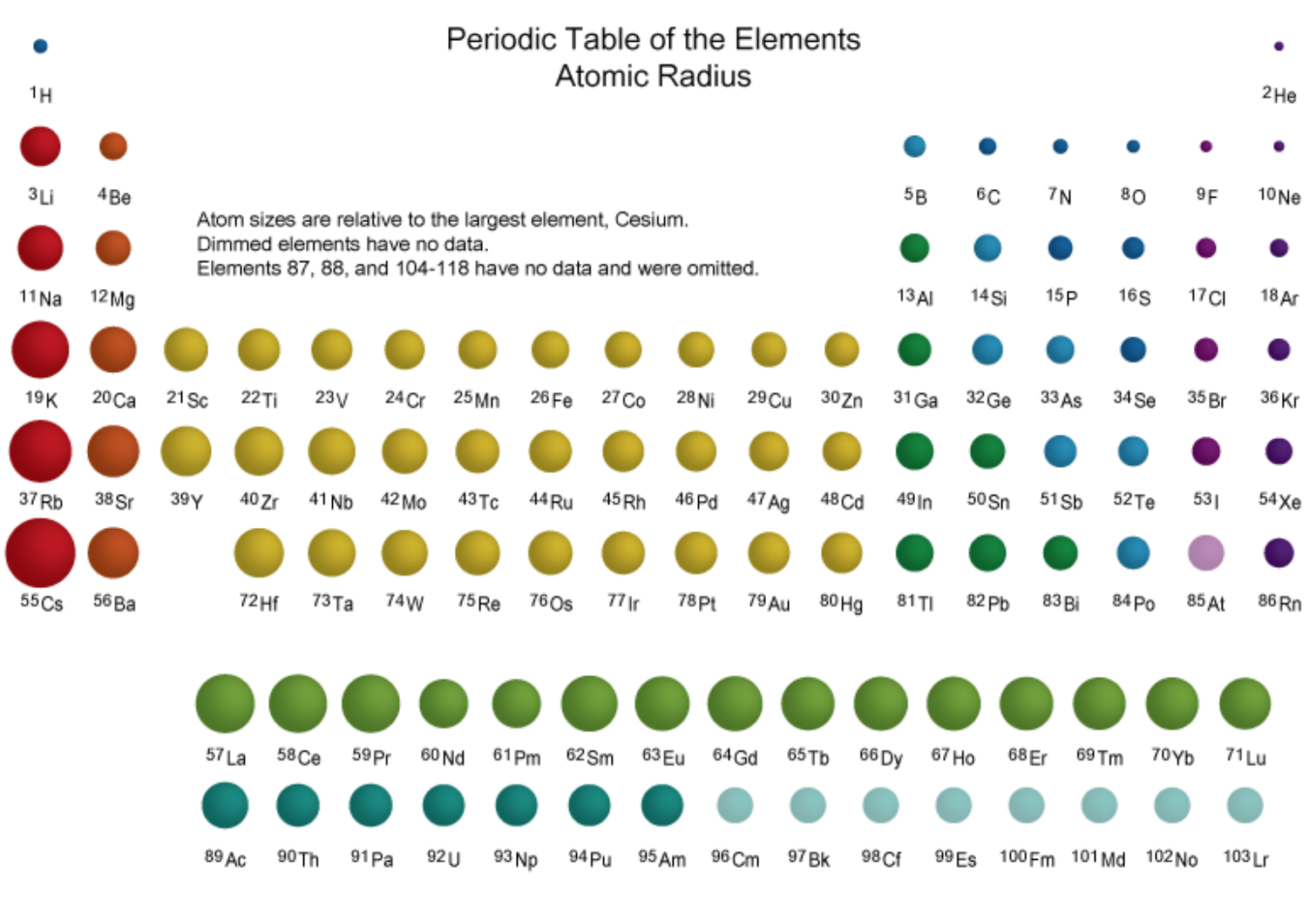
- Next, look at atomic radius. What patterns do you notice? What do you think causes these patterns? For example, why is F smaller than Li?
- Is there a relationship between atomic radius and ionization energy (listed under the elements)?
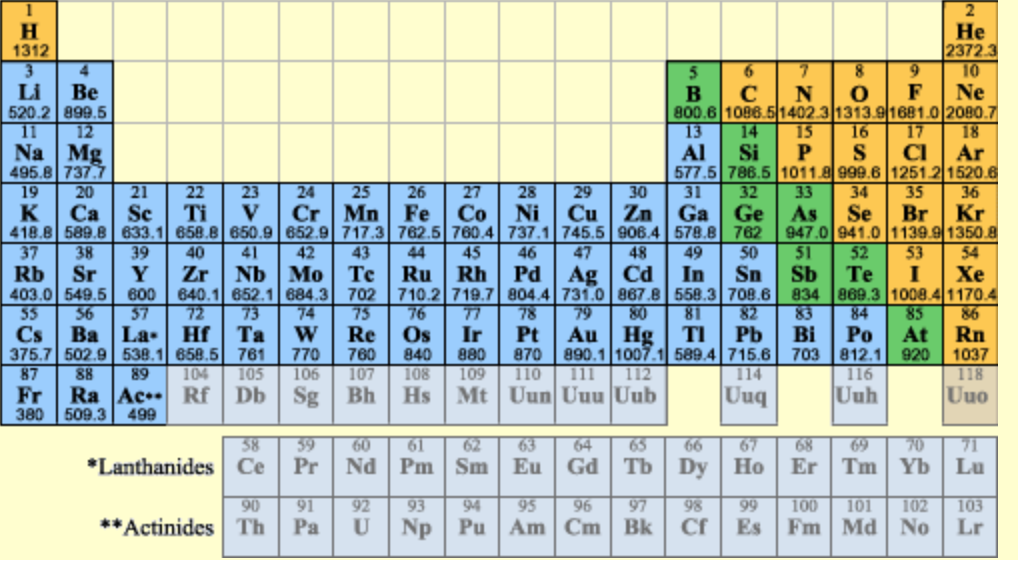
- Look the Alkali Metals labels. What properties peak in these elements? What properties are minimized in these elements?
- How does the arrangement of elements in the periodic table enable the user to identify an unknown element?
2. Have groups record their answers.
3. Each group will present their responses and engage in a class discussion.