SCH4U - Chemistry 12 (2024-25) - A
SCH4U-25A
5-3A: Galvanic Cells
Description
In this activity you will learn about how redox reactions can be used in practical applications.
Page content
A. Galvanic Cells
Picture yourself using a battery powered device, a flashlight for example. When the switch is off, no light shines yet by simply flipping that switch, electricity starts flowing, and enough power is generated to produce light. The production of this electricity occurs in the batteries, which take advantage of the properties of redox reactions to generate the power needed. The production and transmission of electricity, or electrical power requires the movement of electrons. As we have learned in this unit already, redox reactions involve the transfer of electrons between species. By isolating paired oxidation and reduction reactions in a setup called a galvanic cell, scientists are able to harness the transfer of electrons in redox reactions to create electrical power. In the following video, you will learn how galvanic cells work and the important components of a galvanic cell. Watch the full video and then use the navigation buttons to explore all of the important components of a galvanic cell. Below the video, you will learn a method to represent galvanic cells using a simplified format known as line notation.
Click here to download a handout that will help you organize the relevant information about Galvanic cells.
Source: Youtube
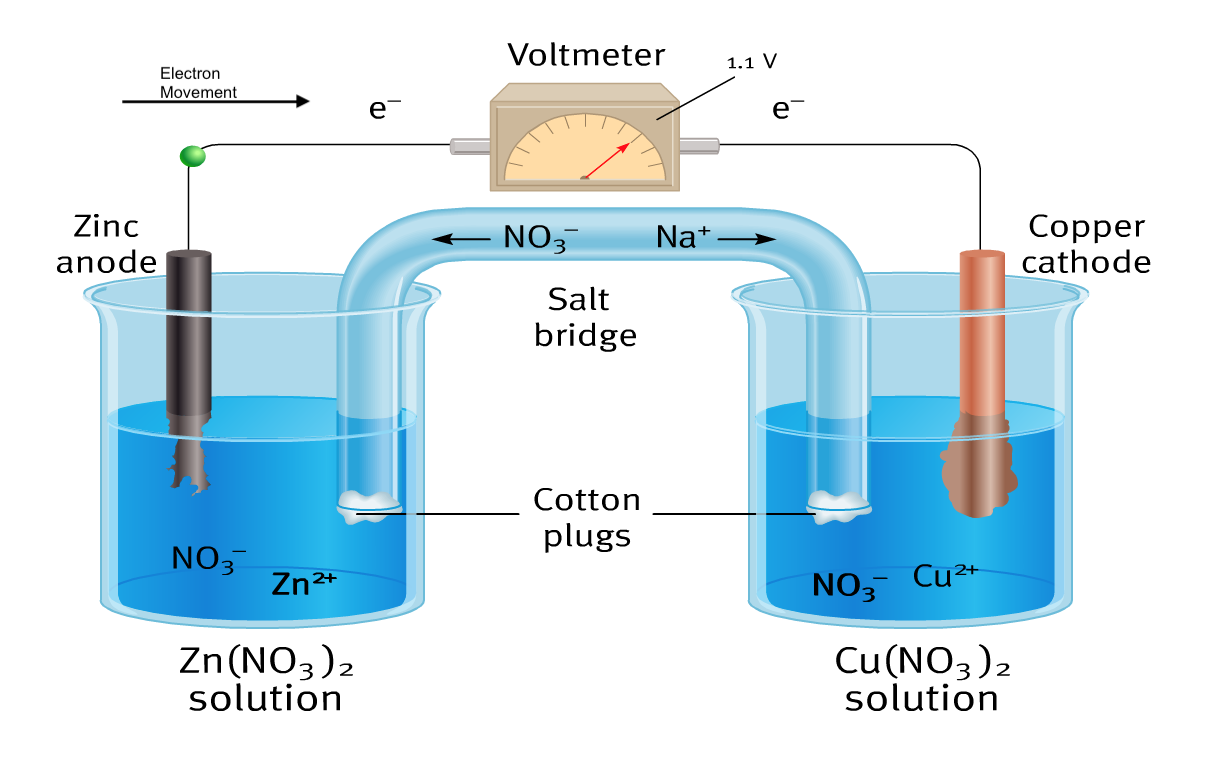
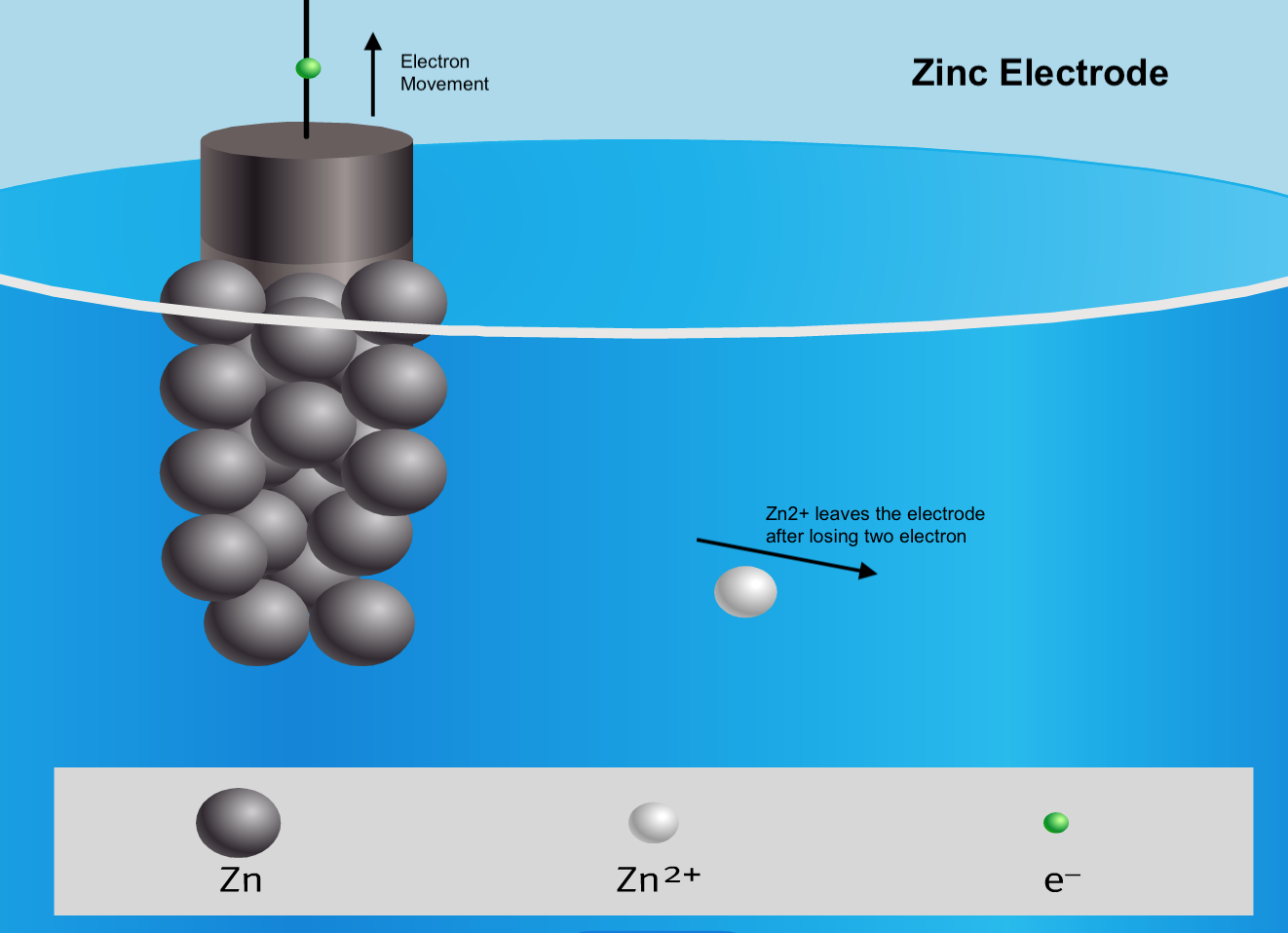
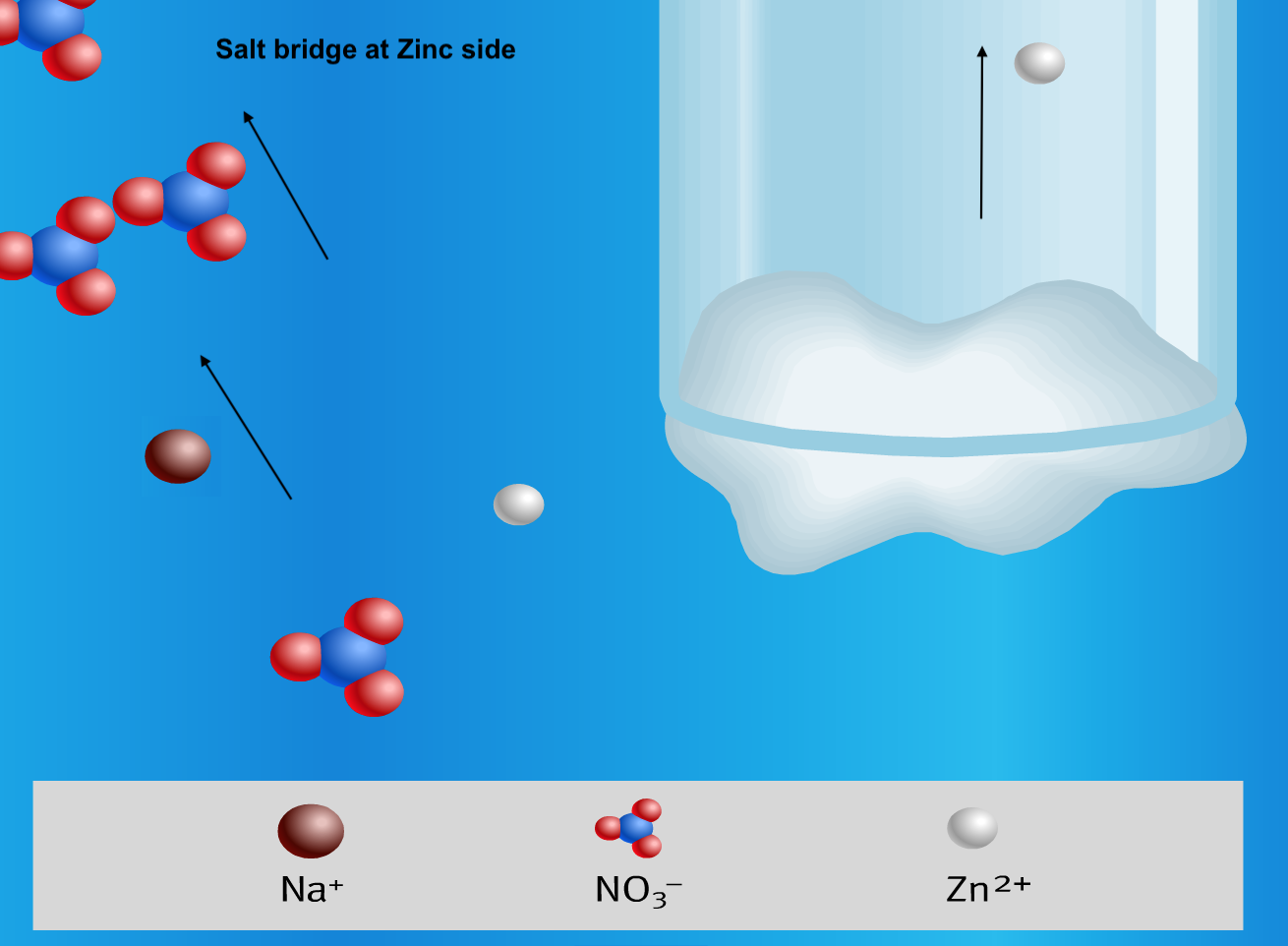
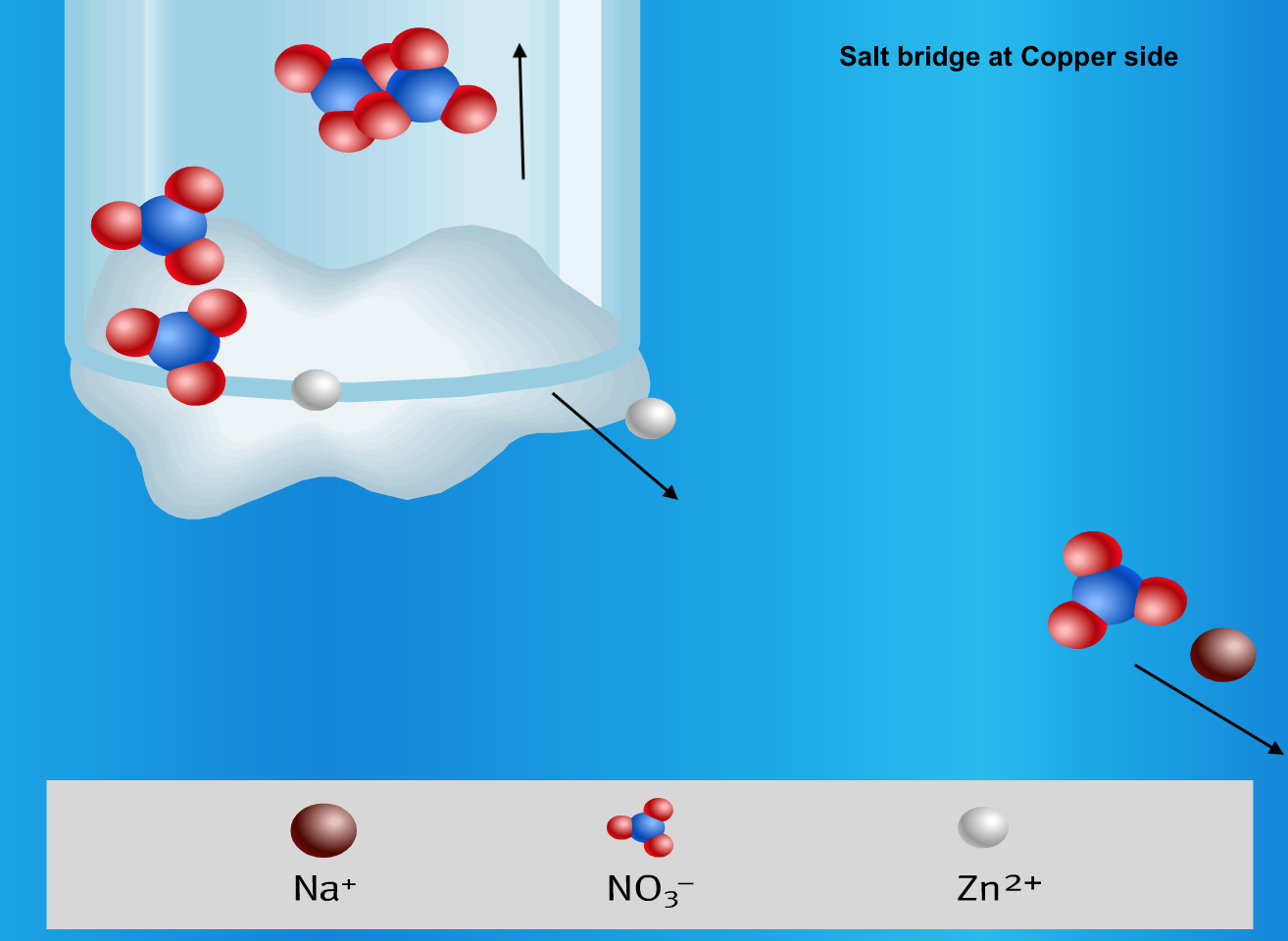
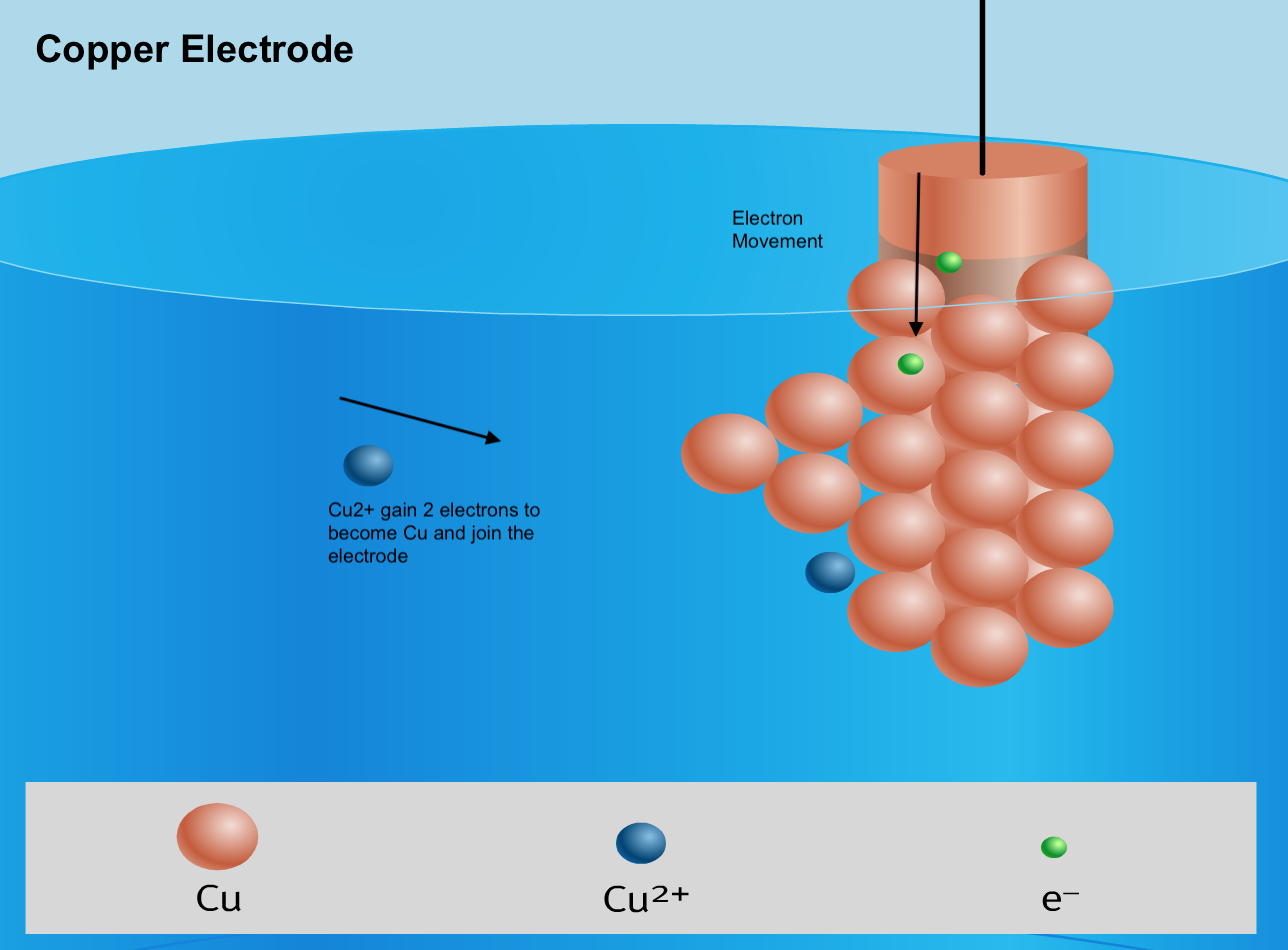
Below is a visual summary of a galvanic cell, with 4 diagrams representing what happens at the 2 electrodes and the 2 ends of the salt bridge.
Line Notation
Line notation is a simplified format for representing galvanic cells. When using line notation, the components of the anode (the ion at the anode and the ion in solution, also known as the electrolyte) are listed on the left separated from each other by a single line, the components of the cathode (the ion at the cathode and the ion in solution, also known as the electrolyte) listed on the right separated by a single line, and the cathode and anode components are separated from each other by a double line.
