SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
**Where Does the Energy Released in Combustion Come From? Group Activity 3-1
This page is part of the content downloaded from **Where Does the Energy Released in Combustion Come From? Group Activity 3-1 on Monday, 3 March 2025, 9:20 AM. Note that some content and any files larger than 50 MB are not downloaded.
Description
n,
Page content
In this group activity you will lead a discussion to explore where the energy that is released in a combustion reaction comes from.
Instructions
This activity is meant to begin as a class (or small group) discussion about the nature of energy and then progresses to an individual and group activity where students can explore their own knowledge and understanding of energy. Through this discussion you will uncover ideas relating to the transfer of energy, stored energy and may extend the discussion to introduce the Law of conservation of energy.
- Begin the activity by brainstorming with the whole class or by breaking students up into groups and asking the leading question ‘Where does energy come from?’
- To help lead the discussion you can re-frame the question to ask students to also provide examples of energy sources.
- As the teacher write the responses out for the class to observe or have the groups record their responses using a brainstorming format.
- After the brainstorming session describe the combustion of methane or show a video of this process.
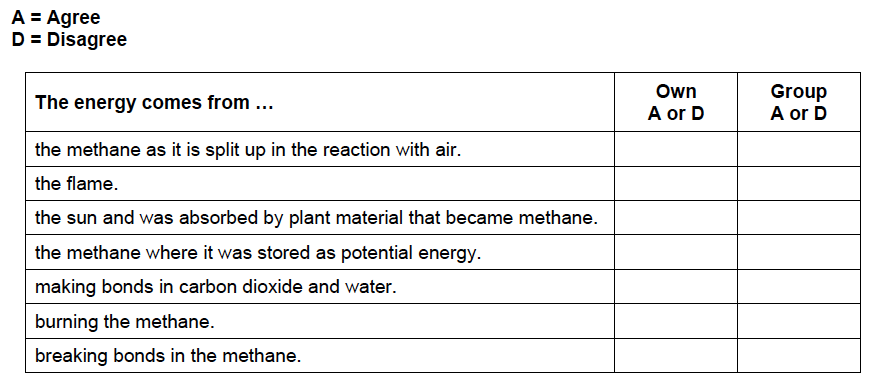
- Next handout the questionnaire below and have students answer the questions individually.
- After they have completed the questionnaire individually have students group together and discuss each of the options on the questionnaire until they come to a consensus as to whether they agree or disagree with each statement.
- Review the responses as a class.
- Ask groups to write the chemical equation for the reaction between methane and oxygen.
- Use molecular models (or ball and stick drawings or online models) to demonstrate that energy needs to be supplied to break bonds – pull the model molecules apart.
- Reform the models into carbon dioxide and water; show that the reverse process, energy release, occurs when new chemical bonds are formed. This reforming of bonds is what causes the energy to be released in the combustion of methane.
Combustion of methane reaction equation:
CH4 + O2 --> H2O + CO2
Click here to open a printable version of the questionnaire below.