SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
Review: Law of Conservation of Mass
Description
In this review activity you will explore the law of conservation of mass through simulated experiments.
Page content
The Law of Conservation of Mass
Use this worksheet to work through the following lab.
Problem
When you go camping an have a fire to roast marshmallows, what happens to the wood at the end of the night? It burns and we call this a chemical change. However, the next morning there is less material in the fire pit than the night before. What happened to all of the wood?

In a chemical reaction, is the mass of the products equal to the mass of the reactants? What do you think?
Please fill in your hypothesis on your worksheet before you continue.
Materials Needed

Procedure

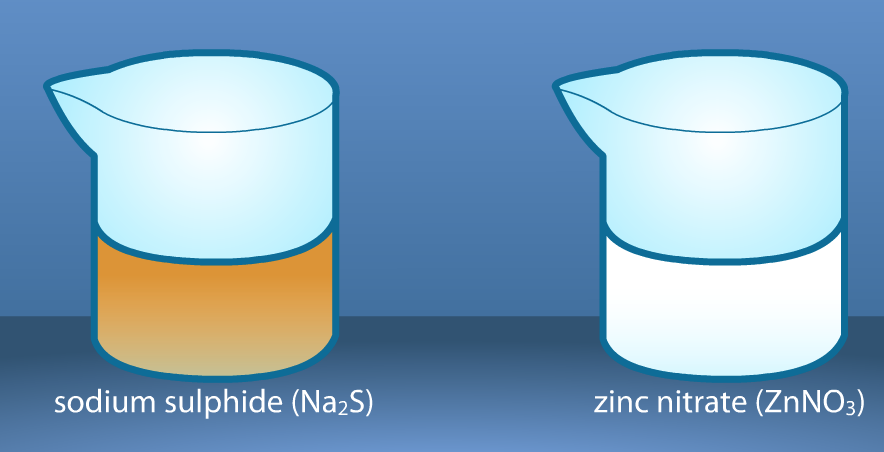
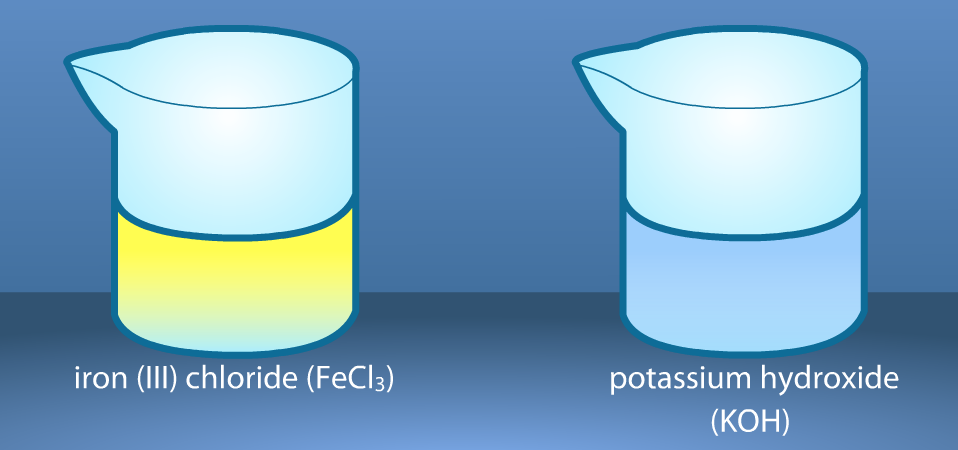
The crystals have been dissolved in water. Notice the color of the solutions. Record the description of the reactants in the observation chart on your worksheet.
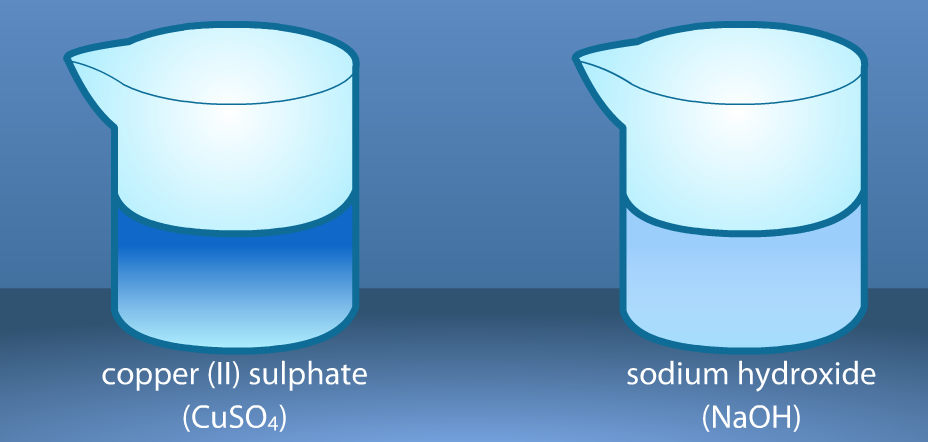
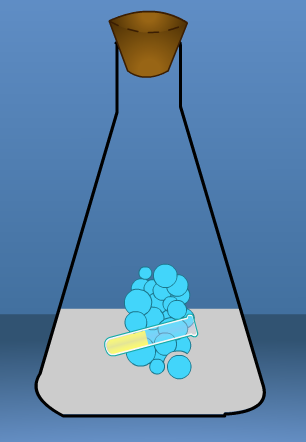
We will pour a small amount of one solution in the flask and a small amount of the other solution in a test tube. Then, we will put the test tube in the flask with a rubber stopper and find the mass of the solutions before mixing. Record the results.

Next, we'll tip the flask to mix thesolutions. We keep swirling to allow the reaction to take place. The color has changed so we know the reaction has taken place. Record your observation in the chart under the Description of the Reaction and Products.

Next, lets measure and record the mass of the flask. Record the results under Mass of Products.

Let's continue the same procedure with the next two reactions.