SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
1-5B: Molecular Polarity
Description
In this activity you will learn how electronegativity and bond polarity can produce polar and non-polar molecules.
Page content
A. Introduction
We learned in the last lesson about polar bonds, this learning activity will now explore polarity in entire molecules. We have seen that bonds can be polar or non-polar and the same principal applies to polyatomic molecules. For this lesson we will apply our knowledge of electronegativity, bond polarity and VSEPR theory.
B. Molecular Polarity Investigation
Using the molecular polarity simulator below answer the following questions on a separate piece of paper or download the handout by clicking here.
- Explain any ways to change the molecular polarity of the three-atom molecule.
- How does the ABC-bond angle effect molecular polarity? (Tip: try changing the bond angle in the simulation)
- Explain the relationship between the bond dipoles and the molecular dipole.
- Can a non-polar molecule contain polar bonds? Use an example to explain your answer.
Click here to open the Molecular Polarity Simulator or click the image below.
C. Molecular Polarity Explanation
One of the most interesting polar molecules is water (H20), which as you saw in the earlier video, produces some unique effects when exposed to a magnetic field. We will use water as our example for how to determine the polarity of a molecule, which follows many of the steps we've already covered from producing Lewis Structures, to predicting shapes using VSEPR to determing bond polarity through ∆EN.
Step 1. Determine the Lewis structure for the molecule
Step 2. Determine the predicted shape of the molecule using VSEPR theory
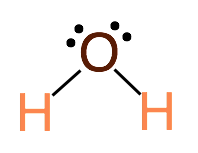
Step 3. Identify the electronegativity of each atom in the molecule and determine the partial charges and polarity of each bond. Click for the Periodic Table with Electronegativity Values.
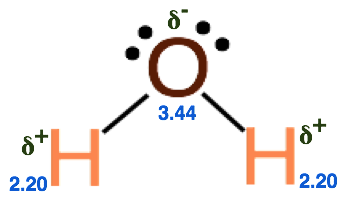
Step 4. Draw the dipoles and determine if the molecule has a net dipole.
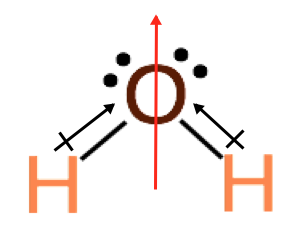 When
a molecule is polar, and therefore has a net dipole we represent this as an arrow pointing in the direction of the negative pole, which is represented by the red arrow above.
When
a molecule is polar, and therefore has a net dipole we represent this as an arrow pointing in the direction of the negative pole, which is represented by the red arrow above.
D. Non-Polar Molecules
From our drawing above we can determine that water is a polar molecule. It is important to keep in mind that molecules with dipoles aren't always polar, it is possible for dipoles to cancel each other out if they are of the same magnitude and opposite direction. For example in a linear molecule, such as CO2, the 2 dipoles end up cancelling each other out, and therefore CO2 has no net dipole and is a non-polar molecule.
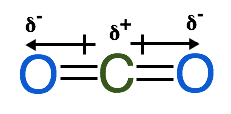
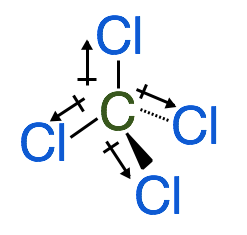
Another example of a non-polar molecule is CCl4 which forms a tetrahedral. The dipoles in this molecule are all of the same magnitude and their directions cancel each other out therefore there is no net dipole. See the image below:
Check your understanding:
Question 1-5-2: Use your knowledge of Lewis Structures, VSEPR and polarity to determine if the following molecules are polar or non-polar
For your reference: Periodic Table with Electronegativity Values.
a) NF3
b) PO4
c) H2
Answer 1-5:2
E. Summary
- Polar molecules display distinct characteristics compared to non-polar molecules
- Polyatomic molecules can be polar or non-polar depending on the polarity of the bonds within the molecule and the net dipole of the entire molecule
- To determine molecular polarity, we require Lewis Structures, electronegativity values and VSEPR theory