SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-5A: Electronegativity & Bond Polarity
Description
In this activity you will explore the nature of bond polarity and how to use electronegativity values to predict the polar nature of a bond between two atoms.
Page content
Introduction
Atoms are made up of protons, neutrons and electrons and we've already learned that the structure of atoms is directly related to many of their characteristics and properties. In this lesson we are going to explore the characteristic of atoms known as electronegativity.
A. Electronegativity
Elements in the periodic table are a lot like humans in some ways, some like to share and some like to take. One measure of the sharing capacity of an element is known as electronegativity. Electronegativity is the ability of an atom in a molecule to attract shared electrons. Some atoms have very high electronegativity values, such as Fluorine, and they tend to hog all of the shared electrons in a molecule. Others, such as potassium, have very low electronegativity values, and they tend to give up their shared electrons rather easily. Linus Pauling, a famous chemist determined the relative electronegativity values (EN) of the elements on the periodic table and we can use these values to determine the types of bonds that form between atoms.
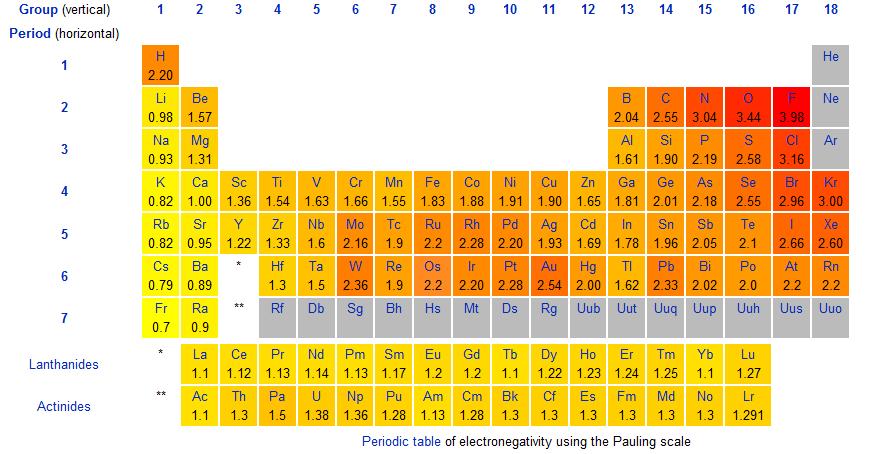
As you can see, EN increase as you move left to right and bottom-to-top across the periodic table.
We can use EN to determine the types of bonds that form between atoms by calculating the difference in electronegativity between them.
B. Ionic Bonds:
Ionic bonds form when an electron is transferred from one atom to another resulting in a positive ion and a negative ion. These types of bonds form when there is a large difference in EN. To represent the difference in electronegativity values we use the symbol ∆EN (where ∆ stands for difference). If the ∆EN between 2 atoms is greater than 1.7, an ionic bond forms. For example KCl:
∆EN = ENCl - ENk = 3.16 - 0.82 = 2.34
C. Covalent Bonds:
Covalent bonds form when 2 atoms share electron pairs between them. However, atoms do not necessarily share the electrons equally, if one of the 2 atoms has a higher EN it will tend to pull the shared electrons closer to itself. In this case the more electronegative atom will have a higher electron density and a more negative character. There are 2 types of covalent bonds that form based on polarity.