anode | electrolyte || electrolyte | cathode
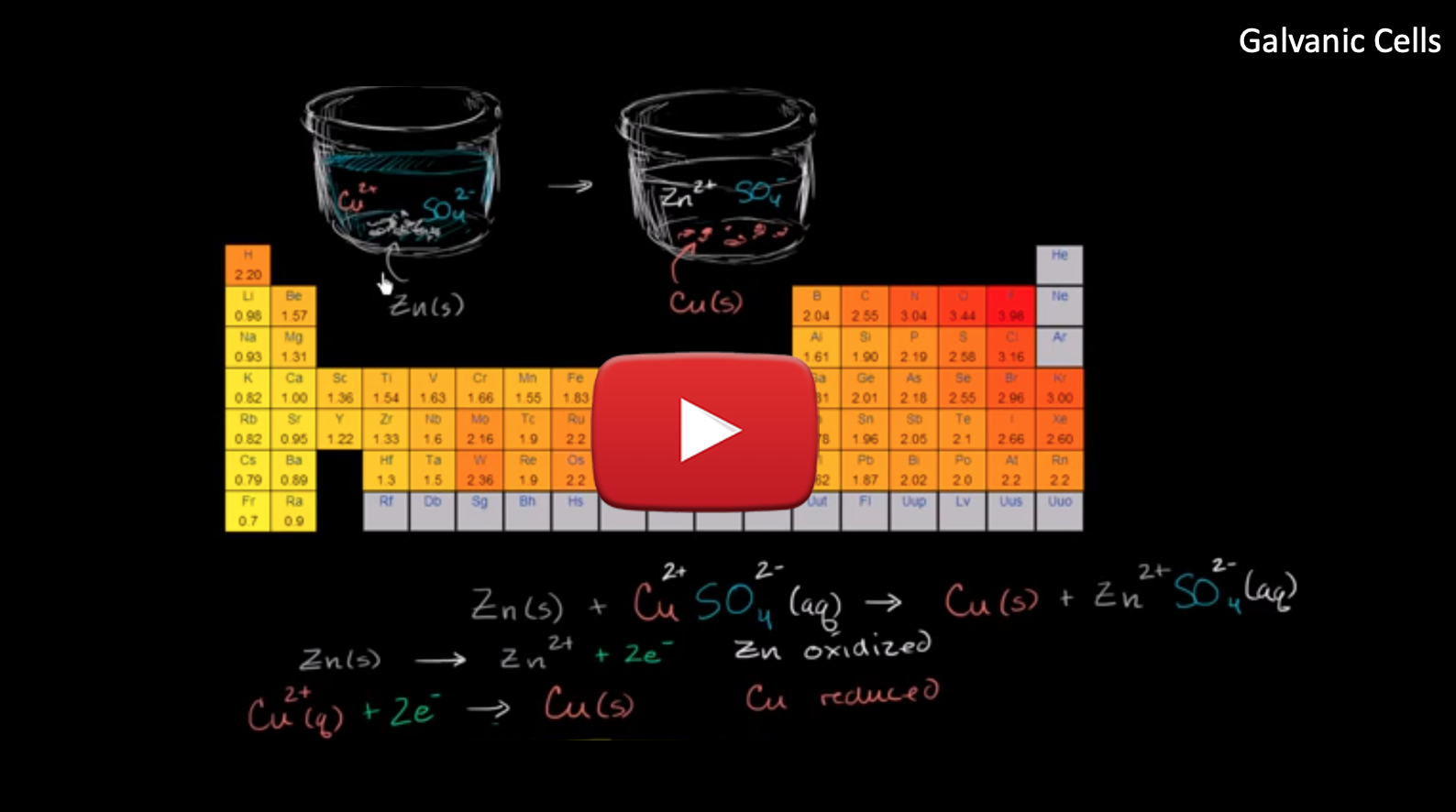
For example, using the Galvanic cell in the animation above, the net ionic equation for the reaction would be:
Zn(s) + Cu2+ (aq) --> Zn2+ (aq) + Cu(s)
Using line notation, this galvanic cell would be written as:
Zn(s) | Zn2+ (aq) || Cu2+ (aq) + Cu(s)
NOTE: To write the line notation, the anode and cathode are identified based on the information given about the reactions. The line notations reflect the actual chemical reactions occurring, not necessarily the diagram.
Review:
The following animation will review important information about Galvanic cells.
B. Summary
- The goal of a galvanic cell is to produce electricity through the movement of electrons.
- A galvanic cell is made up of 2 half-cells, in one half-cell, a reduction reaction takes place while in the other half-cell an oxidation reaction takes place.
- The two half-cells of a galvanic cell are connected externally by a metal wire through which electrons move, this external wire is often connected to a voltmeter or a component that requires electricity.
- The two half-cells are connected internally by a salt bridge that functions to maintain electrical neutrality in each half cell. The salt bridge is made up of a tube that is filled with an electrolyte solution such as KNO3 or KCl.
- Each half-cell is composed of a strip of metal (the electrode) immersed in a salt solution that contains ions made up of the same element as the metal strip.
- The half-cell where reduction takes place is called the cathode.
- The half-cell where oxidation takes place is called the anode.