Non-Polar Covalent Bond: When ∆EN is less than 0.5 the bond is considered a non-polar covalent bond. For example an H-H bond (2 hydrogen atoms bound to each other):
∆EN = ENH - ENH = 2.2 - 2.2 = 0
The ∆EN is zero in this case, therefore the 2 atoms will share the electrons equally
Polar Covalent Bond: When ∆EN is between 0.5 and 1.7 the bond is considered polar covalent. In this scenario the 2 atoms are sharing the electrons unequally and the atom with the higher EN will pull the electrons closer to itself, and therefore have a higher electron density. Example: O-H (an oxygen atom bound to a hydrogen atom)
∆EN = ENo - ENH = 3.44 - 2.20 = 1.24
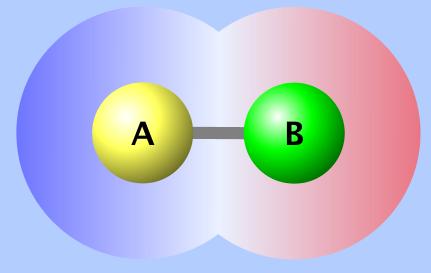
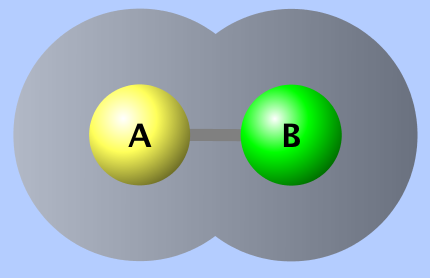
In this case the oxygen atom will pull the shared electrons closer to itself and it will have a slightly more negative character. In the images below, this polar-covalent bond is shown as a difference in charge, called the electrostatic potential (where red = negative and blue = positive) and electron density (darker = higher electron density).
Electrostatic Potential
Electron Density
Test your understanding:
Question 1-5-1. Use the periodic table above or click for Periodic Table: Electronegativity Values
Calculate the ∆EN and determine what type of bond exists between the following atoms:
a) S-H
b) Cl-H
c) Na-Cl
Answers 1-5:1
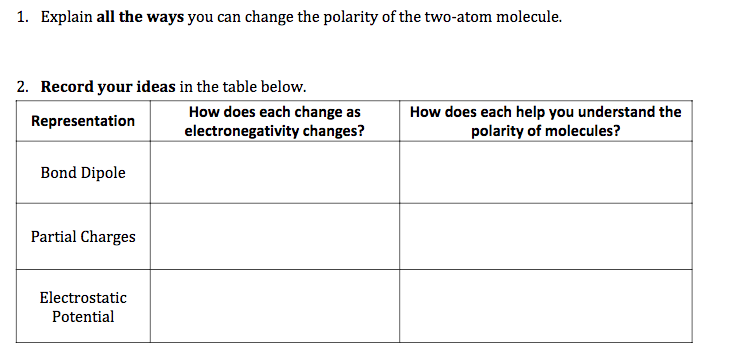
D. Polarity Simulator
Click here to open the molecular polarity simulator.
Using the Two-Atoms Screen Tab answer the following, click here to open the questions in a document that you can save.
E. Bond Polarities - Dipoles:
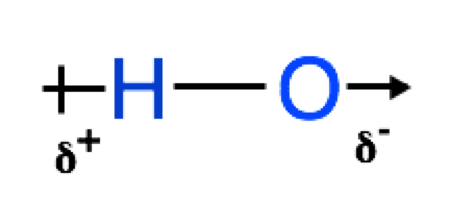
As we have seen above, 2 atoms bound to each other can have polar characteristics to them if the ∆EN is large enough. The polar quality of the bond is caused when one atom pulls the shared electrons closer to itself. When this happens, the more electronegative atom becomes more negative and the less electronegative atom becomes more positive, this creates what is known as a dipole - a separation of posititive and negative charges in space. We can represent the dipole using some common symbols: δ+ is used to represent the positive end of a dipole and δ- is used to represent the negative end of a dipole, we also use an arrow with a line at the back of it to show the difference in charge across a bond, the head of the arrow points towards the negative end of the dipole ![]() So for example if we are representing an H-O bond, we can label it as follows:
So for example if we are representing an H-O bond, we can label it as follows:
F. Summary
- Electronegativity (EN) is the ability of an atom to attract shared electrons
- The difference in electronegativity (∆EN) between 2 bonded atoms determines the nature of the bond
- ∆EN > 1.7 = ionic bond
- ∆EN < 0.5 = non-polar covalent bond
- 0.5 < ∆EN < 1.7 = polar covalent bond
- When there is a difference in electronegativity between 2 bonded atoms, the difference in polarity is called a dipole